Abstract
Background: Osteonecrosis of the femoral head commonly occurs in patients aged under 50 years. Because of a high rate of complications associated with joint replacement surgery in this population, surgical techniques, such as vascularized fibular grafting, have been devised in an attempt to salvage the femoral head. The purpose of this study was to investigate the use of bone scintigraphy to predict graft retention after vascularized fibular grafting for osteonecrosis of the hip.
Methods: We evaluated single photon emission computed tomography images from 104 subjects whose hips were treated with vascularized fibular grafts between 1994 and 2000. We compared the signal intensity of the graft with the intensity of the ipsilateral proximal femoral diaphysis and assigned a score of 1 if less than diaphysis, 2 if equal to diaphysis and 3 if greater than diaphysis. We defined graft failure as conversion to or on the waiting list for total hip arthroplasty.
Results: Thirty percent of hips failed treatment (n = 31, mean graft survival 34.9 mo), while 70% of grafts were retained (n = 73, mean follow-up 56.6 mo). Bone scan scores were significantly lower in the failed group (mean 7.1, range 6–12), compared with the retained group (mean 8.5, range 6–18; p = 0.03). Logistic regression demonstrated that a bone scan score > 6 was associated with graft retention (p = 0.028), with an odds ratio of 3.08 (range 1.13–8.40).
Conclusion: These results suggest that having a well-perfused graft in the early postoperative period improves the chances of graft retention in the future.
Osteonecrosis of the femoral head is a debilitating disease that commonly occurs in patients aged under 50 years. The natural history of the disease is one of progressive necrosis, subchondral fracture of the femoral head leading to collapse and resultant arthrosis of the hip.1,2 Total hip arthroplasty has been demonstrated to have higher complication rates and a poorer prognosis in younger patients (aged < 50 yr)3,4; therefore, joint preserving procedures have been proposed to arrest disease progression. Several treatment methods have been reported, including core decompression,5 transtrochanteric rotational osteotomy6 and nonvascularized structural grafting7; however, the success rates of these procedures have been inconsistent.
The use of vascularized fibular grafts was initiated in an effort to not only provide structural support but also to enhance revascularization of the femoral head. Several studies have demonstrated improved function and pain relief after vascularized fibular grafting.8–10 Although many authors have suggested that it is preferable to preserve the femoral head in the precollapse stage, a recent study has demonstrated good overall graft survival and improved Harris Hip Scores after vascularized fibular grafting in patients with postcollapse, predegenerative osteonecrosis. 11 This suggests that patients with early and late stage osteonecrosis can benefit from vascularized fibular grafting.
It is believed that a viable and well-perfused vascularized bone graft is a prerequisite for graft survival.8,10 However, there is little evidence that graft viability is related to a successful clinical outcome. Several techniques have been proposed to assess graft viability, such as clinical observation of a buoy flap,12 Doppler ultrasound and flowmetry,13,14 angiography,8 bone scintigraphy15 and dynamic enhanced MRI.16 The lack of conformity in the use of these different techniques indicates the variability of their usefulness. Postoperative bone scintigraphy has been shown to be predictive of radiographic outcome after rotational osteotomy of the femoral head for osteonecrosis17; however, little information exists regarding scintigraphic assessment of the femoral head after vascularized fibular grafting. Malizos and colleagues15 evaluated bone scintigraphy and digital subtraction angiography after vascularized fibular grafting for osteonecrosis. They had a small cohort of subjects (n = 40) and relatively short follow-up (mean 22 mo), making it difficult to draw meaningful conclusions. At present, no immediate postoperative technique has been established to accurately predict graft retention. The purpose of this study was to investigate the use of bone scintigraphy to predict graft retention after vascularized fibular grafting for hip osteonecrosis.
Methods
We retrospectively reviewed prospective data, and the study was approved by the Research Ethics Board of St. Michael’s Hospital. From 1994–2000, 177 vascularized fibular grafts were performed in 152 consecutive subjects. Fifty subjects were ineligible for the study because they did not have a bone scan performed (n = 41) or the bone scan was technically inadequate (n = 9). Subjects without bone scans had their surgery when bone scans were not routinely performed (1994–1996). One subject died, 1 refused follow-up, 3 were excluded due to ipsilateral subcapital hip fracture and 18 were lost to follow-up. Thus, single photon emission computed tomography (SPECT) images of 104 hips from 93 subjects treated with vascularized fibular grafting were retrospectively reviewed by a radiologist not involved in the care of the patients and blinded to the outcome. Sixty-nine subjects were men and 35 were women. The mean age was 36.1 years (range 18–55 yr). The 73 subjects excluded were not statistically different from the analyzed group with respect to demographic parameters such as age, sex, involved side or etiology (p > 0.05).
Due to standard purging by the medical imaging department, it was not possible to review preoperative radiographs for 62 subjects. Further, inconsistencies in these subjects’ radiological reports prevented us from reliably classifying them solely from their office charts. As a result, severity of disease was radiographically classified in only 42 subjects according to Steinberg’s classification. There was 1 stage I, 19 stage II, 2 stage III and 20 stage IV. The associated etiological factors included trauma in 4 (4%), ethanol use in 8 (8%), steroid use in 70 (67%) and a combination of ethanol and prednisone use in 2 (2%). The remaining 20 (19%) were classified as idiopathic, because no etiological factor was identified. The operative procedure was a variation of the method of Urbaniak and colleagues.8 We used postoperative medical and rehabilitation protocols, as described by Louie and others.9 Graft failure was defined as conversion to or on the waiting list for total hip arthroplasty (THA); otherwise, subjects were classified as having retained their graft.
SPECT images of the hips were acquired 2 to 3 hours after intravenous injection of 1000 MBq Technetium 99m methylene diphosphonate. Axial oblique and coronal oblique delayed SPECT images were obtained of the hip in question at a mean of 2.7 days (range 1–6 d) from the time of surgery. A scoring system was developed for evaluating the grafts. The graft was divided into 3 regions for the scoring system: greater trochanter, femoral neck and femoral head. The intensity of activity in the vascularized fibular graft in each region was visually compared with the intensity of activity in the cortex of the ipsilateral proximal femoral diaphysis in both the axial and coronal planes. The signal intensity in the ipsilateral proximal femoral diaphysis served as an internal control. The comparison was scored on an ordinal scale of 1 through 3. Graft activity less than that of the proximal femoral diaphyseal cortex was given a score of 1, activity that equalled that of the proximal femoral diaphyseal cortex was given a score of 2 and activity greater than that of the proximal femoral diaphyseal cortex was given a score of 3 (see Fig. 1, Fig. 2, Fig. 3). Consequently, the final value for each hip was termed “bone scan score” and could range from 6 to 18 (i.e., scores 1 to 3 for each of the 3 regions on axial and coronal images). Repeated blinded scoring of a random subgroup of 20 subjects was performed, and intraobserver variability was calculated to be 0.93. Only 1 experienced nuclear medicine radiologist participated in the study, and as a result, interobserver analysis was not performed.
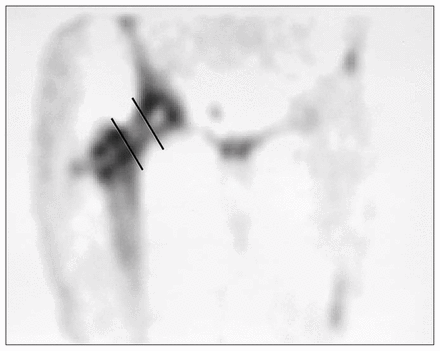
Subject 1. Coronal single photon emission computed tomography image demonstrating poor signal intensity along the graft. An example of the division of the graft into thirds is represented by the inserted bold lines. This represented a bone scan score of 3 and combined with an axial score of 3 for a total score of 6.
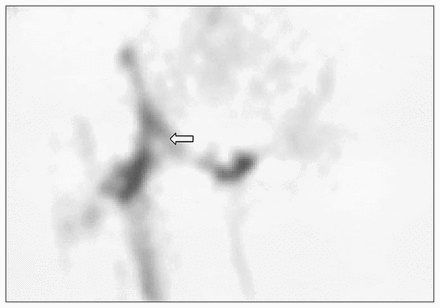
Subject 2. Coronal single photon emission computed tomography image demonstrating improved signal intensity along the graft. Poor signal in the femoral head region (see open arrow) limits the coronal score to 7; this combined with an axial score of 6 for a total score of 13.
Subject 3. Coronal single photon emission computed tomography image demonstrating excellent signal intensity along the graft. This represented a bone scan score of 9 and combined with an axial score of 9 for a total score of 18.
We performed statistical analysis with the SAS system software package (SAS Institute, Cary, NC) and used the Mann–Whitney U test to determine differences of bone scan scores between graft failure and retained groups. We used chi-squared analysis to determine proportional differences between graft failure and retained groups, and we used logistic regression analysis to determine the predictive value of the bone scan score on graft retention. A p value < 0.05 was considered significant.
Results
Thirty percent of subjects were converted to or on the waiting list for THA (n = 31 hips), at a mean graft survival of 34.9 months (range 5–98 mo). Graft survival time for subjects on the waiting list was determined from the time of surgery to the time of being placed on the waiting list (n = 5). Seventy percent of grafts were retained (n = 73) at a mean follow-up of 56.6 months (range 22–100 mo). Of the subjects who had preoperative radiographic staging (n = 42), there was no difference in the proportion of those who were Steinberg stage III and IV between graft retained and graft failure groups (56.3% v. 41.6%, respectively; χ2 = 0.288, p = 0.59).
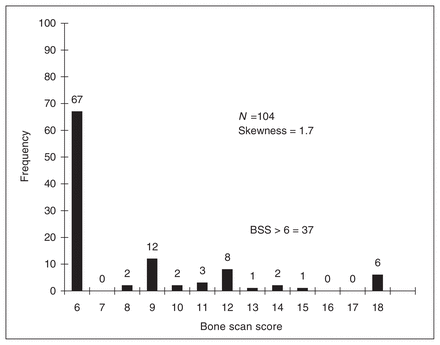
Bone scan scores were significantly lower in the graft failure group (mean 7.1 [standard deviation {SD} 2.7], range 6–12) compared with the graft retained group (mean 8.5 [SD 3.6], range 6–18; p = 0.03). Bone scan scores were positively skewed (1.7) in their distribution (see Fig. 4); 64% of subjects scored 6 while 36% scored above 6 (range 8–18). Owing to the skewness of the data, we divided it into 2 dichotomous groups, those with a score of 6 and those with a score above 6. Logistic regression revealed that a score greater than 6 was associated with graft retention (p = 0.028), with an odds ratio of 3.08 (95% confidence interval 1.13–8.4). Prediction of graft retention with a bone scan score greater than 6 was accurate in 84% of cases (31 of 37) in this study.
Distribution of bone scan scores (BSS) demonstrating positive skewness.
Discussion
Radionuclide bone scintigraphy has been shown to be an effective method of evaluating vascular patency and the viability of the bone. Controversy exists regarding which phase of the bone scan is more appropriate for assessing graft viability. Phase I of a 3-phase bone scan has been suggested to be the more sensitive phase for monitoring the graft viability15; however, this phase is simply a radionuclide angiogram and thus does not directly and fully evaluate graft bone function. Phase III of the scan represents both the metabolic activity of the bone and the patency of the vasculature supplying the bone. The uptake of radioactive ligand requires both an adequate delivery system and metabolically active osteocytes. Berggren and colleagues18 demonstrated that a positive phase III scan a week or longer after surgery does not reflect patency of the vessels or bone viability, because new bone is generally formed at the surface of the graft due to “creeping substitution.” Thus, it has been recommended that bone scans be performed within a week of surgery to avoid false positive uptake. This study used phase III of the bone scan, all phases being performed within 6 days postsurgery. We believe this is the most accurate phase to analyze for assessing graft perfusion and viability.
Scintigraphic assessment of free vascularized fibular grafting after mandibular or extremity reconstruction (or both)19,20 has suggested that bone scintigraphy correlates with graft survival. However, these studies had small sample sizes, they reported high false positive rates and they had difficulty interpreting the bone scans that were not clearly positive or negative and hence labelled indeterminant.
A recent study investigated the predictive value of dynamic enhanced MRI and SPECT after iliac crest vascularized grafts for osteonecrosis of the hip.16 Again, a small series was used (n = 40) and SPECT was performed within 1 month postoperative from the time of surgery and not within 1 week, as advocated by Berggren and others.18 Nevertheless, dynamic enhanced MRI was reported to be more sensitive and specific for predicting surgical outcome. Owing to the obvious methodological differences, it is difficult to relate the above-mentioned results to the present study.
To our knowledge, there has been no previous study examining the predictive value of SPECT after vascularized fibular grafting for osteonecrosis of the hip. Significant differences were found in bone scan scores, because subjects with graft failure had lower scores than did subjects with graft retention; this suggests improved graft perfusion post-operatively in the subjects who would ultimately retain their grafts. Further, subjects with a bone scan score above 6 were 3 times more likely to retain their grafts than were subjects with a score of 6. The over-all graft survival rate was 70% (73/104) for the hips observed for a minimum of 2 years. This value is consistent with those reported in the literature.8,9,11,21
It is important to recognize that a bone scan score of 6 is not equivalent to failure of the anastomosis with no flow to the graft. It simply means that there is less flow than that to the surrounding femoral cortex. There are several potential causes for this phenomenon, including vessel spasm, partial occlusion and outflow obstruction. Potential causes of graft failure include vessel thrombosis, anastomosis failure or complete occlusion from vessel kinking.
These results suggest that having a well-perfused graft, as predicted by bone scan score in the early postoperative period, improves the chances of retaining the graft in the future. Although this information has limited use with regard to acute intervention postoperatively, it would be useful in educating the patient regarding their prognosis and, in turn, may ultimately influence later orthopedic management. It might also be used to investigate the effects of variations in surgical technique, postoperative care or any other factors that could influence graft perfusion.
Acknowledgement
Thanks to Dr. Dawn Pearce for radiographic assistance.
Footnotes
Presented at the Canadian Orthopaedic Association Annual Meeting, Calgary, Alta., 2004.
Competing interests: None declared.
- Accepted February 10, 2006.