Abstract
Objectives: To report early experience with the advanced breast biopsy instrumentation (ABBI) system and to compare the results with those of other published studies.
Design: A nonrandomized case series.
Setting: An outpatient breast diagnostic centre at a large urban community hospital.
Patients: Thirty-four women; 27 had suspicious calcifications, 2 had a nonpalpable mass and 5 had both.
Intervention: The ABBI procedure to excise a breast lesion or obtain a representative sample for histologic examination.
Main outcome measures: Success of the procedure with respect to diagnosis, sample quality, technical problems, margins of tumour free tissue and patient satisfaction.
Results: Malignant tissue was diagnosed in 7 women (21%) and atypical ductal hyperplasia in 2 (6%). In all cancers, the obtained samples had malignant cells present at the margins or less than 1 mm away. Technical problems were encountered in 32% of cases. Manual extraction of the specimen was required in 21% of cases.
Conclusions: The preliminary data correlate well with those of other published results. Although it is possible that a small number of cases and a relatively high proportion of technical difficulties may represent a normal learning curve, there is a definite need for improvement of some ABBI components. ABBI does not appear to provide adequate margins of uninvolved tissue in patients with cancer and thus should not be used with curative intent. ABBI provides excellent quality samples for pathological study and good patient satisfaction. There are not yet enough data for meaningful comparison of ABBI with stereotactic core biopsy and excisional biopsy with needle localization.
Many national clinical consensus bodies have accepted mammographic screening for breast cancer as the essential strategy for reducing deaths from breast cancer.1,2 This approach inevitably led to an increase in the detection of nonpalpable radiologically suspicious or indeterminate lesions. The proper management of these lesions is still controversial. Needle-guided excisional biopsy (NLEB) has been traditionally considered the “gold standard” for the evaluation of non-palpable mammographic abnormalities. It is, however, rapidly falling out of favour because of a failure rate up to 22%, high cost, patient dissatisfaction with the 2-step intervention and poor cosmetic results.3–5 Stereotactic core biopsy (SCB) has an excellent record of sensitiving ranging from 71% to 100% and specificity ranging from 96% to 100%.6 However, multiple passes are required to achieve that degree of accuracy,7,8 and they may significantly alter the tissue, resulting in a false-negative rate of up to 5.4% 9 and a technical failure of up to 6%.10 When a subsequent definitive procedure is required, the identification of the lesions may be difficult.11 Finally, there is some evidence for cancer dissemination as a direct result of the procedure.12
The advanced breast biopsy instrumentation (ABBI) system was developed in Canada in 1995. ABBI can be readily performed under local anesthesia on an outpatient basis. The ABBI gun can excise tissue fragments up to 20 mm in diameter and 20 to 80 mm long with the precision of the stereotactic technique. This provides excellent, reliable samples for histologic examination.
The indications for, statistical characteristics and outcomes of, ABBI procedures have been poorly reported. There are only 3 publications to date describing experiences with ABBI in the United States, yet already ABBI is a subject of heated debate. The first Ontario ABBI procedure was performed at North York General Hospital, Toronto, on Sept. 16, 1997. We present our preliminary experiences with this technique and compare our data with the other published results.
Patients and method
The ABBI procedure was offered to all 34 patients with suspicious nonpalpable mammographic lesions, which were deemed accessible by this technique. All candidates were screened for contraindications to the procedure: inability to tolerate a prone position for 30 minutes because of back or neck problems; extreme obesity; chronic obstructive pulmonary disease; a breast compressible to less then 2 cm; lesion less than 1 cm from the surface, too close to the nipple or in the axillary tail; and patients taking coumarin. Patients taking acetylsalicylic acid were requested to stop intake 1 week before the procedure. No preoperative laboratory investigations were required.
All procedures were performed at the Breast Diagnostic Centre at North York General Hospital by surgeons and specially trained technicians in the presence of experienced breast radiologists. The majority of the procedures were performed by 2 surgeons (20 and 11 procedures respectively), 2 other surgeons under supervision did the remaining 3 procedures. In the latter 3 cases, there were no complications or technical difficulties, and no malignant lesions were found. A detailed questionnaire was completed for each case. The referring radiologic diagnosis, surgical intent, timing of the procedure, adequacy of the excision, technical problems and immediate complications were recorded promptly after the procedure.
The technique of ABBI is described in detail elsewhere.13–15 Briefly, the patient was positioned prone on a specially designed table, with her breast hanging freely through the aperture. The breast was compressed and views were taken at 15° angles, which yielded the coordinates of the target. Skin was prepared and a local anesthetic applied. A targeting needle was stereotactically advanced to the centre of the target area. A T wire was deployed to secure the position and to stabilize the tissue during the excision. An incision was made to accommodate a 0.5-cm to 2-cm cylindrical ABBI gun with an oscillating blade, which was engaged after confirmatory magnified views were taken. The blade of the gun was advanced manually through the opening to 15-mm beyond the target area and a snare wire then cut the specimen while providing simultaneous electrocoagulation. Additional hemostasis if required, was achieved by conventional electrocautery. Final scout images were obtained to document the complete removal of the abnormality, and a film of the specimen was obtained. All women were followed up in the surgeons’ offices 1 week after the procedure. Postoperative pathological findings and follow-up notes were reviewed in each case.
Results
The 34 women, aged 37 to 75 years (mean 57.6 years), underwent ABBI procedures between Sept. 16, 1997, and May 10, 1998. Twenty-seven women (79%) were referred for the investigation of calcifications, including 5 diffuse widespread calcifications and 3 multiple calcified clusters. Fifteen of those calcifications were described by a radiologist as indeterminate probability for malignancy, 7 as suspicious for carcinoma and 5 as likely benign. Two women (6%) had masses without calcifications and 4 had both, 1 of which was a palpable nodule. In addition, 1 woman had undergone stereotaxic core biopsy (SCB) for the diagnosis of a small nodule, which demonstrated a noninvasive intraductal carcinoma, subsequently excised by ABBI.
The operative intents were full excision of the lesion (28), acquisition of a representative sample (6) and wider excision of a previously diagnosed noninvasive cancer (1). The 20-mm gun was used in 31 cases, 15-mm gun was used in 2 more cases, and the 10-mm gun was used in 1 case. The mean (and standard deviation) specimen size was 17.7 (14.2) cm3. The mean (and SD) procedure time was 47 (11) minutes.
A malignant lesion was diagnosed in 7 women (21%) and atypical ductal hyperplasia in 2 (6%). Three malignant lesions were ductal carcinomas with microinvasions; 3 were intraductal carcinomas with no evidence of invasion. Out of those 6, 2 were comedo type, 2 were cribriform, 1 demonstrated mucinous metaplasia and 1 was a widespread multifocal intraductal carcinoma. The remaining specimen was a 0.9-cm invasive ductal carcinoma, grade II/III (Bloom and Richards classification) with mucinous metaplasia. There were no cases of indeterminate histopathology or inadequate samples. In 1 case, part of the radiologically targeted microcalcifications were not present in the ABBI sample and were not found in the breast postoperatively, but the pathologist deemed the specimen adequate. In 5 cases when malignancy was determined, the operative intent was “full excision of the lesion,” in another one — “wider excision of a known cancer,” and in the remaining one — “obtaining a representative sample.” In 4 malignant lesions including 3 invasive ones, the obtained samples had disease present at the margin. That constituted 57% of all malignant lesions. All of these women were referred to surgery (lumpectomy or modified radical mastectomy). In the remaining 3 cases, including 1 invasive ductal carcinoma, cancer cells were identified at 1 mm or less from the margins. Two women from the latter group were referred for NLEB and 1 woman with no invasive disease chose to be followed up by mammography.
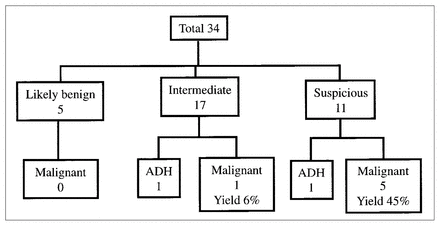
Overall, out of our 34 mammograms, 11 were deemed “suspicious” by the referring radiologists (7 calcifications and 4 masses). In that group, 5 were proven to be malignant masses on ABBI specimens, a yield of 45% (Fig. 1). Another 17 were reported initially as “indeterminate probability for cancer” (15 calcifications and 2 masses). One malignant mass was identified in this group, a yield of 6%. In the 5 cases with “likely benign” interpretation, no malignant lesions was found. The remaining case was the reexcision of a known cancer. Two cases of atypical ductal hyperplasia (ADH) were identified in each of the “indeterminate” and “suspicious” groups. Both were fully excised, and no malignant cells were noted in the specimens. No further procedure was required.
Correlation between radiologic interpretation and pathological findings of the specimens obtained with advanced breast biopsy instrumentation. ADH = atypical ductal hyperplasia.
Table I summarizes the technical problems and complications encountered during the procedures. There were difficulties in 11 (32%) patients. The most common was failure of the cautery snare to divide the remnant of the breast tissue (3 procedures). Manual extraction of the specimen was successfully completed with Metzenbaum scissors in 7 cases. In 3 cases we attributed the failure of cautery to a lower setting than the usual operating room cautery. After the settings were increased, we did not encounter any failure of hemostasis, yet in 3 more cases the snare wire had difficulty separating the specimen in dense breasts. In 4 cases the target calcifications were found at the edge of the extracted specimen. In another case target calcifications were not found on the extracted specimen at all. One lesion, 4 mm in diameter, was displaced by a small (10-mm) blade size, which required readjustment of the gun’s target settings and led us to avoid using gun blades less than 20 mm. Two women experienced discomfort related to positioning. Four patients experienced intraoperative nausea and vomiting or hypotension. One of them required sedation and an overnight admission. One woman had extensive postoperative bruising, and another experienced an anxiety attack. We encountered 1 significant infection, requiring use of antibiotics, and 1 wound dehiscence with serosanguineous discharge. Both resolved quickly. In addition, 2 women presented with uncomplicated hematomas, and 2 others complained of prolonged local pain, all of which eventually resolved.
Technical Problems Encountered During Advanced Breast Biopsy Instrumentation
Discussion
The total cost of ABBI per case is US$700.16 compared with US$1000 for needle-localization-excisional biopsy and US$400 for SCB.16 ABBI shares all the advantages of the SCB over NLEB, such as stereotactic precision, ability to be performed as an outpatient 1-step procedure and better cosmetic results. In addition ABBI has the potential to remove selected lesions in toto, resulting in better quality of the tissue samples for pathological examination, and eliminating the need for subsequent NLEB or definitive surgery. However, there is no adequate study that compares ABBI’s sensitivity and specificity in the diagnosis of breast lesions with that of SCB and NLEB. ABBI’s impact on subsequent morbidity and mortality of the target population is also unknown. Although it is not an alternative to such large-scale undertakings at the present time, we believe our short experience with ABBI technology raised some important questions, which are also reflected in previously published reports.13–15,17
Our patient sample was small, but some conclusions may be drawn. First, the amount of tissue loss is consistently smaller than that reported for NLEB, specifically 13 to 17 cm3 compared with 26 to 32 cm3.15 Second, in about 30% of the cases both we and Ferzli’s group13 encountered various technical problems. For example, in about 20% of the cases manual extraction of the specimen was necessary. Kelley and associates18 indicated that in their series of 350 ABBI procedures, they encountered less than 5% of technical difficulties: the T fastener displaced the lesion in 3 out of 350 cases, failure of the cautery snare to cut through the specimen in 4 cases, and an overall “miss rate” requiring repeat biopsy was less than 1%. In our experience, in 1 case in which we used a smaller (10-mm) blade, it displaced the target tissue, so in all the subsequent cases we elected to use guns with 20-mm blades only, pending further refinement of the technique.
The striking finding in our study was that ABBI was not able to include uninvolved margins in the large proportion of malignant lesions. In our 7 patients with malignant lesions, the margins were involved in 4 (including invasive carcinomas), and in the remaining 3 patients, malignant cells were identified 1 mm or less from the margins. Of note is that in 5 patients when malignancy was determined, the operative intent was “full excision of the lesion,” in another 1 was “wider excision of a known cancer” and in the remaining 1 was “obtaining a representative sample.” Six of the 7 women with an identified malignant lesion had to undergo a second procedure (mastectomy, lumpectomy or NLEB). Even when ABBI targeted a known malignant mass, we could not achieve margin clearance of more than 1 mm. In their initial series of 47 patients, Ferzli and associates13 reported a 30% incidence of margin involvement in the malignant specimens. However, 1 year after the initial publication, in the series of now 134 patients, Ferzli reported that malignant cells were found at the margins in 92.8% of cancers.17 In the study by D’Angelo and associates, 15 all 4 out of 23 patients with ABBI-diagnosed cancer had involved margins.18 Based on these cumulative data, we cannot advocate ABBI’s use for cure in the treatment of lesions that carry a high suspicion of malignancy or for known malignant lesions. In its present form ABBI might not spare the patient a second procedure.
The rate of clinical complications we encountered is higher than the 0.2% reported for SCB.9 However, none of the procedures were interrupted compared with a 3.5% interruption rate found in 1 of the SCB series.10
One of the indisputable advantages of ABBI is its ability to provide excellent specimens for pathological evaluation. In our series (as well as in other published results), pathologists had no difficulty in identifying and diagnosing the lesions. There were no reports of “indeterminate histopathology” or “inadequate tissue sample,” which are responsible for up to 19% rates of repeat biopsy after SCB.19,20
A finding of atypical ductal hyperplasia on SCB has been associated with invasive carcinoma or carcinoma in situ in 50% of cases.18,19,21 The present recommendations require a subsequent NLEB to be performed in such cases. However, in the 2 cases of atypical ductal hyperplasia that we encountered, all margins were clear of malignant cells, and there were no malignant cells present in the specimen. If further study confirms that ABBI is able to provide adequate clearance in women with atypical ductal hyperplasia, it will be a strong positive aspect and may define ABBI’s role versus SCB.
Conclusions
ABBI is a new exciting minimally invasive technique that provides pathological specimens of excellent quality. It is easily performed under local anesthesia on an outpatient basis and is relatively well tolerated by the patients. The amount of the tissue loss is consistently smaller than that reported for NLEB. However, the rate sof technical difficulties and malfunctions are high. At present, ABBI should not be used to completely excise a malignant lesion under the premise that it can leave clear margins.
Finally, there is a definite need for more scientific data regarding the actual indications and appropriate patient selection for the ABBI procedure. The attainment of these objectives, as well as ABBI’s improved technical quality may ultimately secure the role of this technique in the management of nonpalpable breast lesions.
Acknowledgments
We thank the staff and volunteers of the Breast Diagnostic Centre at the North York General Hospital and the radiologists, Drs. Bass, Cranstoun, MacAdam, Durrant and Yates for their assistance with the advanced breast biopsy instrumentation procedures.
Footnotes
Presented at the conference “Controversies in the Etiology, Detection and Treatment of Breast Cancer: 1998,” sponsored by the Breast Cancer Site Group of the Interdepartmental Division of Oncology, University of Toronto, Toronto, Ont., May 1998.
- Accepted September 27, 1999.