Abstract
The hepatitis C virus (HCV) infects more than 250 000 Canadians, many of whom are at increased risk of perioperative complications with surgery and all of whom represent an important risk for the entire surgical care team. The virus likely remains undiagnosed in one-third, while others have a limited understanding of the implications of the disease. All face an increased risk of liver failure and hepatocellular carcinoma. We review hepatitis C infection, including the challenges presented in operating on a patient with HCV infection and the inherent risks for the care team.
Nearly 3% of the world’s population, some 180 million people, is estimated to be infected with the hepatitis C virus (HCV).1 Chronic HCV infection has almost doubled in the last decade; it is now the leading indication for liver transplantation in developed nations and will continue to pose an important health and economic burden during the next 10–20 years. Although the incidence of HCV infection has decreased, the number of HCV-related deaths is likely to rise over the next decade owing to the long latency of the disease.2 A study of the population of the United States for 2010–2019 suggests that the morbidity and mortality associated with chronic HCV will increase dramatically, resulting in 165 900 deaths from chronic liver disease, 27 200 deaths from hepatocellular carcinoma and US$10.7 billion in direct medical expenditures related to HCV.3–5
The prevalence of chronic HCV in Canada is relatively low, believed to be between 0.8% and 1.0%, but is likely to increase because of an increasing number of people with chronic disease and because of migration from highly endemic areas such as Africa and Asia. Currently, 65% of all estimated infections in Canada have been identified.2
Hepatitis C virus poses a particular hazard to health care workers; 2.6% of all health care workers are exposed to HCV infection every year, translating into 16 000 new infections and 142 deaths annually worldwide.6,7 Estimates suggest that hospital-based health care personnel sustain 385 000 needlesticks and other sharps-related injuries each year — an average of 1000 sharps-related injuries per day, 25% of which occur in the operating room.4
Surgeons are likely to have a much higher lifetime risk of exposure to blood-borne viral pathogens than other health care workers. Given the high prevalence of the disease, it is very likely that a proportion of patients presenting for elective or emergency surgery are infected with HCV. It is thus important for surgeons to be aware of the epidemiology and infectious risk of operating on such patients and of management guidelines for health care workers inadvertently exposed to the disease.
We review the epidemiology, clinical course and the complications of HCV infection, as well the impact of the disease on the clinical care of these patients and the risk to the care team. We also discuss the guidelines for the management of accidental exposure to HCV.
Epidemiology
Hepatitis C virus belongs to the Flaviviridae family. There are 6 major genotypes and more than 50 subtypes. Among the 6 known HCV genotypes, most patients in Canada and the United States have either genotype 1 (70%–80%) or genotypes 2 and 3 (20%–30%). Whereas the genotype has very modest impact on the clinical outcome of the disease, it influences the response to and the duration of antiviral treatment.6,8
Mode of infection
The incidence of post-transfusion HCV has dramatically declined since the introduction of HCV screening for blood products in 1991. Currently, the sharing of needles among intravenous drug users is the most common cause of HCV infection, accounting for most new infections in Canada. Immigration from high-prevalence areas accounts for 33% of all cases.2
Most infections occur by percutaneous or intravenous introduction of the virus into the bloodstream, which is most commonly due to shared or unsterile needles. The virus is transmitted relatively inefficiently through sexual intercourse; 6% of all infections are thought to be acquired owing to unprotected sex. The risk of transmission from mother to fetus is small, but vertical transmission of HCV is higher in HIV-positive women.9
Clinical course
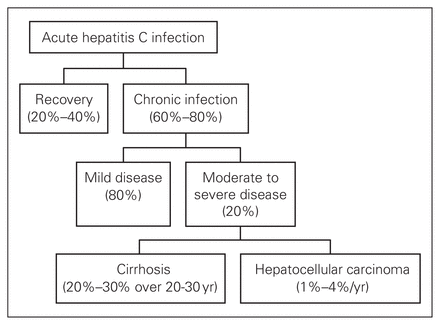
Acute HCV infection is usually subclinical: overt hepatitis develops in 10% of infected patients.1 Fulminant HCV is rare, but may occur more commonly in liver transplant recipients. The incubation period is around 7 weeks but can range from 2 to 12 weeks. Anorexia, myalgia and right upper quadrant pain may be present but are nonspecific. Chronic infection develops in about 60%–80% of all infected patients, and cirrhosis may be expected to develop in 20%–30% of all such patients over 20–30 years (Fig. 1). Hepatocellular carcinoma develops in 1%–4% of patients with cirrhosis every year. Several viral and host factors affect the rate of progression to fibrosis (Box 1).8
Natural course of hepatitis C infection.
Factors affecting the progression of hepatitis C infection to cirrhosis
Viral factors
Hepatitis C virus RNA load
Genotype 1b
Host factors
Age at infection
Male sex
Nonwhite race
Coinfection (hepatitis B virus, HIV-1)
Comorbid disease (iron overload, nonalcoholic steatohepatitis)
Genetic polymorphisms
Disease expression (elevated levels of alanine aminotransferase, stage of fibrosis at diagnosis)
Metabolic factors (obesity, insulin resistance, steatosis)
Immunosuppression (organ transplantation)
Other factors
Alcohol (greater than 50 g/d)
Smoking
Environmental toxins
Diagnosis
Diagnostic tests for HCV infection include serological assays for antibodies and molecular techniques for detecting virus particles. Positive antibody test results usually require confirmation of HCV RNA using a molecular amplification assay to provide an assessment of active viral replication. Quantification of HCV RNA provides important prognostic information on the likelihood of response to antiviral therapy. Several quantitative and qualitative assays are available with dynamic ranges between 101 and 107 IU/mL. Routine liver function tests have limited diagnostic value as more than 50% of patients infected with HCV will have normal transaminase levels.10
Liver biopsy remains the most accurate measure of the extent of liver disease, and assessing necroinflammatory activity and liver fibrosis provides useful information for determining requirements for antiviral therapy, prognosis and the potential for progression of fibrosis. Most clinicians base the decision for antiviral treatment on the necroinflammatory activity on liver biopsy, but this may be unnecessary for patients with genotype 2 or 3 infection.10
Treatment
The current standard of care for treating previously untreated patients with chronic HCV infection is combination pegylated interferon-α by subcutaneous injection once weekly and oral ribavirin daily. Combination pegylated interferon-α and ribavirin therapy can achieve a sustained virological response in 54%–56% of patients, including 42%–46% of patients with genotype 1 infection and about 80% of those with genotype 2 or 3 infection. Patients with genotype 1 infection require 48 weeks of pegylated interferon-α therapy with daily ribavirin, whereas patients with the more treatment-favourable genotypes 2 and 3 need be treated for only 24 weeks. Patients with very high levels of HCV RNA respond less well to antiviral therapy than do those with lower levels. In the postexposure scenario, a sustained course of therapy can clear the virus in 90%–100% patients.10,11
Care of patients with HCV
A patient with HCV may present to a surgeon either for surgery related to complications of HCV (e.g., surgery for hepatocellular carcinoma or a liver transplant) or, more commonly, for an entirely unrelated problem. Patients with decompensated liver disease and complications of cirrhosis are best managed by specialized multidisciplinary liver units. The risk of exposure to HCV varies for different types of surgery but increases in general with longer, more invasive procedures with higher blood loss.
Surgical and anesthetic concerns
Patients with liver disease are at particularly high risk for morbidity and mortality due to both the stress of surgery and the effects of general anesthesia. Decompensated liver disease increases the risk of postoperative complications (e.g., acute hepatic failure, sepsis, bleeding). With cirrhosis, the reciprocal flow relation between the hepatic artery and the portal vein is not well maintained, and the liver architecture is disrupted by fibrosis and regenerative nodules. These pathologic alterations of the normal patterns of liver blood flow make the cirrhotic liver more prone to ischemia.12
Prediction of surgical risk is based on the degree of liver dysfunction, the type of surgery and the preclinical status of the patient. Comorbid conditions responsible for perioperative morbidity and mortality (e.g., coagulopathy, intravascular volume, renal function, electrolytes, cardiovascular status, nutrition) should be identified and addressed before surgery. Risk stratification of patients with liver disease is usually performed using the Childs–Pugh score or the Model for End-Stage Liver Disease (MELD) score. The MELD score is based on a patient’s serum bilirubin, creatinine and international normalized ratio for prothrombin time and can be calculated using an online calculator.13 On average, patients with Child–Pugh class A disease can expect a 10% perioperative morbidity and mortality rate with intra-abdominal surgery. Predicted perioperative morbidity and mortality rates are 30% and 82% for patients with class B and class C cirrhosis, respectively.14 Elective surgery for patients with class C cirrhosis should be avoided without a comprehensive multidisciplinary preoperative assessment from anesthesiologists, surgeons and hepatologists.
Of all the inhaled anesthetics, halothane and enflurane appear to reduce hepatic artery blood flow the most because of systemic vasodilation and a mild negative inotropic effect.15 Halothane is also associated with the greatest risk of hepatotoxicity. Isoflurane is a safer choice than halothane in patients with liver disease because it may increase hepatic blood flow. Newer haloalkanes, such as sevoflurane and desflurane, also undergo less hepatic metabolism than halothane or enflurane. Atracurium has been recommended as the neuromuscular blocking agent of choice because it relies on neither the liver nor the kidneys for excretion. Likewise, drugs such as morphine, meperidine, benzodiazepines and barbiturates should be used with caution because of their dependence on the liver for metabolism.
The type and duration of surgery and amount of blood loss adversely affect outcome in patients with decompensated liver disease. In a study at the Mayo Clinic, investigators reviewed 733 patients with cirrhosis who underwent surgery.12 They found an 11.6% perioperative mortality rate (within 30 days of surgery) and a 30.1% complication rate. Emergency surgery is an important predictor of adverse outcome. The general considerations for anesthesia and surgery in patients with liver disease are listed in Box 2.14
Surgery and anesthesia in patients with liver disease
Preoperative assessment
Surgery is contraindicated in patients with Child–Pugh class C, acute hepatitis, severe coagulopathy, severe chronic hepatitis or severe extrahepatic manifestations of liver disease (e.g., acute renal failure, hypoxia, cardiomyopathy).
Avoid surgery if possible in patients with a MELD score ≥ 8 or Child–Pugh class B unless they have undergone a thorough preoperative evaluation and preparation.
Use caution with sedatives and neuromuscular blocking agents.
Optimize medical therapy for patients with cirrhosis.
Anesthetic considerations
Correct coagulopathy with vitamin K and fresh frozen plasma to achieve prothrombin time within 3 seconds of normal.
The goal platelet count is > 50–100 × 103/L.
Minimize ascites to decrease risk of abdominal-wall herniation, wound dehiscence and problems with ventilation. Address nutritional needs.
Monitor renal function.
Monitor and correct electrolyte abnormalities, especially hypokalemia and metabolic alkalosis.
Perform close postoperative monitoring.
Postoperative care
Admission to the intensive care unit may be appropriate after prolonged surgeries, intraoperative hypotension, excessive blood loss or cardiac and/or pulmonary surgery.
Monitor for signs of acute liver failure, including worsening jaundice, encephalopathy and ascites.
MELD = Model for End-Stage Liver Disease.
Adapted with permission from eMedicine.com, 2009.19
Injury in an operating room
Cuts or needlestick injuries may occur in up to 15.0% of all surgeries (range 1.7%–15.0%). Surgeons and first assistants sustain 60% of such injuries, but all health care personnel working in a surgical suite are vulnerable. Suture needles account for 77% of such injuries, and most injuries occur while closing fascia or muscle.7 Most injuries are self-inflicted. One-third of devices causing injury come in contact with the patient after the event, thus raising the possibility of disease transmission from the surgeon to the patient. The risk of HCV transmission depends on the volume of blood transmitted, but less than 0.5% of such injuries are high risk (e.g., hollow-bore needle). Surgeons do not report 70% of such injuries and often fail to participate in postexposure strategies.
Nevertheless, the risk of an individual surgeon becoming infected with HCV remains small, even in areas with extremely high prevalence of the disease. The estimated probability for such transmission is 0.001%–0.032% per year, translating into a 0.035%–1.120% risk over a 35-year professional career. Liver transplant surgeons, who are at particular risk of exposure, are estimated to have a 0.0009%–0.001% per year risk of infection (0.96–1.35 HCV transmissions per 1000 years) for surgical practice and a 0.001%–0.002% per year risk of infection (1.46–2.23 HCV transmissions per 1000 years) for liver transplant practice.16,17
Risk to health care workers
The prevalence of HCV in health care workers ranges from 0.5% to 3.0%, which is almost the same as the general population.18 Most infections in health care workers do not appear to be occupationally related.19 However, the risk of blood-borne virus transmission after percutaneous exposure is 1.8% (range 1.0%–7.0%), which is nearly 6 times higher than the comparable risk for HIV transmission. The average health care worker sustains 0.5–4.7 sharps-related injuries per year. Almost all HCV infections are due to percutaneous transmission, but isolated cases of transmission through mucus membranes and non-intact skin have also been reported.20,21
Risk to patients
The risk of infection to a patient from an HCV-infected surgeon is small. Six documented cases of transmission were reported after surgery by a cardiac surgeon who was subsequently found to have a high viral load (107 IU/mL of blood). Six other cases of transmission have also been reported.10
Universal precautions
Universal precautions for handling blood and body fluids were first recommended by the Centers for Disease Control in 1987 and have been updated regularly since.23 Published data suggest low compliance for universal precautions among surgeons, but these are the most important primary preventive measures to stop HCV infection.
A patient with known HCV infection should preferably be the last patient of the day to undergo surgery, with only the essential personnel present. Sharps-related injuries can be minimized by the creation of a neutral zone in which to pass instruments (hands-free technique), the use of needle-less instruments, staples and blunt needles to close fascia and muscle. Effective barrier precautions such as plastic aprons, face masks, goggles or shields and water-resistant aprons should be used. Double-gloving decreases the risk of exposure to patient’s blood by 87% and the transmitted volume of blood by 95%, thus decreasing the potential viral load from an infected patient.7
All health care workers performing high-risk procedures, as defined by the appropriate professional organizations, should be aware of their serologic status.
Management of exposure to HCV
Health care workers who are exposed to HCV should initially undergo adequate wound management. An assessment of infection risk should be made based on the type and severity of exposure and the blood-borne infection status of the source person.
Wound management
The wound should be cleaned with soap and running water. Mucous membranes must be washed with water. There is no evidence to support the use of antiseptic agents or of squeezing (“milking”) puncture sites. Bleach and caustic agents should not be used.
Assessment of infection risk
The type of exposure should be reported (e.g., percutaneous, mucous membrane, nonintact skin, bites resulting in blood exposure), as should the body substance (e.g., blood, potentially infectious fluid or tissue). The source person should be tested for the presence of HBsAg, HCV antibody and HIV antibody. If the source is unknown, the epidemiologic and clinical evidence should be assessed: namely, where and under what circumstances the exposure occurred and the prevalence of HBV, HCV or HIV in the population group.
Postexposure management
The exposed health care worker should undergo a baseline antiHCV titer and liver function test. Demonstration of HCV RNA has little role in the initial workup. There is no benefit of immunoglobulin administration in this setting. Additionally, interferon-α has limited benefit in the absence of hepatitis or in acute disease and is not recommended prophylactically. Currently, there is no available prophylaxis for HCV.
The health care worker should be offered serial anti-HCV testing, along with liver function tests every 6, 12 and 24 weeks after exposure. No special precautions need to be taken, and there is no need to modify patient care activities or sexual practices in the interim. Health care workers should not donate blood products, organs or semen until confirmed to be free of HCV. A positive antiHCV test should be confirmed by demonstration of HCV RNA by polymerase chain reaction. If 2 sequential antiHCV titers are positive, then data from the literature suggest that therapy (interferon-α or pegylated interferon-α with or without ribavirin) may lead to viral clearance in almost all patients. The current University of Alberta guidelines recommend treatment if HCV RNA–positive status fails to clear after 3–6 months of follow-up. Current directions of study include neutralizing antibodies to HCV, antiHCV vaccines or serine protease inhibitors, which are under trial and may eventually be used for postexposure prophylaxis.8,18
The HCV-infected health care worker
There is very little evidence that suggests that patients are at increased risk owing to surgeons with chronic HCV infection. The overall probability of transmission in such a situation is estimated to be 0.0001% (or 1 per 10 000 procedures).22 The American College of Surgeons position statement on hepatitis suggests that surgeons with chronic HCV infection have no reason to alter their practices but should adhere to strict infection-control procedures and use barrier precautions. We believe that the ethics of disclosure support notifying patients before a procedure whether the surgeon is HCV-positive. There may, however, be confidentiality concerns, as patients are not bound by the same confidentiality requirements as treating physicians. Surgeons infected with HCV should seek expert medical help, because interferon-α and ribavirin treatment can clear the virus in more than 50% of people with chronic infection.23
Conclusion
The patient infected with HCV presents a unique set of challenges. Surgery in such an individual demands careful preoperative evaluation and meticulous perioperative care to minimize morbidity and mortality risk. Whereas surgeons are occupationally prone to exposure to blood-borne diseases, the risk of contracting HCV is small. Prevention of HCV infection is currently only possible through the prevention of blood exposure. Surgeons have a duty of care to every patient, and no one should be denied appropriate medical treatment because of their HCV status.
In case of accidental exposure, prompt adverse event notification as per local policies, wound management and serial follow-up by serological testing should be undertaken. Current data do not support empirical therapy for HCV exposure, but acute HCV infection can be effectively treated with interferon-α and ribavirin.
Footnotes
Competing interests: None declared.
Contributors: Dr. Kneteman designed the study and reviewed the article. Dr. Asthana acquired and analyzed the data and wrote the article. Both authors approved the final version for publication.