Abstract
Background: The literature examining clinical outcomes and readmissions during extended (> 1 yr) left ventricular assist device (LVAD) support is scarce, particularly in the era of continuous-flow LVADs.
Methods: We completed a retrospective cohort study on consecutive LVAD patients from June 2006 to March 2015, focusing on those who received more than 1 year of total LVAD support time. Demographic characteristics, clinical outcomes and readmissions were analyzed using standard statistical methods. All readmissions were categorized as per the Interagency Registry for Mechanically Assisted Circulatory Support 2015 guidelines.
Results: Of the 103 patients who received LVADs during the study period, 37 received LVAD support for more than 1 year, with 18 receiving support for more than 2 years. Average support time was 786 ± 381 days, with total support time reaching 80 patient-years. During a median follow-up of 2 years, 27 patients died, with 1-year conditional survival of 74%. Median freedom from first readmission was 106 days (range 1–603 d), with an average length of stay of 6 days. Readmissions resulted in an average of 41 ± 76 days in hospital per patient. Reasons for readmission were major infection (24%), major bleeding (19%) and device malfunction/thrombus (13%). There were a total of 112 procedures completed during the readmissions, with 60% of procedures being done in 13% (n = 5) of patients.
Conclusion: Continuous-flow LVADs provide excellent long-term survival. The present study describes marked differences in reasons for readmissions between the general LVAD population and those supported for more than 1 year. Prolonged LVAD support resulted in decreased susceptibility to major bleeds and increased susceptibility to infection.
Heart failure (HF) is widespread among Canadians, with more than 500 000 total cases throughout the country and 80 000 new patients per year.1 Stage D heart failure, defined as the failure of medical therapy, necessitates intervention in the form of mechanical circulatory support or cardiac transplantation.1–4 Although the treatment of choice remains transplantation, donor heart shortages have led to increasing numbers of patients being bridged with a left ventricular assist device (LVAD).2–5 Clinically, there are increasing numbers of patients supported on LVADs for more than 1 year in direct relation to their blood type, body size and antibody sensitization.5,6 These devices improve outcomes and provide a reasonable quality of life for patients with HF. Often, short-term and long-term mortality decrease and patients with refractory HF experience improved symptom control when supported with an LVAD.7–9 Newer continuous-flow devices (CF-LVADs) have improved durability and have allowed prolonged support for patients. The Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) has reported 80% survival rates after 1 year of LVAD support, which is comparable to cardiac transplantation.10 These favourable outcomes have resulted in an increased use of CF-LVADs as a bridge to transplantation and destination therapy (DT). In fact, 40% of total LVAD implantations in the United States in 2012 were indicated as DT.8,10,11
Although implantation is considered relatively safe, adverse events and pre-existing comorbidities can lead to poor prognosis.12–14 Hospital readmissions reduce patient quality of life and impose a financial burden, particularly in the single-payer health care system that exists in Canada. 12–15 Patients who were enrolled in the Randomized Evaluation of Mechanical Assistance for the Treatment of Congestive Heart Failure (REMATCH) trial experienced higher rates of readmission after LVAD implantation than their medical counterparts (88 v. 24 d).8 This is of particular concern in patients supported for long periods of time with expected readmissions as a result of increased support times.
Caring for patients with implanted LVADs supported for more than 1 year presents the health care team with a unique set of medical concerns that are largely unknown. Previously described complications post-LVAD leading to readmissions include gastrointestinal and cerebrovascular bleeding events, infections and thromboembolism. 16–19 Unfortunately, although the proportion of patients with LVADs supported for more than 1 year is increasing, the literature exploring clinical outcomes, readmissions and procedures during prolonged LVAD support remains scarce. The goal of the present study is to report on clinical outcomes of patients supported for more than 1 year on a CF-LVAD. Additionally, we explored reasons for and predictors of readmissions and all subsequent procedures in our patient cohort.
Methods:
Study population
In this retrospective cohort study, we included consecutive adult patients undergoing CF-LVAD implantation between June 2006 and March 2015 at the Toronto General Hospital, with a focus on patients supported on LVAD for more than 1 year. Devices implanted included HeartMate II (HMII; St. Jude Corp), HVAD (Heartware) and Duraheart (Terumo) devices. Our institutional research ethics board approved our research protocol before the study began.
Exposures and outcomes
We collected data on patient demographic characteristics, preoperative comorbidities, preoperative laboratory values, operative values, postoperative medications, postoperative characteristics and postoperative complications and outcomes. Follow-up was 100% complete.
Device implantation and patient follow-up
Surgical implantation of the devices was performed according to previously described techniques.2–4,6 On discharge from hospital, all patients were followed at Toronto General Hospital until transplant or death. Appointments included a physical examination, investigation of the device, optimization of medical therapy and discussion of patient concerns. Visits occurred weekly until there were no active issues, at which point the visits were scheduled biweekly or monthly.
Readmissions and procedures
Readmission data included diagnosis, length of hospital stay (LOS) and interventional procedures performed. Reasons for readmission were categorized using the INTERMACS 2015 adverse events definitions. Elective transplant readmissions were excluded from our analysis. Two research team members (S.R. and F.F.) compiled all readmission and procedure data simultaneously.
Statistical analysis
Data are reported as means ± standard deviations for continuous variables or percentages for categorical variables. Factors associated with readmissions were examined. We compared characteristics between readmitted and nonreadmitted patients using 2-sided 2-sample t test analyses for normally distributed continuous variables. We used the Mann–Whitney U test for non-normally distributed data and the χ2 test for categorical variables. Survival analysis was completed using the Kaplan–Meier method, with censoring for transplantation. Statistical differences in survival were computed using the Mantel log-rank test. All comparisons were 2-sided, and we considered results to be significant at p < 0.05. Competing outcomes were analyzed by adding the incidence of each outcome over time. All statistical analyses were conducted using SPSS software version 22.0 (IBM Corp.).
Results:
Patients and devices
In total, 103 patients were implanted with a CF-LVAD between 2006 and 2015 (Table 1). Of those, 37 patients were supported for at least 1 year, with 18 (49%) patients receiving more than 2 years of support. Overall mortality was 29% (30 deaths) during a mean follow-up of more than 2 years (IQR 489.5–1030 d; Table 2). Baseline characteristics of patients receiving prolonged support are reported in Table 3 and Table 4. Average support time was 786 ± 381 days, amounting to a total of 79.6 patient-years. Indications for device implantation included bridge to transplant in 25 (68%) patients, bridge to candidacy in 6 (16%) patients and DT in 6 (16%) patients. The mean age of the recipients supported over 1 year was 52 ± 13 years, and 40% were women. Etiology of cardiomyopathy was ischemic in 16 (43%) patients, idiopathic in 13 (35%) patients, postchemotherapy in 5 (14%) patients, familial in 2 (5%) patients and hypertrophic (3%) in 1 patient. Devices implanted included 27 (73%) HMII, 8 (22%) HVAD and 2 (5%) Duraheart devices. The median postoperative hospital stay during the index admission was 27 days (range 11–33 d).
Demographic and clinical characteristics of patients who received short-term versus prolonged LVAD support
Overall survival on LVAD support (n = 103)
Preoperative characteristics of patients with prolonged LVAD support
Peri- and postoperative characteristics of patients with prolonged LVAD support
Survival with continuous-flow pumps
Survival of patients receiving LVADs, regardless of support time (n = 103), is shown in Table 3 and Table 4. The greatest risk for patients was during their index admission for LVAD implantation, accounting for 63% of all deaths. Table 3 and Table 4 demonstrate postoperative outcomes in the 37 patients who received prolonged support, stratified by readmission status.
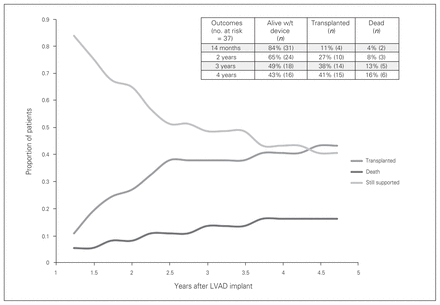
Figure 1 illustrates a competing outcomes analysis in patients with prolonged LVAD support. Transplantation rates increased at a higher rate than mortality. There were 9 patients supported on an LVAD for more than 1000 days. During a median follow-up time of just over 2 years (IQR 489.5–1030 d), 16 (43%) patients underwent transplantation and 6 (16%) died. Causes of death included sepsis, stroke, LVAD malfunction and 3 palliative cases where care was withdrawn.
Competing risk outcomes depiction for long-term left ventricular assist device (LVAD) support. The sum of the proportion of patients reaching the indicated end points equals 1.0 for each time point.
Readmissions
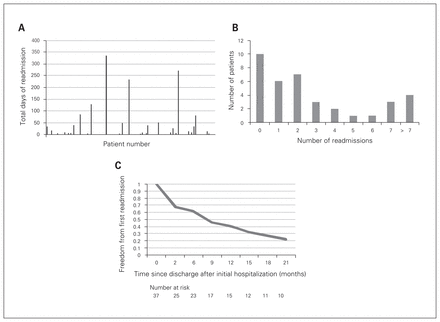
There were a total of 129 readmissions among the 37 patients on prolonged LVAD support. This amounted to 1499 hospital days (Fig. 2A). After index admission, 29 (73%) patients were readmitted at least once, 9 (24%) patients were readmitted 5 or more times, and 4 (11%) patients were readmitted at least 9 times (Fig. 2B). On average, patients were readmitted 3 times, for a median LOS per admission of 6 days in hospital. Patients who were readmitted spent an average of 41 days in hospital during their readmission. Median freedom from first readmission was 106 days (Fig. 2C). More than 60% of patients were readmitted within their first year of support.
A) Total readmission days were 1499, with the average being 41 days. B) There were a total of 129 readmissions among patients with prolonged left ventricular assist device (LVAD) support, with the average being 3 readmissions per patient. Although most patients (75%) were admitted at least once, 23% of patients had 5 or more readmissions and 10% of patients had more than 10 readmissions. C) The median time to first readmission was 106 days. More than 60% of patients were admitted within the first year of support (range of 1–584/603 days [latest readmitted/never readmitted]). The mean and median length of stay were 7 days and 5.5 days, respectively.
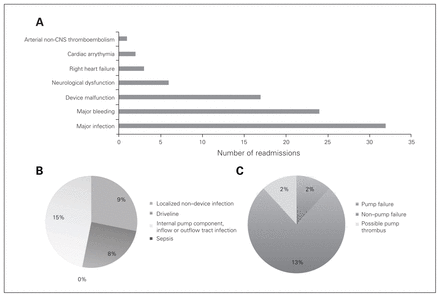
Reasons for readmissions are depicted in Figure 3A. The primary cause of hospitalization was major infection, amounting to more than 30 readmissions. Fifteen (38%) patients experienced a major infection during prolonged LVAD support, and 7 patients experienced recurrent infections. All patients with multiple readmissions (> 5) had at least 1 incidence of infection, and patients with more than 9 readmissions experienced recurring infections. Breakdown of reasons for readmissions according to INTERMACS shows that sepsis was the main cause of readmissions in this patient cohort (Fig. 3B). Although device malfunction was one of the top 3 causes for readmissions, pump exchange was required in only 3 patients (Fig. 3C). A substantial proportion (35%) of readmissions were classified as “other” and included vertigo, fever, dyspnea, syncope, fluid overload, fractures, ischemic colitis, anemia, increased international normalized ratio, slurred speech and back pain.
A) Reason for readmission (Interagency Registry for Mechanically Assisted Circulatory Support 2015). B) Etiologies for infection-related readmissions in patients with long-term left ventricular assist device (LVAD) support. C) Causes of device malfunction in long-term LVAD readmissions. CNS = central nervous system.
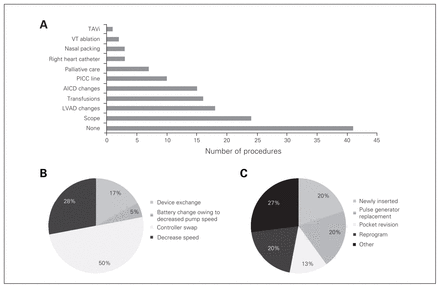
During readmissions, patients underwent several procedures, as shown in Figure 4A. Endoscopy and pump exchange accounted for a large number of the procedures (38%). Endoscopy was generally undertaken to determine the source of gastrointestinal bleeding and included gastroscopy, capsule endoscopy, sigmoidoscopy, colonoscopy and double balloon endoscopy. Device malfunction was related to controller requiring swap in 9 patients and to speed adjustments in 5 patients (Fig. 4B). There were 3 LVAD pump exchanges owing to thrombus. Twelve procedures were classified as “other” and included bone marrow biopsy, brachial emblectomy, femoral–femoral bypass, tissue plasminogen activator for LVAD alarm thought to be secondary to pump thrombus, transjugular liver biopsy, open appendectomy, kryphoplasty, vertebral biopsy, dialysis catheter insertion, enterocolysis and subtotal colectomy.
A) Procedures during readmissions. Most readmissions required supportive care without the need for procedures. B) Left ventricular assist device (LVAD) procedures. C) Automatic implantable cardioverter defibrillator (AICD) procedures. PICC = peripherally inserted central catheter; TAVi = transcatheter aortic valve implantation; VT = ventricular tachycardia.
Associations with readmissions
Of the 37 patients on prolonged LVAD support, 10 (27%) did not require readmission after the index admission for LVAD implantation. A postoperative infection during the index admission was the only variable found to be associated with future readmissions (p = 0.030). Other postoperative complications, including all-cause bleeding and ventricular arrhythmia, approached significance. Additionally, there was a higher prevalence of diabetes, hypertension, and previous cardiac surgery in the highly readmitted patient cohort.
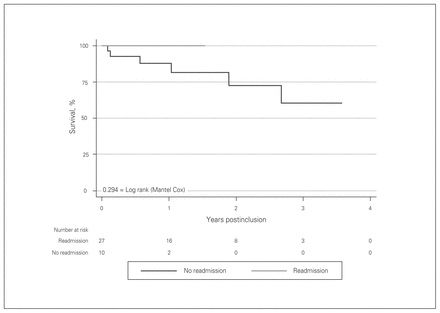
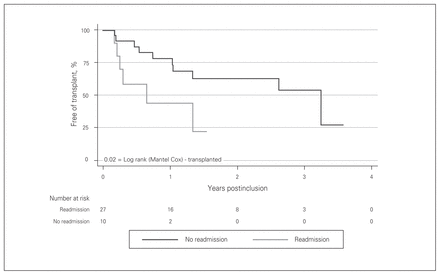
There were no significant differences in mortality between patients who were readmitted and those who were not (p = 0.29). Of note, there were 6 deaths in the readmitted population in contrast to no deaths among patients requiring no readmissions (Fig. 5). Patients who were readmitted at least once experienced longer wait times for transplantation than their nonreadmitted counterparts (Fig. 6, p = 0.021). Finally, risk of recurrent readmissions for patients who were readmitted even once after their index discharge was 78%.
Kaplan–Meier curve for overall survival in patients with prolonged left ventricular assist device (LVAD) support. There were no significant differences in mortality between those who were readmitted and those who were not (log rank = 0.294).
Kaplan–Meier curves for transplant wait times in patients with prolonged left ventricular assist device (LVAD) support. There were significant differences in transplant wait times in the readmitted versus nonreadmitted group (log rank = 0.02).
Discussion
This study explored our single-centre experience with CF-LVADs as a long-term therapy for patients with end-stage HF. Continuous-flow LVADs provide reasonable long-term survival and are suitable for bridging to transplant candidacy as well as DT. In our overall LVAD population, we report promising survival rates of 77% and 74% at 1 and 2 years, respectively. Patients requiring long-term support are increasingly susceptible to infection and device malfunction in comparison to the overall LVAD population.
Our survival rates of 77% at 1 year and 74% at 2 years are comparable to those in the large INTERMACS registry as well as those of several smaller centre analyses. 10,16–18 Increasing experience with device implantation and optimal clinical management of patients has led to improved outcomes in patients with LVADs. The first HMII trial reported a survival of 68%,3 which has now improved to 85%.20 In regards to long-term LVAD support, the HMII DT trial reported 2-year survival rates increasing from 58% to 63% in the current era.21 We report lower mortality at our centre, potentially attributable to our rigorous patient selection process and interprofessional LVAD education. Notably, our results implicated the index admission as the highest-risk time period for LVAD mortality, implicating patient selection as a major contributor. This holds promise for long-term LVAD support, which holds a mortality risk of 8% per year and decreases in relation to years of support. Our results serve as a reminder that overall outcomes for patients with HF have improved significantly. In the REMATCH trial, long-term (2 yr) survival was 8%.8 Although the HM1 was able to increase survival to 23%, nonpulsatile devices have improved that value to more than 60%.20 This illustrates that prolonged LVAD support is beneficial as both bridge to transplant as well as DT.
Patient baseline characteristics, LOS and time to transplant were akin to those previously reported in the literature and similar to that of the general LVAD population. 10,16–19,21 The limited literature examining postoperative LOS in the LVAD population varies widely, ranging from 17 days to 25 days.16,18 Compared with other centres, our centre leans toward having increased length of postoperative stays. At present in our centre, patients are discharged directly home after LVAD implantation, contrary to other institutions where patients are transferred to rehabilitation centres before discharge home. This is due to the lack of “alternate level of care” centres that accept LVAD-supported patients, which likely explains our increased postoperative hospital stays.
Readmissions in the general LVAD population have implicated bleeding, cardiac anomalies and infections as the major causative reasons, in order of prevalence.16–19 In contrast, reasons for 30-day readmissions were somewhat different, with recurrent HF taking the lead, followed by gastrointestinal bleeding.22 Long-term LVAD support predisposes patients to readmissions due to infections as opposed to bleeding events. Interestingly, unlike the overall LVAD population who were at increased risk of LVAD-related infections, the present population experienced increased admissions due to systemic sepsis. We must strive to reduce infections predominantly via increased patient education, focusing on long-term support and exploring prophylactic antibiotics options in high-risk groups. Contrary to our findings, Takeda and colleagues23 found that bleeding, cardiac causes and device-related morbidity were the leading causes of readmissions in long-term support. This discrepancy could be due to the difference in mean follow-up time between the studies. We followed patients for an average of 2 years and noticed a readmissions peak during the first year of support, which subsequently plateaued. Other studies have found similar peak readmissions at 1 year and then subsequently at 3 years.23 It would be worthwhile to further assess these readmissions in order to reduce patient morbidity and mortality. Of note, there was a subset of 4 (11%) patients who made up more than 40% (55 readmissions) of the total number of readmissions. To our knowledge, the present study was the first of its kind examining procedures sustained by patients during readmissions. Notably, although endoscopy, LVAD changes and transfusions were common in our patient cohort, most readmissions amounted to supportive care without additional procedures.
Although readmissions did not have a significant impact on overall mortality, they did increase time to transplant and support times. This delay could have been be due to procedures, particularly transfusions given to patients with bleeding complications. Additionally, patients enduring long-term LVAD support had significantly higher freedom from first readmission (106 d v. 51 d), suggesting that hospital admissions may play a role in time to transplant.16 It would be pertinent to further explore these variables and their effects on transplantation.
In the context of inadequate donor hearts as well as patient contraindications to surgery, there is active discussion surrounding CF-LVADs as an alternative to cardiac transplants. According to the International Heart Transplant registry, the current 1- and 5-year survival rates are 81% and 69%, respectively.24 Data from the INTERMACS registry on 1160 DT patients highlighted 1- and 2-year survival rates of 88% and 80%, respectively, in selected low risk patients.10 Additionally, Takeda and colleagues23 showed similar survival rates in their long-term supported patients. Our survival data also support prolonged use of CF-LVADs. That being said, although existing data are strongly suggestive of the need for a controlled trial, it is difficult to make conclusions on the best treatment option. Additionally, owing to frequent readmissions in this population, patient quality of life and health care costs must be considered as pertinent outcomes.
Limitations
The present study had the limitations of a descriptive retrospective analysis and had a small sample size. We are not funded for DT therapy, thus our DT population is relatively small compared with those of other series. There are a number of large multicentre studies depicting excellent outcomes in patients with LVADs supported for prolonged periods. The strengths of the present single-centre study were the consistent strategies in patient selection, operative procedure and postoperative care. Additionally, our centre serves a wide catchment area and is one of the largest LVAD centres in the country. Thus we believe the findings from our study should pertain to the general Canadian LVAD population. Furthermore, our study provided detailed analysis of readmissions and procedures that are not sufficiently addressed in multicentre studies. The INTERMACS registry demonstrates that patients have long support durations regardless of the initial implant approach.25 We did not capture readmissions to other hospitals; however, because our hospital is the only LVAD-specialized centre in the region, it is unlikely that patients would have been readmitted elsewhere for LVAD-related concerns. Finally, self-assessments of quality of life would have provided a much-needed patient perspective.
Conclusion:
Continuous-flow LVADs provide satisfactory long-term outcomes and are suitable for bridging to transplant candidacy as well as DT. Patients requiring long-term support may be less susceptible to major bleeds but increasingly susceptible to infection and device malfunction. Furthermore, the incidence of postoperative infections may serve as a strong predictor of future readmission to hospital. Our study provides novel Canadian data that describe resource utilization and clinical outcomes in patients receiving prolonged CF-LVAD support and, in our opinion, justifies the continued use of this therapy.
Footnotes
Presented at the Canadian Cardiovascular Congress in Toronto, Ont., Oct. 24–27, 2015
Funding: The present study received support from the Institute of Medical Sciences, University of Toronto and the Heart and Stroke Foundation.
Competing interests: V. Rao is a consultant to St. Jude Corp (Minneapolis, Minn.) and HeartWare International Inc. (Framingham, Mass.). No other competing interests declared.
Contributors: S. Raju, J. MacIver, C. Alba and V. Rao designed the study. S. Raju, F. Foroutan and C. Alba acquired the data, which all authors analyzed. S. Raju and V. Rao wrote the article, which all authors reviewed and approved for publication.
- Accepted January 6, 2017.