Abstract
Background: Tumour budding is defined as the presence of a cluster of fewer than 5 cells along the invasive margin. It may confer a worse prognosis in colorectal cancer, but its importance in pT2N0 colorectal cancer is unknown. This study aimed to determine the prognostic value of tumour budding in pT2N0 colorectal cancer.
Methods: This was a retrospective cohort study with prospective assessment of tumour budding by 2 pathologists. We included all patients who underwent elective curative resection for pT2N0 colorectal cancer except those with hereditary colorectal cancer syndromes, inflammatory bowel disease or positive resection margins, those who received neoadjuvant or adjuvant therapy and those who died within 90 days of operation. Patients were classified as having high-grade tumour budding (≥ 10 budding foci per high-power field) or low-grade tumour budding (< 9 budding foci per high-power field). The main outcome measure was locoregional or distant recurrence.
Results: Of 85 patients, 36 had high-grade tumour budding and 49 had low-grade tumour budding. The overall recurrence rate was 11% (9/85) and median follow-up was 41.0 months (interquartile range 22.0–68.0). Interrater reliability for tumour budding assessment was excellent (κ = 0.86, 95% confidence interval [CI] 0.76–0.96). There were more recurrences in patients with high-grade tumour budding (7/36, 19.4% v. 2/49, 4.1%; p = 0.020). On multivariate analysis, after we adjusted for confounders, the presence of high-grade tumour budding was independently associated with recurrence (hazard ratio 5.11, 95% CI 1.01–25.9).
Conclusion: Tumour budding was independently associated with increased recurrence after pT2N0 colorectal cancer resection. It offers additional prognostic information that may affect treatment strategy.
The prognosis and treatment of colorectal cancer depends largely on the stage of the disease as classified by the TNM staging system. Even though the rate of overall survival for stage I colorectal cancer is high,1 some tumours behave more aggressively and patients have a worse prognosis than would be predicted by TNM stage alone. There are well-established prognostic factors, such as lymphovascular and perineural invasion,2–4 that help clinicians identify such tumours. There is an increasing body of evidence to support the use of tumour budding in identifying these high-risk tumours as well.
First described by Jass and colleagues5 in the characterization of the invasive margin, tumour budding was demonstrated to be a prognostic marker in rectal cancer. Although there are several different definitions,6–8 in essence, tumour budding is defined as the presence of single or small clusters of malignant cells (< 5 cells) scattered in the stroma at the invasive margin. It is thought that the buds represent tumour that has gained vascular and lymphatic invasive ability. Histologic studies have shown that tumour buds are located near or within areas of lymphovascular invasion.6,9 Furthermore, tumour budding can predict isolated tumour cells in lymph nodes of node-negative colorectal cancers.10 Supporting this theory is the strong association between tumour budding and lymph node metastases in tumours confined to the submucosa (pT1).11–13
Among patients with stage II colorectal cancer, tumour budding has been associated with increased disease progression and cancer-related death.14–17 However, few studies have investigated the prognostic value of tumour budding in early colorectal cancer. Two previous studies have reported an association between tumour budding and increased metastases and mortality in stage I and II colorectal cancer,18,19 but no study has specifically investigated the prognostic value of tumour budding in patients with pT2N0 colorectal cancer. We hypothesized that a high number of tumour buds at the invasive margin will predict worse outcomes in patients with early-stage colorectal cancer. The objective of this study was to determine the impact of tumour budding on locoregional and distant recurrence in patients with pT2N0 colorectal cancer.
Methods
After we obtained institutional ethics review board approval, we identified all patients who underwent elective curative radical resection (R0) for pT2N0 (as per the TNM staging system in the 7th edition of the American Joint Committee on Cancer Staging Manual) colorectal cancer between 2000 and 2011 at a single university-affiliated institution through the operating room and pathology databases. Patients with hereditary colorectal cancer syndromes, inflammatory bowel disease or positive resection margins, patients who received neoadjuvant or adjuvant therapy and patients who died within 90 days of operation were excluded. Patient characteristics, operative variables and long-term outcomes were obtained by retrospective chart review. All patients underwent history and physical examination and had serum carcinoembryonic antigen levels drawn every 3 months for the first year, followed by a surveillance colonoscopy at years 1, 3 and 5 after resection. Imaging was performed at the surgeon’s discretion on the basis of symptoms and tumour markers. The primary outcome was tumour recurrence, defined as either locoregional recurrence or distant metastasis. All anastomotic recurrences were detected on surveillance endoscopy and were biopsy proven; locoregional extraluminal recurrences, as well as distant recurrences, were either biopsy proven or diagnosed on the basis of imaging at a multidisciplinary tumour board meeting.
Eligible surgical pathology specimens from the index operation were prospectively re-reviewed by 2 independent pathologists for histopathologic criteria. All specimens were fixated in formalin and stained with hematoxylin and eosin. Tumour budding was assessed according to Ueno and colleagues, who defined budding foci as isolated cancer cells derived from clusters of fewer than 5 cancer cells, situated in the stroma of the actively invasive margin.7 Figure 1 in a report by Zlobec and colleagues20 illustrates tumour budding at the invasive margin. The budding number was determined by counting the foci within 1 high-power field (× 25 magnification) in an area chosen by the pathologist as maximal budding, known as the hotspot method. Tumour budding was then classified using a 3-tier system as low budding (0–4 budding foci per high-power field), intermediate budding (5–9 budding foci per high-power field) or high budding (≥ 10 budding foci per high-power field). Both the hotspot method and the 3-tier system were recommended by the International Tumor Budding Consensus Conference in 2016.21 For this analysis, we divided patients into 2 cohorts, grouping low and intermediate tumour budding together: the low-grade tumour budding cohort had fewer than 9 budding foci per high-power field, and the high-grade tumour budding cohort had 10 or more budding foci per high-power field. Only high-grade tumour budding has been associated with poor oncologic outcomes and this is the prognostic subgroup of interest; this is why we grouped low and intermediate tumour budding together.21,22 Patient, operative and histopathologic characteristics were compared between the 2 groups.
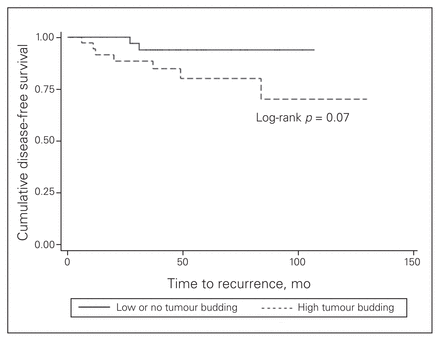
Kaplan–Meier disease-free survival curve for patients with low or no tumour budding compared with that for patients with high tumour budding.
Descriptive and summary statistics were calculated, as appropriate. The Cohen kappa statistic (κ) was calculated to assess the interrater reliability of tumour budding assessment by group (low-grade v. high-grade tumour budding). Univariate analyses were performed using the Fisher exact or χ2 tests for categorical variables and the Student t or Mann–Whitney tests for continuous variables. Multiple Cox proportional hazards regression was performed to investigate the association between recurrence and high-grade tumour budding. Factors that were significant on univariate analysis were included in the regression model. Statistical significance was set at p = 0.05. All statistical analyses were performed using Stata 12 (StataCorp).
Results
A total of 108 patients were identified as having pT2N0 colorectal cancer on final pathology. Twenty-three patients were excluded: 9 patients had received neoadjuvant therapy (ypT2), 2 received adjuvant chemotherapy (patient preference given young age and high-risk features), 5 had inflammatory bowel disease with chronic colitis, 3 had familial colorectal cancer syndromes, 2 had positive resection margins and 2 died in hospital within 30 days of their operation. Therefore, a total of 85 patients met the inclusion criteria, of whom 36 (42.4%) had high-grade tumour budding and 49 (57.6%) had low-grade tumour budding. Median follow-up for all patients was 41.0 months (interquartile range [IQR] 22.0–68.0). The characteristics of these patients are reported in Table 1. Of note, patients with high-grade tumour budding were more likely to be younger (65.7 yr v. 70.5 yr, p = 0.047) and to have rectal tumours (66.7% v. 36.7%, p = 0.006). There were no other differences in patient, operative, and histopathologic characteristics, including tumour differentiation and lymphovascular and perineural invasion. The interrater reliability of tumour budding assessment was excellent (κ = 0.86, 95% confidence interval [CI] 0.76–0.96).
Characteristics of patients with low-grade or high-grade tumour budding
The overall recurrence rate over the study period was 11% (9/85). There was a higher incidence of tumour recurrence in patients with high-grade tumour budding than in those with low-grade tumour budding (7/36, 19.4% v. 2/49, 4.1%; p = 0.020) (Table 2). Of the 7 high-grade tumour budding recurrences, 5 were in patients with rectal primaries (1 with lung metastases, 1 with lung metastases and anastomotic recurrence, 3 with locoregional pelvic recurrences) and 2 were in patients with colon primaries (1 with liver metastases, 1 with intraluminal anastomotic recurrence). Of the 2 low-grade tumour budding recurrences, 1 was in a patient with a rectal primary and the other was in a patient with a colon primary (both locoregional recurrences). There were 4 cases of anastomotic leak: 3 were in patients with high-grade tumour budding and 1 was in a patient with low-grade tumour budding (8.3% v. 2.0%, p = 0.18). All such cases occurred with proctectomies, and 1 of these patients developed a locoregional recurrence (this patient had high-grade tumour budding). Median time to recurrence in patients with high-grade tumour budding was 20.5 months (IQR 11.5–49.5 mo) whereas it was 29.0 months (IQR 27.0–31.0 mo) in patients with low-grade tumour budding. Unadjusted survival curves demonstrated a trend toward improved disease-free survival for patients with low-grade tumour budding compared with high-grade tumour budding, but this trend did not reach statistical significance (log-rank p = 0.07) (Fig. 1).
Postoperative outcomes and follow-up of patients with low-grade or high-grade tumour budding
After accounting for age and tumour location (colon v. rectum) using a multiple Cox proportional hazards regression model, we found that the presence of high-grade tumour budding was independently associated with recurrence (hazard ratio [HR] 5.11, 95% CI 1.01–25.90) (Table 3). Age was also associated with recurrence (HR 1.11, 95% CI 1.02–1.21) but tumour location was not a significant predictor (rectal tumour: HR 2.23, 95% CI 0.54–9.24).
Multivariate Cox proportional hazards analysis for tumour recurrence
Discussion
There may be additional factors other than the TNM disease stage that will predict a worse outcome, even in early-stage colorectal cancer. In this study, we have determined that high-grade tumour budding is associated with a higher risk of recurrence in patients with pT2N0 colorectal cancer than in similar patients with low-grade tumour budding.
Only 2 previous studies have assessed the prognosis of tumour budding in patients with pT2N0 colorectal cancer who underwent surgical resection. Losi and colleagues18 compared 22 patients who died of stage I colorectal cancer and matched them by sex, age, location of tumour, type of resection and tumour depth (pT stage) with patients with stage I disease who were still alive, and they found that there was a higher incidence of tumour budding in patients who died than in those who were still alive at 5-year follow-up. Prall and colleagues19 reported that high tumour budding was associated with increased metastases on survival analysis in patients with stage I/II colorectal cancer, although the authors used a higher cut-off for the number of tumour buds than either Losi and colleagues18 did or we did in the present study. However, both of these studies are limited by a relatively small number of patients with pT2N0 or by the inclusion of patients who received neoadjuvant or adjuvant therapy.
The present study is the largest to evaluate the impact of tumour budding in pT2N0 colorectal cancer and reports a higher recurrence rate (19.4%) than would be predicted on the basis of staging alone. The association between high-grade tumour budding and tumour recurrence remained on multiple Cox proportional hazards regression after we accounted for possible confounders, such as tumour location and age. These findings correlate well with what is already known regarding tumour budding in colorectal cancer, where the negative prognostic impact of high-grade budding has been demonstrated in both early and advanced disease. In a large series of patients with stage III disease, tumour budding was independently associated with worse disease-free survival, and when combined with the N stage (N1 v. N2), a better prognostic stratification was obtained for 5-year disease-free survival when compared with nodal staging alone.23 In node-negative colorectal cancer, tumour budding has been more widely studied and may have larger implications for clinical decision-making. Several studies have reported worse outcomes in patients with stage II disease (pT3–4N0) in the presence of high-grade budding, citing survival rates in such patients equivalent to those of patients with stage III disease.14–17,24
The reason why early-stage cancers with high-grade budding behave more aggressively than expected is not clear. Tumour budding has been shown to be predictive of isolated tumour cells in lymph nodes of node-negative cancers, and it is possible that tumour budding is associated with a higher rate of occult metastases.10 These patients are consequently downstaged and inadequately treated. In our cohort of patients with pT2N0 disease and high-grade tumour budding, a similar underestimation may have occurred, which would explain the higher rate of distant metastases in patients with high-grade tumour budding. We believe that a referral to medical oncology to consider adjuvant treatment is warranted in this subgroup of patients, even in stage I (pT2N0) disease. We also found that tumour budding was associated with locoregional pelvic recurrence in rectal cancer, which has been previously described.25,26 Although our rate of anastomotic recurrence is higher than what would be expected, studies have demonstrated an association between intramural tumour spread and tumour budding, which could result in residual tumour cells at the anastomotic line and explain our findings.27 Perhaps patients with high-grade tumour budding should undergo more frequent surveillance of the anastomosis. Future studies are needed to further investigate the association between tumour budding and recurrence, to better understand the pathophysiology of this adverse marker.
In addition to the prognostic value of tumour budding, we have also reported a very high interrater reliability (κ = 0.86) in the assessment of high-grade versus low-grade tumour budding, which is similar to that reported by other single-centre studies.7,19 Even though reliability decreases slightly when multiple pathologists and institutions are involved, reported interrater reliability remains reasonable. 17,28 Furthermore, our 42% detection rate of high-grade tumour budding is congruent with what is generally described in the literature, ranging around 29%–43%.7,24,26 We assessed tumour budding with the hotspot method as per Ueno and colleagues,7 which was endorsed by the International Tumor Budding Consensus Conference in 2016; however, other methods to count tumour buds have been described.21 We also grouped low and intermediate budding together (both < 10 buds), a decision that is supported by the literature. In studies that use a binary cut-off to define the presence or absence of tumour budding, a cut-off of 10 or more budding foci is most commonly used to signify its presence.22 Furthermore, in the largest systematic review and meta-analysis to date, whenever a 3-tier system was used in individual studies, the authors grouped together the mild and moderate budding cases and compared outcomes with those of the highest budding cases.22 In a cohort study of 159 patients, high-grade tumour budding had a higher hazard of cancer-related death (HR 3.14, 95% CI 1.52–6.49) and disease recurrence (HR 3.24, 95% CI 1.43–7.34) than low-grade budding, while intermediate budding demonstrated no increased risk (HR 1.88, 95% CI 0.90–3.92, and HR 1.83, 95% CI 0.81–4.12, for cancer-related death and disease recurrence, respectively).26 Tumour budding can also be assessed using immunohistochemistry as opposed to hematoxylin and eosin staining, but meta-analyses suggest that the prognostic capabilities of tumour budding do not differ by technique.22,29 Unlike previous studies, we did not identify an association between high-grade tumour budding and other histopathologic markers of adverse prognosis, such as lymphovascular invasion, perineural invasion and tumour differentiation. Although this result is probably due to our small sample size, it lends further support to tumour budding as an independent marker of poor prognosis.
The strength of this study lies in the fact that 2 pathologists blinded to the main outcome independently reviewed all pathologic specimens, preventing observer bias. To our knowledge, this is 1 of very few North American studies that have investigated the prognostic value of tumour budding in colorectal cancer. Previous studies have originated mainly from Japan; their results may not be completely generalizable to a North American population because of differences in social, cultural and genetic factors. We also excluded patients with confounding factors, such as inflammatory bowel disease and hereditary cancer, to preserve the validity of our findings, but at the cost of generalizability to a wider patient population. Lastly, this is 1 of the very few studies looking exclusively at T2N0 colorectal cancer, which can have a very heterogeneous behaviour. A better understanding of the important prognostic factors in early-stage colorectal cancer, which typically does not warrant neoadjuvant or adjuvant treatment, can greatly improve the management of this cohort of patients.
Limitations
Our findings must be interpreted in view of several limitations. First, this was a retrospective analysis with a small sample size, which limited both the number of covariates that we could include in the multiple Cox proportional hazards model and the width of the confidence intervals surrounding the estimates. This was also a single-centre analysis from a tertiary-care institution with specialized pathologists and colorectal surgeons, which may decrease the generalizability of the results to institutions that may not have these specialists available. We also were not able to obtain data on the molecular subtyping of the cancers, such as the presence of KRAS or BRAF mutations, or the presence of microsatellite instability. This information would have been useful to better understand the makeup of high-risk early-stage cancers, as well as their association with tumour budding. Finally, we did not assess cancer-specific or overall mortality, as this analysis would have required a much higher sample size to be adequately powered.
Conclusion
We have provided preliminary evidence for the prognostic value of tumour budding in early-stage pT2N0 colorectal cancer. Future research should focus on validating these findings and evaluating whether additional adjuvant therapy or more intensive surveillance is required in patients with tumour budding. Tumour budding represents an easily measurable prognostic marker that has an increasing body of evidence to support its inclusion as part of the decision-making process in early colorectal cancer.
Acknowledgements
The authors thank Elektra McDermott for her editorial assistance and preparation of the manuscript.
Footnotes
Presented at the American Society of Colon and Rectal Surgeons 2013 Annual Scientific Meeting, Apr. 27 – May 1, 2013, Phoenix, Ariz.
Competing interests: None declared.
Contributors: All authors designed the study and acquired and analyzed the data. R. Garfinkle wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper. All authors agreed to be accountable for all aspects of the work.
Funding: R. Garfinkle was supported by a Canadian Institutes of Health Research Summer Research Bursary from the McGill University Faculty of Medicine. L. Lee was supported by the Quebec Health Science Research Fund (FRQ-S). The funders had no role in the conduct of this study.
- Accepted January 3, 2019.