Abstract
Background: Acute compartment syndrome (CS) is a limb-threatening disease that results from increased intracompartmental pressure. The pathophysiologic mechanisms by which this occurs are poorly understood. This study was designed to measure the effects of increased intracompartmental pressure on skeletal muscle microcirculation, inflammation and cellular injury using intravital videomicroscopy (IVVM) in a clinically relevant small animal model.
Methods: We induced CS in 10 male Wistar rats (175–250 g), using a saline infusion technique. Intracompartmental pressure was controlled between 30 and 40 mm Hg and maintained for 45 minutes. After fasciotomy, the extensor digitorum longus muscle was visualized using IVVM, and perfusion was quantified. We quantified leukocyte recruitment to measure the inflammatory response. We measured muscle cellular injury using a differential fluorescent staining technique.
Results: The number of nonperfused capillaries increased from 12.7 (standard error of the mean [SEM] 1.4 ) per mm in the control group to 30.0 (SEM 6.7) per mm following 45 minutes of elevated intracompartmental pressure (CS group; p = 0.031). The mean number of continuously perfused capillaries (and SEM) decreased from 78.4 (3.2) per mm in the control group to 41.4 (6.9) per mm in the CS group (p = 0.001). The proportion of injured cells increased from 5.0% (SEM 2.1%) in the control group to 16.3% (SEM 6.8%) in the CS group (p = 0.006). The mean number of activated leukocytes increased from 3.6 (SEM 0.7) per 100 μm2 in the control group to 8.6 (SEM 1.8) per 100 μm2 in the CS group (p = 0.033).
Conclusion: Early CS-induced microvascular dysfunction resulted in a decrease in nutritive capillary perfusion and an increase in cellular injury and was associated with a severe acute inflammatory component.
Acute limb compartment syndrome (CS) is characterized by raised pressure within a closed fascial compartment.1–7 If left untreated, CS may lead to tissue necrosis and permanent functional impairment.4,8–10 The clinical sequelae of CS, first described by Richard von Volkmann in 1875, relates irreversible contractures of the hand to an ischemic process in the forearm; von Volkmann put forward the idea that the pathophysiology of the contracture is caused by arterial insufficiency combined with venous stasis.11 Despite the breadth of research dedicated to understanding the pathophysiology of CS, the mechanisms causing the tissue and microvascular injury associated with acute CS are complex and remain only partly understood. Factors hindering our understanding of CS pathophysiology include the limitations in clinical trials owing to the severe acuity of CS, the absence of a clinically relevant standardized animal model and the difficulty of applying invasive tools to help delineate the pathways that propagate the CS injury at a cellular level.
Intravital videomicroscopy (IVVM) is a modern technique allowing for the visualization and study of microvascular perfusion.12 This technique has previously been used in the study of ischemia reperfusion, ischemic preconditioning, sepsis and other disease states that may compromise blood flow.12–16 The purpose of this study was to develop a clinically relevant small animal model of elevated intracompartmental pressure and to employ IVVM to study the microvascular and inflammatory response to CS.
Methods
Animal description and care
The male Wistar rats used for these experiments had access to food and water ad libitum. All protocols and experiments were conducted in agreement with the Committee on the Care and Use of Laboratory Animals of the Institute of Laboratory Animals Resources, National Research Council, and were approved by the institutional Council on Animal Care.
Experimental protocol
Ten rats (175–250 g) were anesthetized with inhalational isoflurane. After induction at 5% isoflurane in a 1:1 O2:N2 mixture, anesthesia was maintained at 2% isoflurane and titrated to maintain general anesthesia. We cannulated the carotid artery for continuous blood pressure monitoring and fluid replacement to maintain a normal mean arterial pressure of 100 mm Hg. Once the rats were anesthetized, we elevated compartment pressure by slowly infusing isotonic normal saline via a 24-gauge angiocatheter into the anterior compartment of the left hind limb in the experimental (CS) group. Compartment pressure was raised to 30 mm Hg and maintained between 30 and 40 mm Hg for the duration of the protocol. An electronic compartmental pressure monitoring system (Synthes USA) was inserted into the anterior and then posterior compartments through a 14-gauge angiocatheter. As the pressure rose within the hind limb, both the anterior and posterior compartments became isobaric (both anterior and posterior compartment pressures were raised to 30–40 mm Hg). To test the effect of time on capillary perfusion and cellular injury, we maintained elevated intracompartmental pressure (EICP) for 45 minutes (n = 5) before the release of the EICP via fasciotomy. Control animals (n = 5) underwent the same preparation; however, no saline was infused into the compartment via the catheter, and we maintained intracompartmental pressure at control levels for the duration of the experiment before fasciotomy.
Surgical technique
The extensor digitorum longus (EDL) muscle was prepared for IVVM, as previously described.12,16,17 In brief, the exposure of the EDL muscle began by incising the skin over the posterior aspect of the hind limb. We retracted the underlying biceps femoris muscle to expose the tibialis anterior and the lateral gastrocnemius muscles. These muscles were divided to expose the EDL, and the overlying fascia was incised. We applied a suture ligature around the distal tendon of the EDL. We then cut the tendon from its bony insertion to allow the EDL to be reflected onto the microscope stage with its proximal arterial and venous pedicle intact. Once prepared, animals were placed onto the stage of an inverted microscope (Nikon Diaphot 300), and the EDL was reflected onto a slide moistened with saline. A cover slip was placed on top of the EDL, and all exposed tissues were covered with a plastic film to isolate the preparation from the atmosphere and to prevent drying. A heat lamp maintained the EDL muscle temperature (32°C) as well as the core temperature (37°C) of the rat. Care was taken to ensure that the time from fasciotomy to the first microscopy recording was no more than 5 minutes.
Intravital microscopy and video analysis
The muscle preparations remained on the microscope with intact circulation postfasciotomy. We randomly chose 5 fields of view within the EDL containing a complete microvascular unit (arteriole, capillary bed and post-capillary venule). These fields were recorded onto video using a 20× objective, for a final magnification of 700× on the monitor. The microscope was connected to a charged-coupled device camera (Dage-MTI VE1000), a time–date generator (WJ-810, Panasonic) and a computer. Appropriate white light illumination was obtained using fibre-optic guides. One-minute video recording of each field of view was obtained postfasciotomy and stored on the computer for later analysis. We recorded an additional 15 seconds for the nuclear dye staining. This period was limited to reduce exposure to excitation wavelength to preserve the fluorochrome contained within the dyes.
Perfusion analysis
We determined an index of CS-induced microvascular dysfunction by counting the number of perfused capillaries crossing 3 equidistant parallel lines drawn on the computer monitor perpendicular to the capillary axis and expressed as the number of perfused capillaries by red blood cells per millimetre line length, following our previously validated methodology.12,14–16,18
Injury analysis
After fasciotomy, we added the fluorescent vital dyes ethidium bromide (EB, 5 μg/mL) and bisbenzimide (BB, 5 μg/mL) to the saline bath, as previously described.16,19 The topical use of EB and BB does not alter microvascular perfusion and is a reliable technique for cellular labelling in live animals.19
Bisbenzimide, a membrane-permeant dye, stains the nucleus of all cells. Ethidium bromide, a larger molecule, is membrane impermeant and acts to stain the nuclei of cells with injured (permeable) membranes.16,19 Since EB labels cells with a range of injuries from minor injury (increased permeability) to cellular death, this technique cannot distinguish injury from cell death. Fluorescent illumination with the appropriate filters for EB (Ex = 482 nm; Em = 610 nm) and BB (Ex = 343 nm and Em = 483 nm) were applied. We examined tissue injury in 5 fields of view for animals in the control and CS groups. Cellular injury was expressed as the ratio of EB-labelled nuclei to BB-labelled nuclei (EB:BB).16,19
Analysis of leukocytes
Leukocyte rolling and adherence were observed in post-capillary venules using the 40× objective (final magnification 1400×) postfasciotomy. The total number of rolling and adherent leukocytes was measured over 30 seconds and expressed as the number per 100 μm2. An adherent leukocyte was defined as a cell that remained stationary for a minimum of 30 seconds. Measurements of rolling and adhered leukocytes from each of the 5 fields of view were observed in both the control and CS groups.
Statistical analysis
Statistical analysis consisted of a repeated-measures 2-way analysis of variance (ANOVA) to compare the degree of perfusion, muscle injury, leukocyte rolling and leukocyte adherence with the presence of CS. We considered results to be significant at p < 0.05.
Results
Microvascular dysfunction
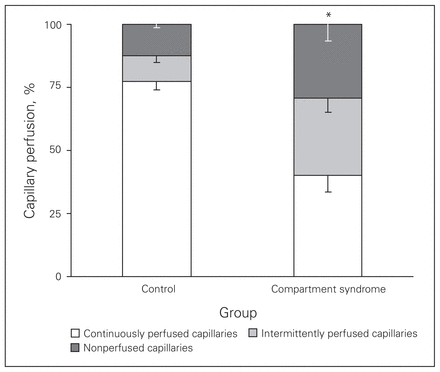
The effects of increased duration of EICP on capillary flow are shown in Figure 1. The capillary profile observed in control animals demonstrates predominantly continuous perfusion, representing normal healthy perfusion.
The effect of elevated intracompartmental pressure on microvascular perfusion measured using intravital videomicroscopy. The graph represents the overall surface microvascular perfusion within the extensor digitorum longus muscle when subject to elevated pressure as a function of time. *Continuous and intermittently perfused capillaries after 45 minutes (compartment syndrome [CS] group) are significantly different than that in the control group (p = 0.001 and p = 0.021, respectively). *The number of nonperfused capillaries increased (p = 0.031) in the CS group compared with the control group.
The mean number (and standard errors of the mean [SEM]) of continuously perfused capillaries decreased from 78.4 (3.2) per mm in the control group to 41.4 (6.9) per mm in the CS group (p = 0.001). Perfusion shifted from a predominantly continuous profile in the control group to an intermittent and nonperfused profile in the CS group. There was an increase in the number of intermittently perfused capillaries from 10.4 (SEM 2.7) per mm in the control group to 31.4 (SEM 6.0) per mm in the CS group (p = 0.021). The number of nonperfused capillaries increased from 12.7 (SEM 1.4) per mm in the control group to 30.0 (SEM 6.7) per mm after 45 minutes of EICP in the CS group (p = 0.031).
Inflammation
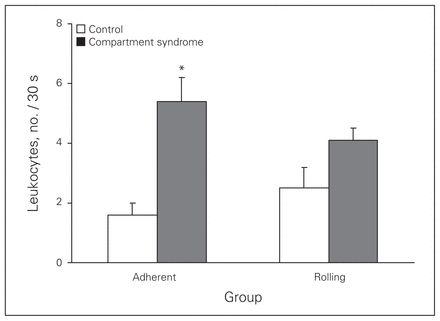
Leukocyte number and flow characteristics increased in response to CS. The mean number of activated leukocytes increased from 3.6 (SEM 0.7) per 30 seconds in the control group to 8.6 (SEM 1.8) per 30 seconds in the CS group. Rolling leukocytes increased from 2.5 (SEM 0.7) per 30 seconds in the control group to 4.1 (SEM 0.4) per 30 seconds in the CS group. Adherent leukocytes significantly increased from 1.6 (SEM 0.4) per 30 seconds in the control group to 5.4 (SEM 0.8) per 30 seconds in the CS group (p = 0.022; Fig. 2).
Leukocyte adherence and rolling in postcapillary venules observed in the control group and after 45 minutes of elevated intracompartmental pressure (compartment syndrome group). *An early and significant (p = 0.022) difference in leukocyte adherence was noted. In inflamed tissue, leukocyte rolling leads to a stationary state in which the leukocyte remains firmly attached to the endothelial cell surface without motion. This high-affinity adhesive interaction (leukocyte sticking or adherence) denotes the absence of movement of the leukocyte along the length of the venule.
Tissue injury
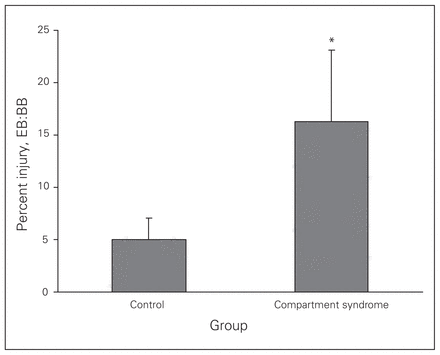
Muscle injury was quantified as the ratio of EB:BB stained nuclei and represents the percentage of injured cells per field (Fig. 3). After application of the fluorescent dyes, the control group demonstrated a baseline level of tissue injury (5.0%, SEM 2.1%), presumed to be secondary to tissue handling during surgical preparation. There was a sudden and significant (p = 0.006) increase in the percentage of injured cells (16.3%, SEM 6.8%) in the CS group.
The effect of elevated intracompartmental pressure on parenchymal tissue injury within the extensor digitorum longus muscle. Sham muscles (0 min) have a low baseline level of parenchymal injury, indexed by the number of ethidium bromide (EB)–labelled nuclei relative to the bisbenzimide (BB)-labelled nuclei. *After 45 minutes of elevated intracompartmental pressure (compartment syndrome CS group) a significant increase (p = 0.006) in muscle cellular injury was noted.
Model characteristics
Carotid artery cannulation demonstrated a normotensive model throughout the duration of CS. Mean arterial pressure was maintained within physiologic limits (data not shown).
Discussion
We studied the effect of EICP on microvascular perfusion, tissue injury and inflammation in a small animal model of CS using IVVM and nuclear fluorescent dyes. Direct imaging of capillaries demonstrated a significant decrease in continuously perfused capillaries (p = 0.001) and a significant increase in intermittent and nonperfused capillaries (p = 0.021 and p = 0.031, respectively; Fig. 1). This observation characterizes the early microvascular response to the CS insult. Continuous perfusion is normal physiologic perfusion observed in uninjured microvasculature. The immediate response to CS is a shift to intermittent and nonper-fused capillaries. This state of diminished microvascular flow produces a non-nutritive perfusion with compromised gas exchange. Intermittent perfusion demonstrates a marked decrease in red blood cell flow, whereas in nonper-fused capillaries red blood cells have no movement. Post-fasciotomy, intermittently perfused capillaries may recover flow; however, nonperfused capillaries do not.20,21
This microvascular dysfunction is accompanied by a substantial inflammatory response. Activated leukocytes are categorized as rolling or adherent, and we measured them in the postcapillary venule. Leukocyte adherence was significantly increased (p = 0.022) in the CS group compared with the control group. There was no observed difference in leukocyte rolling between groups. After 45 minutes of EICP, the observed leukocyte adherence reflects a relatively early time course for leukocyte accumulation.18,22 Leukocyte arrest during rolling is triggered by chemo-attractants and is mediated by the interaction of integrins and immunoglobulins expressed by endothelial cells.23,24 The arrest of leukocytes under conditions of flow and the leukocyte recruitment and emigration observed suggests that CS induces a proinflammatory environment. The inflammatory activity seen in this model of CS exceeds the degree of inflammation noted in complete ischemia and early reperfusion models.18 The exact role of inflammation in muscle damage in CS is unknown but may contribute to the nonreflow of capillaries as well as cellular injury.
Parenchymal injury was evidenced by the sudden significant increase in the number of EB-labelled nuclei in the CS group compared with the control group (p = 0.006). Ethidium bromide is a fluorescent dye that does not penetrate the cell membrane of uninjured cells.19 Injured cells develop increased membrane permeability and allow EB to enter the cell and stain the nucleus, thereby reflecting the amount of injury within the capillary networks observed. Whether these cells are able to recover or become functionally viable remains unknown. This technique for detecting injury has been used in vivo for many years in studying microcirculation and ischemia reperfusion.12,14,16,19
Compartment syndrome as low-flow ischemia
After 45 minutes of EICP, nearly all of the capillaries observed in the EDL muscle displayed altered perfusion. Despite microvascular dysfunction in acute CS, some degree of perfusion remains at all times, creating a partial ischemic environment or low-flow ischemia within the limb. This allows neutrophils to be activated immediately, which may contribute to the degree of cellular injury noted.25,26
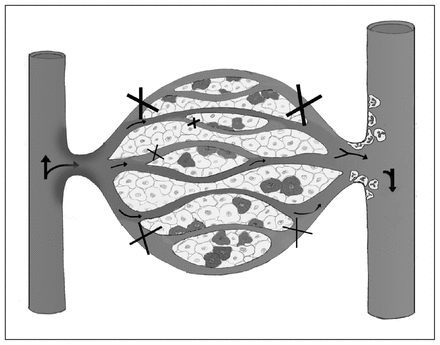
Following complete ischemia, revascularization leading to the reintroduction of oxygen into ischemic tissue results in an increase in reactive oxygen metabolites, initiating an acute state of inflammation.27–29 These reactive metabolites serve as a trigger to increase the overall rate of cellular apoptosis and necrosis.29 During EICP (30 mm Hg) in a normotensive model with partially sustained perfusion, a concurrent amplification of the inflammatory system from reactive metabolites may occur since oxygenated blood continues to perfuse the compartment, in contrast to complete ischemia. In a murine model comparing complete hind limb ischemia to partial ischemia, Conrad and colleagues30 reported that partial ischemia causes a significant early increase in the proinflammatory cytokine KC, which is analogous to human interleukin-8 expressing neutrophil chemotactic activity. This finding corroborates the early inflammatory response that we observed in CS, which we believe is physiologically similar to a partial ischemic state. In a canine model comparing complete ischemia to CS, Heppenstall and colleagues31 observed that the CS stimulus caused severe acidosis and metabolic stress. They also concluded that CS rendered a more severe degree of muscle ultrastructural deteroriation than ischemia alone.31 Compartment syndrome was found to be more injurious to muscle than complete ischemia, possibly owing to the cytotoxic inflammation induced by this low-flow ischemic state. Our physiologic model of CS included a low-flow ischemic state with associated inflammatory activation and muscle tissue injury (Fig. 4).
Proposed conceptual model of compartment syndrome (CS)–induced microvascular dysfunction. Oxygenated blood flows from the arteriole through the capillary, unloading oxygen to cells. With elevated compartmental pressure, nonperfused and intermittently perfused capillaries become visible within capillary beds and are ineffective at gas exchange (X), contributing to cellular injury (darkened cells). Furthermore, maintenance of capillary perfusion during CS allows oxygenated blood into the compromised compartment, which may lead to reactive oxygen metabolites contributing to the chemotactic stimuli for the expression and activation of leukocytes. In the postcapillary venule, activated leukocytes that may contribute further to tissue injury can be observed.
Compartment syndrome modelling
The severity and acuity of CS restricts the study of its pathophysiology in humans. Animal models have been used in the study of CS since 1926, when Jepson published an inaugural study in canines.11 He experimentally induced CS and detailed the functional benefit of decreasing venous obstruction via fasciotomy. Animal models of acute lower-extremity CS have been developed using various techniques in both large and small animals. Skin fold chambers, arterial occlusion via fogerty balloon, arterial ligation, inflation of latex balloons within compartments, external compression and tourniquet application are some of the techniques published.32–40 Large animal canine models deemed clinically relevant have induced CS using pressure-controlled autologous blood or plasma infusion into compartments.
In the present study, we used a model with pressure-controlled isotonic normal saline infusion in a rodent hind limb. We studied the EDL muscle, as it is composed of a mixture of muscle fibre types, with up to 54% of the muscle being fast twitch,17 similar to human anterior compartment musculature. The EDL preparation has been established in the study of microcirculation,14–19 its advantages being that it is a deep muscle and sustains minimal mechanical manipulation in its preparation and therefore minimal reactive hyperemia and injury. Most of the muscle remains in situ when its microcirculation is studied. Its surgical preparation does not demonstrate deterioration of perfusion with time and, hence, experimental controls can be easily applied.
The time chosen for EICP (45 min) was based on previous work demonstrating that 1 hour of ischemia in rodents approximates 4 hours of ischemia in humans.33 The experimental time of 45 minutes was applied to observe the early microvascular response to EICP and its subsequent effects on the surrounding tissue. Small animal models are not identical to metabolic and cellular derangements in humans, and, hence, experimental effects need to be compared with the existing body of literature. This model is reliable and simple to use for the study of microcirculation, inflammation and injury in acute CS and allows for the detailed study of the mechanism underlying CS.
Conclusion
To our knowledge, this study provides the first evidence of the in vivo microvascular perfusion changes that occur with early CS. The use of IVVM in conjunction with fluorescent stains in a small animal model has demonstrated the specific perfusion changes, inflammation and tissue injuries that occur in early CS. These data suggest that the injury process in CS begins early and causes a severe inflammatory response. Further study is required to fully delineate the mechanism causing the severe injuries observed clinically in CS.
Footnotes
Competing interests: None declared.
Contributors: Dr. Lawendy designed the study and wrote the article. Ms. Bihari acquired the data. All authors analyzed the data, reviewed the article and approved its publication.
- Accepted March 23, 2010.