Summary
Noncompressible hemorrhagic control remains one of the most challenging areas in damage control medicine and continues to be a leading cause of preventable death. For decades, emergency thoracotomy or laparotomy and aortic cross clamping have remained the gold standard intervention. Recently, there has been a movement toward less invasive techniques for noncompressible hemorrhagic control, such as resuscitative endovascular balloon occlusion of the aorta (REBOA). The REBOA technique involves inflation of an endovascular balloon within the abdominal aorta proximal to the vascular injury to temporarily inhibit bleeding. Although the literature is robust on this new technique, skepticism remains about whether REBOA is superior to aortic cross clamping, as it has been associated with complications including organ and limb ischemia, limb amputation, femoral aneurysm, and thrombosis.
Resuscitative endovascular balloon occlusion of the aorta (REBOA) is a less invasive method of aortic occlusion as a temporary measure to manage noncompressible hemorrhagic control. This technique consists of a multitude of steps, including arterial access, sheath placement, catheter insertion, balloon introduction, position selection, balloon inflation, balloon deflation, and removal. Although the technique has remained relatively similar, the manufacturing of the device has changed over time. The first generation REBOA used a 12-French sheath to deliver the balloon within the common femoral artery (CFA) requiring surgical repair. The introduction of the new second-generation ER-REBOA catheter allows for the use of a 7-French sheath and a built-in-pressure monitoring lumen; it is also guidewire-free and can be used in a fluoroscopy-free setting.1 These improvements allow for a further reduction in time to occlusion. We describe the use of a second-generation ER-REBOA in an unresponsive, hypotensive polytrauma patient.
Case
An otherwise healthy 61-year-old man presented to the emergency department at our level-I trauma centre after a 2-storey fall. The patient presented with a Glasgow Coma Scale score of 13, which deteriorated to 9, resulting in immediate intubation for airway protection. The patient was tachycardic with a heart rate of 112, and was initially normotensive with a systolic blood pressure (SBP) of 144 mm Hg, which rapidly declined to 85 mm Hg. The patient was treated according to Advanced Trauma Life Support principles. A chest radiograph showed right subcutaneous emphysema with no evidence of thoracic bleeding. As a result, a right-sided chest tube was inserted. A pelvic radiograph showed an unstable pelvis fracture with a left vertical shear pattern. Therefore, a pelvic binder was placed. As the focused abdominal for sonography in trauma (FAST) was positive, our massive transfusion protocol was initiated. The patient was immediately given 2 units of packed red blood cells (PRBCs), and an emergency trauma laparotomy was scheduled.
Despite aggressive resuscitation the patient was a transient responder with an elevated lactate of 5 mmol/L. Owing to deteriorating hemodynamics, the patient was transported to the operating room; during the preparation, we decided to obtain noncompressible hemorrhage control using REBOA at zone 3 (bifurcation of the abdominal aorta). Zone 3 was selected as the patient had severely displaced pelvic fractures, which we believed to be the primary source of hemorrhage. Zone 1 was not selected, as we wanted to avoid complications of visceral and spinal ischemia. During preparation for emergency laparotomy, the trauma team leader percutaneously accessed the left CFA by ultrasound guidance using an 18-G needle.
To ensure accessibility of the left CFA, a notch was placed in the pelvic binder. External landmarking was used to determine the placement site for balloon occlusion. A 7-French arterial inducer (AVANTI) was inserted into the left CFA and we advanced the ER-REBOA (Prytime Medical) for zone 3 aortic deployment.1
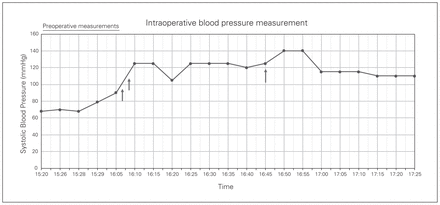
The balloon was deployed at a catheter length of 27 cm. External landmarking and catheter length were used to estimate zone 3 balloon location. As this was our first REBOA case, we also verified location using fluoroscopy before inflation with 4 mL of saline. Time from ultrasound-guided percutaneous access to balloon inflation in zone 3 was 43 seconds. There was an immediate increase in systolic blood pressure from 90 mm Hg to 125 mm Hg after balloon deployment (Fig. 1). Total balloon occlusion time was 36 minutes while surgical hemorrhage was controlled. The balloon was safely deflated by the trauma surgeon in conjunction with the anesthesia team and confirmed through fluoroscopy before removal (Fig. 2). During the trauma laparotomy, the following injuries were identified: grade III splenic laceration, grade I liver injury, blunt hollow viscus injury, and a zone 3 retroperitoneal hematoma. Surgical management consisted of splenectomy, preperitoneal pelvic packing, small bowel repair and liver packing. Intraoperative angioembolization was performed through access via the right groin to embolize bleeding branches of the iliac artery. The patient received an additional 4 units of PRBCs, 5 units of platelets and 4 units of fresh frozen plasma. Estimated blood loss was 2 L. As the patient was initially unresponsive to hemodynamic management, it was our clinical judgment that if the ER-REBOA was not placed, the patient would have required additional blood products and may have required resuscitative thoracotomy.
Intraoperative blood pressure measurement in a 61-year-old man; systolic blood pressure (mm Hg) is plotted on the y axis and time on the x axis. Preoperative measurements were those taken in the trauma bay upon arrival. Arrow 1 is time of catheter insertion (16:08:15), arrow 2 is time of balloon inflation (16:09), and arrow 3 is time of balloon deflation (16:45), at which point the catheter was also removed. The sheath was left in place until postoperative day 1, at which point it was removed in the intensive care unit.
Fluoroscopy image showing balloon deflation (white arrow) in zone 3. The inferior vena cava filter can be seen on the right (black arrow), in addition to pelvic packing material (dotted arrow).
Distal Doppler pulses were obtained for dorsalis pedis in both limbs to confirm patency of lower extremity arteries. We placed a temporary abdominal closure, and the patient was then transferred to the intensive care unit (ICU) for ongoing resuscitation.
The femoral sheath was removed in the ICU 17 hours after the procedure, and CFA imaging was performed using CT angiogram to confirm adequate limb perfusion and absence of complications. The patient’s postoperative course of 48 hours proved to be uneventful; he experienced no limb ischemia or reperfusion injuries. At 30 days postadmission, the patient was on the trauma service ward for ongoing management of an open abdomen.
Discussion
Noncompressible hemorrhage is a leading cause of preventable death in patients with traumatic injury.2 The concept of resuscitative endovascular aortic occlusion as a method of hemorrhage control is not novel.3 This technique was originally reported by Hughes during the Korean war, where the procedure was performed on 2 critically wounded soldiers.4 Although the first patients succumbed to their wounds, the potential for use as a resuscitative method was confirmed and is now showing promise to improve survival through temporary occlusion of the aorta, thereby restoring perfusion to the brain and heart.5 The technique of REBOA in trauma with off-label endovascular catheters has been used, but large arterial sheaths of up to 14-French has limited their use.5
To our knowledge, this is the first report of use of the ER-REBOA catheter in Canada since it obtained Health Canada approval in October 2017, and we have since placed a second device for a junctional hemorrhage due to a gunshot wound to the proximal superficial femoral artery. The ER-REBOA system offers a compliant aortic balloon on 5-French catheters with an integral arterial line. The device also does not require a guidewire, which allows for very rapid deployment.1 Although frequently used in the United States and Japan, REBOA has yet to be widely adopted in Canadian trauma centres.
Multidisciplinary training of the trauma program, emergency, and operating room staff allowed for rapid mobilization for REBOA placement. This training included practitioners undergoing external REBOA courses, hands-on practice in gaining CFA access with interventional radiology, didactic presentations and trauma team simulations.
Conclusion
The endovascular revolution has now placed an easily and safely deployable REBOA device in the hands of non-interventional radiologists and nonvascular surgeons. Our case report shows that with multidisciplinary training, ER-REBOA can be performed successfully in Canadian trauma centres; however, further studies are needed to fully define its role in Canadian Trauma Systems.
Acknowledgements
The authors acknowledge the generous support of the Montreal General Hospital Foundation, and the McGill University Health Centre Emergency Medicine and Adult Trauma Programs.
Footnotes
Competing interests: None declared.
Contributors: All authors contributed substantially to the conception, writing and revision of this article and approved the final version for publication.
- Accepted July 27, 2018.