Abstract
Objective: Selection of the optimal treatment strategy for patients with rectal cancer requires appropriate investigation, but published guidelines provide no clear consensus. We examined the current practice pattern for the investigation of primary rectal cancer by general surgeons in the province of Ontario, Canada.
Methods: A telephone interview was completed by 124 surgeons in Ontario who manage patients with rectal cancer, who indicated the investigations they routinely perform in assessment before treatment. An exploratory cluster analysis was used to identify surgeon-related variables that predicted the pattern of investigation; these were tested in univariate and multivariate analyses.
Results: Cluster analysis identified 3 distinct groups of surgeons based on patterns of test usage. Univariate analysis showed that the use of chest radiography, computed tomography of the abdomen and pelvis, and ultrasound of the abdomen varied significantly with the surgeon’s subspecialty training, practice location and years in practice. Regression analysis confirmed that each of these 3 variables independently predicted the pattern of preoperative investigation. There were no significant predictors of the use of colonoscopy, sigmoidoscopy or carcinoembryonic antigen level. Over half of surgeons reported that they would have ordered additional imaging tests but did not because of lack of availability. The perceived desirability of additional tests, endorectal ultrasound in particular, varied with training.
Conclusions: The current practice pattern for the preoperative investigation of primary rectal cancer by general surgeons varies significantly with specific surgeon-related variables, with potential impact on the management and outcome of patients with rectal cancer.
Colorectal cancer is the third most common cancer in North Americans. In one-third of patients with colorectal cancer, the rectum is the location of the primary tumour, accounting annually for over 46 000 new cases.1,2 Advances in the investigation and management of rectal cancer include improved imaging techniques such as endorectal ultrasound (ERUS) and MRI, the use of neoadjuvant chemoradiation and the advent of total mesorectal excision.3–5 However, despite these and other improvements, up to 50% of patients with primary rectal cancer still die of recurrent disease.3
Appropriate management of rectal cancer depends on the stage of the tumour. Knowledge of the location and extent of locoregional disease, and whether distant metastases are present, is important in pretreatment planning of the sequence of therapy and choice of operative procedure.6–13 Locoregional staging of rectal cancer may involve digital rectal examination, sigmoidoscopy, ERUS, CT, MRI and/or positron-emission tomography (PET) scans. Distant disease may be assessed with ultrasonography (US), CT, MRI and/or PET scans as well as chest radiography (CXR) and serum carcinoembryonic antigen (CEA) level. However, the reported accuracies of different techniques for T-, N- and M-staging of rectal cancer varies markedly.7,8 For instance, in several recent reports7,8,12 the accuracy of determining wall penetration of primary rectal cancer (T-stage) ranged from 57% to 100% for pelvic CT, 43%–97% for ERUS, and 73%–95% for MRI. Reported accuracies of different techniques in assessing local nodal involvement and distant spread also vary considerably. In practice, choice of a staging technique is based as much on availability and perceived cost-effectiveness as on its sensitivity and accuracy.7
Guidelines for the preoperative investigation of rectal cancer published by professional bodies such as the Society of Surgical Oncology (SSO) and the recommendations of individual experts (Table 1) are not only often inconsistent but sometimes conflicting. 6,11,14–21 This variability, coupled with the selective application assigned to particular tests (Table 1), does not permit easy comparisons or consensus development. Indeed, a recent review20 of 41 studies evaluating the role of local excision revealed salient differences in the use of imaging techniques for staging and selecting patients, implying a lack of consensus among clinicians.
Guidelines for the preoperative investigation of primary rectal cancer*
Given the primacy of surgical resection in the management of primary rectal cancer, the individual surgeon is a critical determinant of pretreatment investigation. Because few studies have attempted to determine current practice patterns for the preoperative investigation of rectal cancer among surgeons,20,22 we surveyed surgeons in a defined geographic area under uniform governance, the province of Ontario.
Methods
Inclusion criteria
We sought to obtain population-based data on the investigation of rectal cancer by surgeons across the province. Registries of general surgeons practising in Ontario were obtained from the College of Physicians and Surgeons of Ontario (CPSO) and the Ontario Association of General Surgeons (OAGS).
Membership in CPSO is compulsory for all physicians in clinical practice in Ontario, whereas membership in OAGS is voluntary. Both registries include surgeon name, practice location and office telephone numbers. In 1999, 859 general surgeons were registered as active with the CPSO, including all of the 431 listed with OAGS. From an internal OAGS survey, 134 of these general surgeons were known to manage patients with rectal cancer.
These 134 surgeons, plus the first 377 general surgeons listed in alphabetical order in the CPSO registry (for a total of 511), were selected for initial office-telephone contact. Of these, 150 reported that they did not manage rectal cancer; another 220 confirmed that they did. (The offices of the remaining 141 surgeons could not be contacted in 4 telephone attempts.) An attempt was then made to call the 220 surgeons who do manage cases of rectal cancer, to ask each directly to participate in a telephone survey. Of the 220, 96 could not be contacted in person after 3 attempts; 4 declined to be surveyed; and 124 completed the survey.
Demographic data on all surgeons practising in Ontario in 2000 were obtained from the Ontario Physician Human Resources Data Centre.
Survey content and format
The telephone survey had an open-ended, scripted format developed after field-testing for reproducibility on a small sample of general surgeons (n = 15) practising in Toronto, Ontario. During the survey, no prompting or choice of responses was provided, except for categories for number of operations per year (see the second question, below). The survey consisted of 7 questions, in this order:
Anatomically, how do you define the rectum?
How many operations for primary rectal cancer do you perform in a year? There are four choices: < 5, 5–15, 16–40, > 40.
What examinations or investigations do you routinely perform in evaluating a new patient with primary rectal cancer?
Are there investigations that you would like to order routinely but do not due to lack of availability or time constraints?
From which general surgery program did you graduate, and in what year?
If you completed further specialty training after your general surgery residency, what was the program? And for how many years?
How many years have you been in practice?
This method of data collection was chosen to obtain responses that best described the surgeon’s routine preoperative work-up without providing options to bias their answers.
Statistical analysis
Physicians were assigned numbers for anonymity, and their data entered into an Excel spreadsheet. Descriptive data were summarized via counts and percentages. All analyses were performed by the University of Toronto Statistical Consulting Group using SAS software, with α = 0.05.
To determine if patterns of investigation were associated with particular surgeon-related characteristics, a cluster analysis was performed on the reported use of 7 tests: CEA, CXR, abdominal and pelvic (A+P) CT, abdominal US, ERUS, colonoscopy and sigmoidoscopy. Surgeons were divided into 10 clusters based on the tests that they reported using routinely. Of these 10 clusters, only 2 were large (41 and 37 surgeons, respectively); 3 clusters were therefore used in the analysis, with cluster 3 made up of all other survey respondents (n = 46). Frequency tables of test performance by cluster were created, and χ2 tests performed to assess differences between clusters.
In an effort to avoid investigator bias in developing a list of candidate surgeon-related variables, we used this initial exploratory cluster analysis to identify demographic variables that varied significantly between the clusters that had been created on the basis of test usage. Additional frequency tables were created and χ2 tests performed to examine demographic differences between clusters. To then test directly for correlations between the demographic variables identified as significant by cluster analysis and test usage, univariate analysis was performed on the original raw data (χ2), and logistic regression analyses for each of 7 individual types of investigation.
Results
Demographics
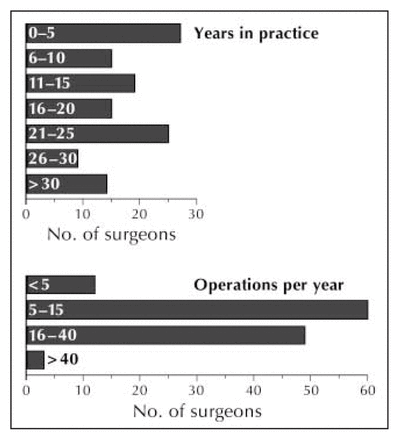
Of 361 potential respondents, 124 (34%) completed the telephone survey. For the demographic characteristics we documented (Table 2, Fig. 1), this group was representative of general surgeons across Ontario: 75% had been in practice for at least 5 years, and 66% for more than 10 years; 41% practised in rural settings (towns of ≤ 100 000 inhabitants). This distribution reflects the overall population of general surgeons practising in Ontario in the year 2000, when 79% had been in practice at least 5 years, and 36% practised in a rural setting as just defined.23 In addition, around 60% of the surgeons surveyed had, after completion of their general surgery residency, undertaken at least 1 year of further clinical training of some type. Ninety percent of respondents said they performed at least 5 operations for rectal cancer per year (Table 2, Fig. 1).
Distribution of the 124 surgeons surveyed, according to their years in practice and number of operations for rectal cancer performed per year.
Demographic characteristics of all survey respondents (n = 124)
Definition of the rectum
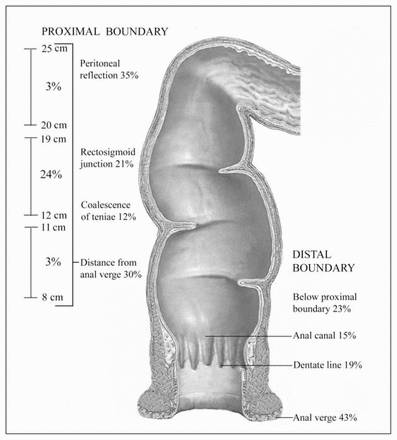
In the first part of the survey, we asked surgeons to define the proximal and distal boundaries of the rectum. There was considerable variation in the definitions of both (Fig. 2): 66% of respondents defined the upper boundary of the rectum as an anatomic landmark (e.g., the peritoneal reflection); 30% defined it as a given distance (in cm) from the anal verge. Seventy-six percent of respondents provided an anatomic landmark for the distal boundary of the rectum, whereas the remaining 23% did not, describing the rectum as anything below their definition of the upper boundary. The single most common definition of the distal boundary of the rectum was the anal verge (43%).
Proximal and distal boundaries of the rectum as defined by all respondents who completed the survey (n = 124). The percentage of surgeons naming a particular proximal or distal boundary is indicated. (For the distal boundary, 1 surgeon did not respond.)
Summary of preoperative tests ordered
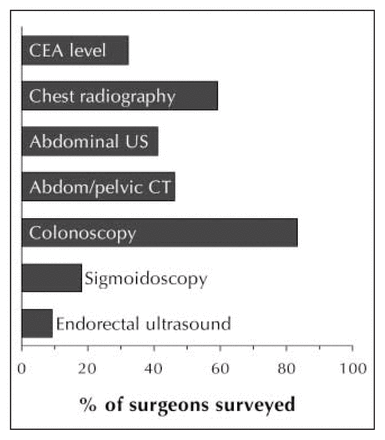
Percentages of respondents who order specific tests as part of their routine preoperative work-up are shown in Fig. 3. The most common test, colonoscopy, was routinely performed by 83% of the physicians surveyed. The percentages ordering additional investigations for locoregional or distant disease staging varied from 9% for ERUS to 59% for CXR. However, 56% of those surveyed said they would have ordered 1 or more additional tests on a routine basis, but did not do so because of lack of availability. Of this group, 52% said they would obtain ERUS and 42% a CT scan. A smaller fraction of physicians said they would order either MRI (5%) or measurement of a CEA level (6%) if these tests were more readily available.
Proportion of the 124 respondents who reported routinely ordering the indicated test in the preoperative investigation of rectal cancer. Abdom = abdominal; CEA = carcinoembryonic antigen.
Cluster analysis
As described in the Methods, exploratory cluster analysis was used to identify patterns of routine preoperative testing that might distinguish groups of surgeons within the sample; 3 clusters of surgeons were defined by the frequency with which 7 individual investigations were routinely employed in their preoperative work-up (Table 3). Use of A+P CT, abdominal US and CEA level were factors that distinguished between the 3 clusters with a high level of significance (p < 0.0001). Smaller but still significant differences were seen for sigmoidoscopy (p < 0.0002) and colonoscopy (p = 0.0009). Although use of ERUS also differed between clusters, only a small number of surgeons overall order the test on a routine basis (n = 11). Frequency of CXR use was similar in the clusters, and the use of other investigations (e.g., liver function tests, MRI) did not meaningfully contribute to the assignment of clusters.
Summary of cluster analyses on pattern of routine preoperative investigation for primary rectal cancer
These 3 clusters were examined for frequency of surgeon-related variables. Differences between clusters in number of operations performed for rectal cancer per year (p = 0.82) in and surgeons’ anatomic definition of the rectum (p = 0.13) were not significant. Cluster 3 included more surgeons with colorectal or surgical oncology subspecialty training (p = 0.033) and more who practised in an urban teaching centre (p = 0.009) than did clusters 1 and 2. Surgeons in clusters 1 and 2 were more likely to have been in practice for at least 5 years than those in cluster 3 (p = 0.008).
Univariate analysis
Years in practice
The surgeons surveyed were in practice for a median of 15 years (range 3 mo to 44 yr). Number of years in practice (subdivided as ≤ 5, 6–16 or > 16) was significantly associated with use of A+P CT and abdominal US (Table 4). Recent graduates were more likely to routinely order CT scans (p = 0.0003) and less likely to order abdominal US (p = 0.045) compared with surgeons with more than 5 years of experience. Differences in the use of other tests as a function of years in practice or between surgeons in practice 6–16 versus > 16 years were non-significant.
Routine preoperative investigations according to subspecialty training, years of experience and type of practice location
Subspecialty training
Of the 124 surgeons surveyed, 27 (22%) had surgical oncology or colorectal (SO/CR) subspecialty training. The preoperative investigations routinely ordered by this group differed significantly from those by surgeons with other (n = 45) or no (n = 52) subspecialty training (Table 4). Overall, surgeons with SO/CR training ordered 42% more tests than other surgeons, and were more likely to order preoperative CT scans (p = 0.0002) and CXR (p = 0.044) and less likely to order abdominal US (p = 0.030) than surgeons with other or no fellowship training. Subspecialty training did not predict the use of any other tests, but 67% of surgeons with SO/CR training said that they would order ERUS routinely if it were available, compared with 30% of all other surgeons who would do so (p = 0.004).
Practice location
The distribution of surgeons surveyed who practice in urban teaching, urban non-teaching or rural hospital centres was well balanced (Table 4). Surgeons in urban teaching centres were significantly more likely to order preoperative CT scans and less likely to order abdominal US than other surgeons. Practice setting did not predict the use of other tests. Surgeons practising in rural or urban non-teaching centres were no more likely to indicate constraint by limited test availability than surgeons practising in urban teaching hospitals.
Self-reported operative volume and anatomic definition of the rectum did not predict the pattern of preoperative investigation in univariate analysis.
Multivariate analysis
When the 7 tests originally included in the cluster analysis underwent logistic regression analysis to determine whether specific surgeon-related variables remained predictors of test usage when multiple variables were considered, no individual demographic variable significantly predicted use of CEA, colonoscopy, sigmoidoscopy or ERUS. There was, however, moderate evidence that ordering a CEA was less frequent among surgeons who operate on 5–15 cases of rectal cancer per year than among others (p = 0.0536).
Surgeons with SO/CR training ordered CXR more often than those with other (p = 0.0112) or no subspecialty training (p = 0.0256). Surgeons in urban teaching hospitals were more likely to perform A+P CT than those in urban non-teaching (p = 0.0117) or rural hospitals (p = 0.0003). There was also moderate evidence that surgeons in practice for 5 or fewer years ordered A+P CT more than those who had been in practice longer (p = 0.06). Abdominal US was requested less frequently by surgeons with 0–5 years in practice than by others (p ≤ 0.03). Surgeons in urban non-teaching hospitals used abdominal US more than those in urban teaching hospitals (p = 0.0222).
In summary, then, surgeons with SO/CR training ordered more cross-sectional imaging for staging of locoregional disease, and more CXRs to stage distant disease. To stage the liver, A+P CT (rather than abdominal US) was used more by surgeons working in urban teaching centres and by recent graduates.
Discussion
This study reveals significant variation in the techniques currently used by surgeons in the province of Ontario to investigate primary rectal cancer. Colonoscopy was the only investigation that more than 80% of surgeons said they performed routinely. Although over 71% ordered cross-sectional imaging (CT or US) of the liver, only 59% said they obtained a CXR. About 50% of surgeons routinely obtained cross-sectional imaging of the primary tumour and regional nodes preoperatively. Such imaging is important to identify locally advanced disease that should be managed with neoadjuvant chemoradiation, 6,8,24 and can preclude an unnecessary laparotomy and futile dissection that will render subsequent resectional surgery more difficult. Also, it can prevent an inappropriate attempt at local excision for T3, T4 or node-positive tumours.
A cluster analysis identified 3 common patterns of preoperative testing, most strongly distinguished by differences in the use of A+P CT, abdominal US, CEA and ERUS. Surgeons’ post-residency subspecialty training, years in practice and practice setting varied significantly between clusters and were significant predictors of the pattern of preoperative investigation. In our study, surgeons with SO/CR subspecialty training were more likely than others to use cross-sectional imaging to assess the extent of locoregional disease. In addition, more of the SO/CR-trained surgeons (67%) said they would routinely request ERUS if it were readily available. These results echo those of Hool and associates,21 who found that the majority of colorectal surgeons in teaching centres in the United States routinely ordered ERUS preoperatively. However, ERUS appears to be employed much less consistently in other settings. Only 9 of 41 trials evaluating local excision utilized ERUS as part of the selection protocol. 20 In the National Surgical Adjuvant Breast and Bowel Project Protocol (NSABP) R03 trial,25 only 14% of patients underwent ERUS as part of their pretreatment work-up. Other studies and authors have used or advocated an inconsistent combination of CT, MRI and ERUS to stage locoregional disease,26,27 and some28,29 do not specify how staging was performed. The lack of emphasis in such trials on consistent techniques for locoregional staging is unsurprising, given that, except for digital rectal examination and colonoscopy, no other single technique is recommended by a majority of contemporary guidelines (Table 1).
In designing this study, we considered the possibility that some part of the expected variation in preoperative investigation would be attributable to differences in how individual surgeons define the rectum. The definition of the rectum did indeed vary considerably, with surgeons using a variety of endoscopic and intraoperative criteria to identify the proximal and distal boundaries of the rectum (Fig. 2). About a quarter of the responses were consistent with the recent National Cancer Institute endoscopic definition of the rectum–colon junction as being 12 cm from the anal verge.26 However, we found no significant correlation between the individual surgeon’s definition of the upper and lower boundaries of the rectum and the investigations routinely used to evaluate a patient with rectal cancer.
Up to 25% of all patients with rectal cancer present with distant metastases, the most common sites being the liver and lungs.22,30 An appropriate treatment strategy depends on the resectability of distant disease, and may include radiotherapy and/or chemotherapy to achieve control of local disease.30–32 Published guidelines present no consensus regarding the appropriate technique(s) for the routine assessment for distant spread of rectal cancer (Table 1). Moreover, in trials evaluating adjuvant therapy or different operative approaches, the preoperative work-up for distant disease may be CXR and abdominal US, CXR and abdominal CT, or CT of the chest and abdomen.25,27,28,33 In our study the results were varied, but SO/CR-trained surgeons were more likely to order A+P CT (78%) and CXR (82%), and less likely to order abdominal US (22%) than surgeons with other or no specialty training (Table 4). All surgeons practising at urban teaching centres obtained pretreatment cross-sectional imaging of the liver, in contrast to results from Hool’s group,21 who found that only 43% of attending surgeons in American colorectal training programs routinely ordered preoperative imaging of the liver. The choice of CT over US among SO/CR surgeons in our study may reflect an appreciation of synchronous imaging of the primary tumour, regional nodes and periaortic nodes, and also the surgeon’s desire to personally review imaging studies before operating.11,30
In summary, this study demonstrates a lack of consensus on an appropriate preoperative work-up for patients with primary rectal cancer among surgeons in Ontario. Subspecialty training, practice location and years in practice were factors that predicted the choices made. Limited test availability, particularly of ERUS, was also cited as an important determinant. The lack of consistent guidelines from professional societies or experts, together with a continuing trend toward increased subspecialization after general surgical training, may drive further surgeon-dependent differences in preoperative planning. Surgeon-specific variables have also been correlated with a variety of outcome measures in patients undergoing resection of primary rectal cancer. 3–5,31,34–43
The differences we have identified in preoperative staging could account for some part of the observed differences in choice of operative procedure, local recurrence and survival rates observed between individual surgeons.31,34–44 Indeed, recent prospective studies have shown that pretreatment ERUS increased the use of neoadjuvant therapy and the rate of curative resection in both single and multi-institutional trials.45,46 The limited availability of ERUS evaluation found in our study is thus of particular concern. At a minimum, some form of cross-sectional imaging of the primary tumour should be performed before making a treatment decision,47 and educational initiatives should stress not only proper resection technique but also appropriate preoperative investigation.
Acknowledgements
The authors thank Tamara Arenovich and the Statistical Consulting Service at the University of Toronto for assistance with the analyses. Dr. McMullen was supported by funding from the Ontario Medical Association and the University of Toronto Medical Alumni Association.
Footnotes
Competing interests: None declared.
- Accepted April 7, 2004.