Gastrointestinal stromal tumours (GISTs) are the commonest mesenchymal tumours of the digestive tract, with an incidence estimated between 10 and 20/106 people annually.1 GISTs are characterized by overexpression in the tyrosine kinase receptor through mutations of c-kit or PDGFRA genes.1 The stomach is the most frequent site of GIST occurrence.1,2
Because of their high vascularity, GISTs are frequently associated with gastrointestinal bleeding1 and have been associated with severe gastrointestinal hemorrhage requiring either embolization or emergency surgery.3,4 Lifethreatening hemoperitoneum due to GIST rupture has not been described except after percutaneous diagnostic biopsy and in patients receiving imatinib mesylate treatment.1
We describe a case in which life-threatening GIST rupture was associated with nontraumatic hemoperitoneum.
Case report
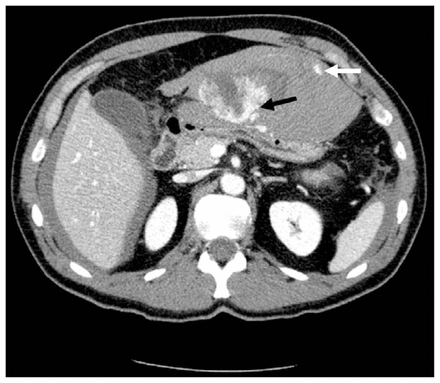
A previously healthy 49-year-old man presented with acute abdominal pain and severe hypotension. His medical history revealed recurrent epigastralgia over the last 6 months, which was treated by over-the-counter medications. He had no history of abdominal trauma. After resuscitation, diffuse peritoneal irritation and abdominal swelling were noted. Abdominal ultrasonography revealed the presence of free peritoneal effusion. Abdominal CT showed a large heterogeneous antegastric mass with active arterial bleeding into the peritoneal cavity (Fig. 1). Because of his hemodynamic instability an emergency laparotomy was performed. Blood (2.5 L) was evacuated from the peritoneum. An anterior gastric mass suggesting a GIST was found. It was associated with active arterial bleeding into the peritoneum. Complete resection and bleeding control were achieved by distal gastrectomy with Roux-en-Y reconstruction. Free tumour fragments due to tumour rupture were found within the peritoneum, which was irrigated with distilled water, for its cytolytic effect, and saline. The patient recovered well without postoperative complication.
CT scan (venous phase) shows a hypervascular gastric tumour (black arrow) associated with accumulation of intravenous contrast within the peritoneal cavity (white arrow).
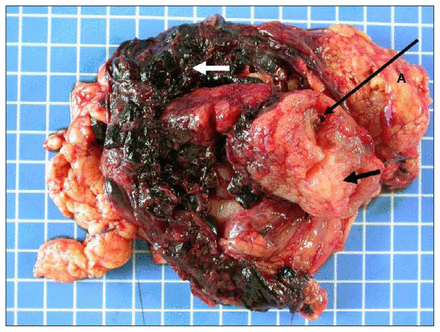
Pathological examination revealed a malignant gastric GIST (Fig. 2). The tumour measured more than 10 cm in dimension, the mitotic rate was of 15/50 high-power fields, with abundant tumour necroses. The tumour was focally positive for KIT (CD117). Genotypic analysis revealed a mutation of the PDGFRA gene.
Excised gastrointestinal stromal tumour. Note that tumour capsule is absent owing to tumour rupture. Fresh hematoma (white arrow) is present within the tumour (black arrow). Arterial stump (black arrow with A) responsible for peritoneal bleeding can be seen within the tumour.
The patient had no evidence of tumour progression on CT and positron emission tomography 18 months postoperatively. No adjuvant treatment was given.
Discussion
Our case suggests that GIST should be considered when acute nontraumatic hemoperitoneum is present, particularly if a heterogeneous mass consisting of a necrotic cavity and high vascularity is detected on CT.1
Complete surgical resection is the mainstay and the only radical treatment for GIST.1,2,5 Completeness of the primary resection (R0 v. R1 or R2) influences the prognosis. This is mainly important in low- and intermediate-risk GIST, whereas the prognosis for malignant GIST is primarily dictated by tumour aggressiveness.5 Resection of the primary lesion in metastatic GISTs is warranted for palliation of local symptoms and mostly to prevent bleeding. For these reasons, we believe that emergency surgery is the treatment of choice for severely hemorrhagic GIST. In the present case, complete resection was achieved by distal gastrectomy with more than 2 cm of tumour-clear margins, which is the recommended approach for GIST resection.1,2,5 Abundant peritoneal lavage should be performed in cases of tumour rupture in an attempt to reduce the risk of peritoneal tumour spillage. For this purpose, irrigation with distilled water, for its cytolytic activity on suspended cells, followed by a large volume of saline solution could be used.
The development of imatinib mesylate has dramatically improved the prognosis for metastatic GIST.1 Until now there has been no place for adjuvant therapy in patients treated surgically for ruptured GIST outside research protocols. 2 In fact, there is no proof that adjuvant treatment with tyrosine kinase inhibitor improves patient survival.2
Footnotes
Competing interests: None declared.
- Accepted July 7, 2007.