Abstract
The optimal fluid for resuscitation in hemorrhagic shock would combine the volume expansion and oxygen-carrying capacity of blood without the need for cross-matching or the risk of disease transmission. Although the ideal fluid has yet to be discovered, current options are discussed in this review, including crystalloids, colloids, blood and blood substitutes. The future role of blood substitutes is not yet defined, but the potential advantages in trauma or elective surgery may prove to be enormous.
The realization that significant blood loss must be replenished with extraneous fluids dates back several centuries. Shortly after William Harvey’s description of the process of blood circulation (circa 1628), the first successful blood transfusion was reported in dogs, and attempts to transfuse humans ensued. These early attempts, however, were not only unsuccessful but were frequently fatal because of incompatibility. This led to a search for an effective alternative, resulting in the production of physiologic saline in 1875. With the discovery of the major blood types in 1900 to 1902 and Rh factor in 1939, blood transfusion regained acceptance as a safe and effective resuscitative therapy. Over the past few decades, the increased concern over the risks of transmitting such diseases as HIV and hepatitis has led to a resurgence of research into the development of blood substitutes.
In this review we examine the options available for fluid resuscitation of hemorrhagic shock and comment on a number of blood alternatives under investigation. In addition, those circumstances under which consideration should be given to limiting or delaying fluid resuscitation are discussed. Although beyond the scope of this review, it must be recognized that to be successful, fluid resuscitation must be used in conjunction with efforts to prevent further blood loss and limit tissue injury. Also, any concurrent causes of shock (e.g., cardiogenic, neurogenic, septic, anaphylactic) must be managed appropriately.
Options for fluid resuscitation
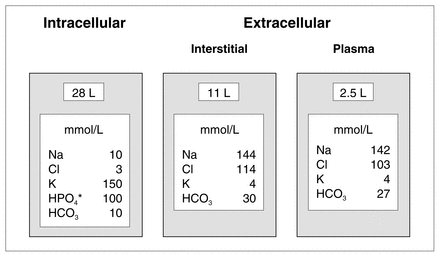
Ideally, the optimal fluid for resuscitation would combine the volume expansion and oxygen carrying capacity of blood, without the need for crossmatching or the risk of disease transmission. In addition, it would restore and maintain the normal composition and distribution of body fluid compartments (Fig. 1). Although the ideal fluid has yet to be found, a variety of options are currently available for fluid resuscitation (Tables 1 and 2).
Volume and composition of functional compartments of body fluids in a man weighing 70 kg. *Inorganic phosphorus exists as a buffered mixture, mostly as HPO4, and is the primary intracellular anion.
Options for Fluid Resuscitation
Effects of Various Resuscitation Fluids
Crystalloids
Normal saline or lactated Ringer’s solution
At present, the first-line fluid of choice for resuscitation remains isotonic crystalloids (0.9% sodium chloride or lactated Ringer’s solution) for a number of reasons, including proven effectiveness, high availability, low morbidity and low cost. For losses of up to 30% of the circulating blood volume, crystalloid alone will often suffice. The volume of fluid required for adequate resuscitation is approximately 3 times that of the estimated blood loss. However, with ongoing blood loss or more significant hemorrhage, prompt surgical intervention or blood therapy becomes necessary.
Hypertonic saline
Hypertonic saline (HS) combines several of the advantages of crystalloids with those of colloids. For instance, by drawing intracellular fluid into the vascular space, HS can restore circulating volume and normal hemodynamics with a smaller volume infused than isotonic crystalloids. This has been suggested as a mechanism to reduce cerebral edema in patients with a head injury and to reduce pulmonary edema in patients with lung contusion.1,2 There are a number of studies suggesting that HS may also improve outcomes by altering immune function.3–7 However, concern has been raised in animal studies that HS resuscitation in the presence of uncontrolled hemorrhage may adversely increase blood loss and mortality.8
Clinically, a role for HS in resuscitation has yet to be clearly defined. A randomized controlled trial comparing the effect of HS, hypertonic saline with dextran (HSD) or isotonic saline in patients with hypovolemia treated in the emergency department found no difference in outcomes (although less fluid was needed to restore normal blood pressure in the HS groups).9 Similarly, in a study examining 359 trauma patients with hypotension (defined as a systolic blood pressure less than 90 mm Hg) in which patients were given either 250 mL of HSD or isotonic saline as needed, no significant difference in the death rate was found.10 A meta-analysis11 and a cohort analysis12 of the individual patient data from prospective randomized controlled trials comparing HSD and HS alone, as compared to isotonic crystalloid solution, suggested some benefit of hypertonic solutions in certain trauma patients, specifically, those with penetrating injuries11 and those with combined shock and severe head injury.12 However, another meta-analysis using the pooled study data of 1200 patients in 14 trials (8 examining HSD, 6 HS) found no differences in survival with HS, HSD or isotonic crystalloid.1
It is notable that very few adverse effects of HS administration were observed in the various clinical trials.13 Rare episodes of hyperosmolarity were found, usually in association with ethanol intoxication, and although minor electrolyte imbalances occurred, no clinically significant sequelae such as central pontine myelinosis were reported.
In summary, a significant advantage of HS over standard crystalloid solutions remains to be proven. The decreased volume of hypertonic solution required to obtain the same degree of resuscitation may be of some benefit in prehospital environments, where storage or transport of fluids is a logistical concern. There is some data to suggest that for patients with shock and traumatic brain injury, HS may improve outcome.
Colloids
Colloid solutions have the advantage of providing more rapid restoration of circulating volume with a smaller infused volume than physiologic salt solutions. Another purported benefit is their ability to maintain or restore serum protein levels. However, recent meta-analyses of randomized, controlled clinical trials of fluid resuscitation with colloids versus crystalloids suggest that there is little, if any, role for colloids in the resuscitation of critically ill patients.14–16 In particular, an increased absolute risk of death of 4% was found with colloids by Schierhout and Roberts,15 and increased mortality in trauma patients resuscitated with colloids versus crystalloid was found by both Choi and associates14 and Velanovich.16 Considering that colloids are also more expensive than crystalloids, it would appear that their role in resuscitation should remain limited outside clinical trials. Further trials are indicated, however, as there were methodologic problems within many of the primary studies on which these meta-analyses were based (e.g., few were blinded).14 As well, the results of meta-analysis are not necessarily replicated by more rigorous, large, randomized controlled trials.17 For completeness we shall briefly review the colloid solutions currently available.
Albumin
The prototypic colloid is 5% or 25% albumin, purified from human blood. A purported advantage of albumin solutions over crystalloids is that the larger molecules increase the oncotic pressure within the intravascular space; this action alters Starling’s forces and increases intravascular volume as a result of water being drawn into the vessels. Unfortunately, in the presence of leaky capillary membranes, such as those found in sepsis, burns and severe hemorrhagic shock, this is not necessarily true. It has been suggested that under such circumstances, leakage of albumin across the membranes may exacerbate rather than reduce interstitial edema.18 Given the added disadvantages associated with infusing blood products (limited availability, disease transmission) and the increased cost, along with the failure of studies to demonstrate a clear advantage in terms of survival or lower morbidity, the use of albumin in resuscitation is currently not favoured.
Synthetic colloids
There are a number of commercially available synthetic colloids that have the advantages of colloid without the risks and limited availability of blood products. Hydroxyethyl starch (hetastarch) is a synthetic colloid formed from hydroxyethyl-substituted branched-chain amylopectin (average particle molecular weight 450 kD) first introduced 20 years ago. It is available in a 6% solution of normal saline, which contains 60 g/L of hetastarch and has a colloid osmotic pressure of about 300 mOsm/L. Although studies comparing hetastarch to crystalloids confirmed that the colloid was a better volume expander volume for volume, no significant reduction in organ dysfunction or mortality was observed.19,20 Reported adverse effects include prolongation of activated partial thromboplastin time and anaphylaxis.
Pentaspan (Dupont Pharma, Mississauga, Ont.), a mixture of 10% pentastarch in normal saline (average molecular weight 260 kD), similarly provides expansion of the plasma volume in excess of the volume infused and lasts for approximately 18 to 24 hours. As for hetastarch, there are few studies demonstrating an advantage over crystalloid, other than the smaller volume required to achieve similar hemodynamic parameters.21,22 There are a number of limitations to the use of pentastarch. First, since pentastarch is eliminated primarily by renal excretion, it is not recommended for use in patients with renal failure (except as a result of hypovolemia). Second, large volumes of pentastarch can dilute plasma proteins and hemoglobin, and may impair coagulation mechanisms. Third, when given in high doses Pentaspan has been shown to be embryocidal in New Zealand rabbits and Swiss mice, and therefore, should be used with caution in pregnancy. Finally, pentastarch is contraindicated in patients with hypersensitivity to hydroxyethyl starch (although relative to hetastarch, fewer anaphylactic episodes have been reported).
Blood
Although crystalloid alone may suffice for resuscitation of class 1 or 2 hemorrhagic shock (i.e., losses of less than 15% to 30% blood volume), more significant hemorrhage requires blood transfusion to maintain sufficient oxygen delivery to tissues. The reason for this is that oxygen delivery is a function of cardiac output × oxygen content, which in turn is a function of percent oxygen saturation × Hb × 1.34. In emergencies, if typed and cross-matched blood is not readily available, O-negative (or O-positive in men or women older than 50 years) may be used, followed by type-specific or fully cross-matched blood as it becomes available. In order to maximize oxygenation, factors that impair the ability of hemoglobin to give up oxygen (e.g., alkalosis, reduced diphosphoglyceric acid and hypothermia) should be avoided.
Although there is no doubt that blood transfusion can be life-saving, it is reserved for cases of significant or ongoing bleeding because of a number of drawbacks. First, the blood supply is quite limited both in the quantity of blood available, as well as the duration that blood can be preserved after collection. For instance, with increased length of storage time, potassium, lactate and ammonia levels rise, and 2,3-diphosphoglycerate levels (necessary for oxygen release) are adequate for only about 14 days before decline. Also, blood must be stored refrigerated and should be rewarmed to prevent hypothermia during infusion. Blood products also carry a risk of disease transmission, as well as a risk of transfusion reactions. Further, an immunosuppressive effect of allogeneic blood, particularly with respect to increased risk of postoperative infection and tumour recurrence has been suggested.23 There is also evidence to suggest that blood transfusion may be an independent risk factor for post-traumatic organ dysfunction. 24
Autologous blood salvage techniques
In an attempt to overcome the problems of an inadequate blood supply, time required for cross-matching, transfusion reactions and the transmission of viral diseases associated with allogeneic blood, several methods of salvaging autologous blood loss for reinfusion have been developed. The simplest method involves the collection of shed blood into a reservoir (with or without an anticoagulant) followed by immediate transfusion through a micropore filter to remove contaminants. This method can be used, for example, to reinfuse blood loss secondary to a massive hemothorax. A more complex and costly method used more often in elective surgery is the collection and washing of shed blood in saline, followed by concentration of the blood cells before reinfusion. This technique requires equipment set-up and trained personnel and has not been found to be of significant benefit unless more than 3 units of packed cells can be retrieved and transfused.25 Although efforts are made to avoid the use of contaminated blood, it has been demonstrated that even moderate contamination by enteric contents poses little risk to the patient provided antibiotics are given perioperatively.26,27 Other reported drawbacks of autotransfusion include microemboli of platelet plugs and fractured red blood cells, and the use of anticoagulants.
Blood substitutes
Recently, there has been renewed interest in the development of red cell substitutes for use in trauma or elective surgery, either alone or in combination with acute normovolemic hemodilution (allowing more aggressive hemodilution followed by gradual reinfusion of the blood as the synthetic blood substitute is eliminated). In traumatic hemorrhage, the ideal situation would involve resuscitation with blood substitutes until the bleeding has been controlled, followed by the transfusion of allogeneic blood. This approach would decrease the demand on blood supply services. The blood substitutes currently being investigated can be divided into 2 classes, modified hemoglobins and perfluorocarbons.
Modified hemoglobins
A number of modified hemoglobin products are currently in various stages of development. One approach being examined involves the creation of encapsulated hemoglobin molecules with use of various biodegradable polymers to create artificial red blood cells that do not express blood group antigens on their surfaces. A major limitation encountered with such cells, however, is their uptake and destruction by the reticuloendothelial system. In addition, because of the complexity involved in the structure and contents of the cells, most products have yet to reach the stage of clinical trials in humans. Preliminary animal experiments, however, appear promising.
Another approach being examined is the development of cross-linked or polymerized hemoglobin molecules. For example, Polyheme (Northfield Laboratories, Evanston, Ill.), is a pyridoxylated, polymerized hemoglobin product made from out-of-date human banked blood. Each unit of Polyheme contains 50 g of hemoglobin in 500 mL of solution and is equivalent in the amount of hemoglobin to a unit of packed red blood cells. In a randomized study of 44 trauma patients by Gould and associates,28 no significant adverse sequelae were noted after transfusion of up to 20 units of Polyheme, and the blood transfusion requirements of those receiving the blood substitute were reduced by approximately half.28,29 Phase III trials are currently underway. Hemolink (Hemosol, Etobicoke, Ont.) is another polymerized human hemoglobin product about to enter a phase III clinical trial in Canada, the United States and the United Kingdom. Non-human sources of hemoglobin substitutes are also being developed. Hemopure (Biopure/Pharmacia and Upjohn, Cambidge, Mass.) is a bovine hemoglobin-polyethylene glycol conjugate. An advantage of this product is that unlike human hemoglobin products, 2,3-diphosphoglyceric acid is not required for oxygen release, and an ample supply of donors is available. However, a disadvantage is the need for precautions to prevent the transmission of bovine pathogens. A phase III trial of Hemopure in humans is pending. Recombinant hemoglobin-based products are also being developed (e.g., Optro; Baxter Healthcare, formerly Somatogen, Boulder, Colo.) and offer the greatest promise for unlimited supply and safety, although cost may be a limiting factor.
Advantages of the hemoglobin-based blood substitutes include their ready availability without the need for cross-matching, their long shelf-life, their ability to be stored at room temperature and their reduced risk of disease transmission. Also, since they have a lower viscosity than blood they may enhance oxygen delivery to peripheral tissues, and they have not been found to be immunosuppressive. As mentioned, the use of blood substitutes to maintain oxygen carrying capacity during acute hemorrhage would reduce the strain on scarce blood bank supplies.
Unfortunately, the hemoglobin solutions currently available have a number of disadvantages, including their relatively short half-life (24 to 48 hours), their interference with certain biochemical laboratory tests30 and their vasoreactivity. The vasoreactivity is thought to be secondary to binding of nitric oxide, which can lead to the development of significant systemic as well as pulmonary vasoconstriction. This vasoreactivity, however, can be mitigated by measures to reduce extravasation of the hemoglobin solution from the intravascular space (such as encapsulation, polymerization or conjugation to a large molecule such as polyethylene glycol). There is also a concern that ultra-pure hemoglobin products may worsen reperfusion injury owing to their lack of protective red blood cell antioxidant enzymes such as superoxide dismutase. To address this concern, a new generation of hemoglobin blood substitutes are being developed that incorporate various antioxidant enzymes.
As with many of the alternatives to traditional resuscitation fluids, further studies are needed to determine what role these blood substitutes will play in resuscitation. For example, in contrast to the promising results of preclinical studies, a multicentre, randomized controlled trial assessing the effect of diaspirin cross-linked hemoglobin (DCLHb) on the death rate in humans with severe traumatic hemorrhagic shock was stopped at the interim analysis because of concerns related to pulmonary hypertension and a higher death rate in the group receiving the hemoglobin product compared with the saline controls was found (death rate at 48 hours 38% versus 15%, p = 0.01; at 28 days 46% versus 17%, p = 0.003).31 DCLHb differs from the other hemoglobin solutions previously mentioned in that it is not polymerized or conjugated to a large molecule and thus has increased vasoconstictive properties. Nevertheless, although the authors proposed several arguments related to study conduct and baseline differences between groups that may have accounted for their findings, this study led Baxter Healthcare (Boulder, Colo.) to abandon further development of its DCLHb product. Clinical trials with other hemoglobin solutions continue as does the development of new and improved hemoglobin products.
Perfluorocarbons
Oxygent (Alliance Pharmaceutical, San Diego, Calif.) is an example of a perfluorocarbon compound that is being investigated for several applications, including that of a blood substitute. Because of its relatively high solubility for oxygen and carbon dioxide, after intravenous infusion perflubron emulsion particles can transport oxygen from the lungs to the tissues where the oxygen is released by simple diffusion at approximately twice the rate of oxygen dissociation from hemoglobin. Similarly, carbon dioxide is transported from the tissues to the lungs. Purported advantages of perflubron emulsions include compatibility with all blood types, a shelf life of 2 years and no risk of transmission of blood-borne infections. As well, it would provide an acceptable alternative for those having religious objections to the use of blood products. To date, phase I and II studies in elective surgical patients have demonstrated no adverse effects on hemostasis, platelet function or coagulation parameters. The primary indications for this blood substitute that are currently being explored include its use as a diluent to reduce blood transfusion under elective circumstances and a possible bridge to transfusion when blood is not available. One particular disadvantage of perfluorocarbons, however, is the necessity for 100% inspiratory oxygen to provide effective oxygenation. Other disadvantages include interference with certain laboratory tests30 and uptake by the reticuloendothelial system, as well as the current limitation of 20% to 25% perfluorocarbon as the maximum stable concentration (which provides only 5mL oxygen/100 mL). Research into modifications of perfluorocarbons to obviate these limitations continues.
Fluid resuscitation: When and how much?
A final topic of discussion is the recently revived debate over whether or not fluid resuscitation in the rapidly hemorrhaging patient before operative control of the bleeding is beneficial. In fact, some authors propose that it may be detrimental and should be avoided.32,33 Traditionally it has been assumed that the timely restoration of perfusion and oxygen delivery was imperative to improving outcomes in trauma. This tenet was questioned, however, as early as 1918 by Cannon and associates,34 who suggested that normalizing blood pressure during active bleeding may not be optimal therapy as “if the pressure is raised before the surgeon is ready to check any bleeding that may take place, blood that is sorely needed may be lost.”34 Over the past decade, this issue has once again been brought to the forefront.
In 1994 Bickell and associates32 reported the results of a study examining the effect of immediate versus delayed fluid resuscitation in 598 adults having penetrating torso injuries and a prehospital systolic blood pressure less than 90 mm Hg. Increased survival (70% versus 62%), fewer complications (23% versus 30%) and shorter hospitalization were found in those patients in whom aggressive fluid resuscitation was delayed until the time of operative intervention.
Several issues, however, should be borne in mind when interpreting these results. First, this study has been criticized for a number of weaknesses and methodologic problems (e.g., large standard deviations in the amount of fluids the groups actually received, patient heterogeneity, analysis of results only by intent to treat, retrospective stratification of patients prior to statistical analysis). Second, this study took place in a large urban centre, with relatively short transport times from the scene to the operating room. A number of studies examining prehospital fluid resuscitation have suggested that patient outcomes are not improved if the transport time to definitive care is under 30 minutes35–37 (although, given the paucity of randomized controlled trials, no definitive conclusions can be drawn). Third, the study population had sustained penetrating injuries leading to uncontrolled hemorrhage. It is unclear whether victims of multisystem blunt trauma would respond similarly. Certainly in animal experiments, it has been demonstrated that a model in which the hemorrhage stops before resuscitation (as may occur with closed fractures) differs quite significantly from a model in which the bleeding continues. In the latter scenario, too aggressive resuscitation before surgical control of the bleeding may have a number of adverse effects including increased blood loss and mortality.38–41 The postulated mechanisms for this include increased hydrostatic pressure driving ongoing bleeding or dislodging a clot, as well as the decrease in blood viscosity and dilution of clotting factors.42 A caveat that also bears consideration when comparing such studies, is the anesthetic used, which can also significantly affect blood loss.43–45
Although hypotension may be tolerated by a variety of vital organs for periods up to a couple of hours, cardiac arrest is clearly not. Further, the effect of more prolonged hypotension in certain trauma patients, such as those with severe closed head injuries, is especially detrimental. Certainly, increased morbidity and mortality has been found in patients with a head injury and low cerebral perfusion pressures.46–48 It is also unclear how well patients with comorbid conditions or diminished physiologic reserve, such as the elderly, would tolerate delayed or limited resuscitation.
Thus, at present the evidence suggests that in remote or rural communities, in the elderly or in those patients with controlled hemorrhage, the timely restoration of perfusion and oxygen delivery should be the primary objective. In patients with uncontrolled hemorrhage from penetrating trauma in close proximity to a hospital facility capable of definitive care, less aggressive fluid resuscitation pending prompt surgical intervention may be used. The question of how to judge the adequate amount of fluid resuscitation in these patients remains to be answered.49
Future directions
An increasing number of questions with regard to fluid resuscitation remain unanswered. Hemorrhagic shock is a complex process that encompasses not only inadequate oxygen delivery to meet tissue demands but also the vast array of hemodynamic, metabolic, endocrine, hematologic, inflammatory and immune responses of the host to the inciting event as well as its sequelae (Fig. 2). To effectively assess the relative merits of different resuscitation protocols, including the use of novel synthetic blood substitutes or biologically active solutions capable of modifying the host stress response, further understanding of this complex response to injury and shock is required. In addition, the development of improved technologies and end points50 to assess the adequacy of fluid resuscitation in both hospital and prehospital settings is sorely needed, as is better identification of the various groups of trauma victims. Taken together, this will allow the design of resuscitation protocols that can be tailored to optimize the outcome for individual patients.
The process and outcome of hemorrhagic shock. GI = gastrointestinal, PMN = polymorphonuclear leukocytes.
- Accepted June 23, 2000.