Abstract
Background: Literature on the survival rates and function of hinged total knee replacement (HTKR) prostheses is scarce, and to our knowledge there is not yet any published literature on the Legion HK Hinge Knee Replacement prosthesis (Smith & Nephew) with guided-motion articulation. The objective of this study was to establish the early survival rate of this modern HTKR at a single institution and to investigate postoperative patient function and satisfaction.
Methods: This retrospective study included patients who received the Legion HTKR prosthesis with guided-motion inserts as a primary or revision implant between October 11 and March 2016 at a tertiary care centre in Manitoba, Canada. Preoperative and postoperative functional scores on the 12-item Oxford Knee Score and postoperative patient satisfaction were assessed.
Results: Thirty-nine HTKR implantations (38 patients) were included in this study: 12 primary cases and 27 revision cases. Three revision surgeries and 4 perioperative complications were noted at a mean follow-up of 29.1 months. The 2-year survivorship of the HTKR system was 90.7%. Postoperative functional scores improved significantly and the majority of patients were satisfied or very satisfied at all follow-up time points.
Conclusion: The early survival rate of a modern guided-motion HTKR prosthesis is similar to the survival rates of other hinged knee prostheses published in the literature. The prosthesis demonstrated substantial postoperative functional improvement when used in the setting of complex primary or revision total knee replacement.
Ahinged total knee replacement (TKR) prosthesis is a joint replacement device in which the medial and lateral collateral ligaments are supplanted by a hinge mechanism between the femoral and tibial components. The purpose of these specialized TKR devices is to restore knee function to patients with substantial knee soft-tissue defects, including those with ligamentous instability and those who have had large amounts of tissue resected during tumour removal. Hinged TKR (HTKR) designs coupled with additional mechanical support may also be required in cases of excessive bone loss.
Early HTKR designs used a fixed hinge with fully constrained movement of the knee joint that only allowed flexion and extension motions, creating a high amount of shear and rotational stress on the prosthesis. These increased forces were transmitted to the bone–prosthesis interface and are theorized to have increased the likelihood of early failure because of aseptic loosening.1 Newer systems rotationally decouple the prostheses to reduce this risk. In general, HTKR designs are used in cases of severe collateral ligament attenuation or absence when a constrained condylar design will not provide adequate stabilization. Constrained condylar designs also have severe rotational constraint, leading to concerns similar to those noted with early hinged knee designs and premature prosthesis failure.2 Modern HTKR devices are now being used not only for salvage procedures but also for more complex primary and revision arthroplasty cases.3–5
With the increasing demand for primary and revision TKR in North America, along with the increasing applicability of HTKR devices, clinical study of these prostheses is becoming more important.6 Information on survivorship and functional outcomes of modern HTKR devices is limited as these prostheses have traditionally been used in highly complex or salvage cases. The studies published to date on HTKR have relatively small sample sizes and the results tend to be somewhat inconsistent among centres.7,8
The Legion HK Hinge Knee Replacement device (Smith & Nephew) is a modern HTKR prothesis designed to more closely match the original knee anatomy and to more fully restore normal knee function. The system incorporates a rotating hinge mechanism alongside a medial pivot with a lateral rollback design to replicate more natural kinematics and allow for screw home rotation of the knee upon extension through the use of “guided motion” inserts.9 Further, the design is such that a reported 96% of the joint load is applied to the condylar surfaces, thereby removing stresses from the hinge mechanism and improving the wear characteristics of the device.10 To our knowledge, no published clinical data are currently available on this system.
The purpose of this study is first to establish the early survival rates of the Legion HTKR device with guided-motion inserts and to compare these with published data on the survival rates of other hinge prostheses. Second, this study aims to evaluate improvement in patient function following implantation of the HTKR device.
Methods
A retrospective database review was initiated at the Concordia Hip & Knee Institute to study outcomes of HTKR. Ethical approval for this study was granted by the University of Manitoba Bannatyne Research Ethics Board and Concordia General Hospital Research Ethics Board. Patient consent was obtained through the institution’s research database consenting process before surgery. The study included consenting patients who received the Legion HTKR prosthesis with guided-motion inserts as a primary or revision implant between October 2011 and March 2016. Indications for use of the HTKR prosthesis in both the primary and revision setting were incompetence of the medial collateral ligament and/or severe uncorrectable collateral ligament imbalance. All procedures were performed by fellowship-trained, high-volume arthroplasty surgeons.
The standard surgical technique for all cases included complete removal of failed components or adequate resection of bone in the case of primary hinge use. Proximal tibial and distal femoral cleanup cuts were referenced off of reamers. The tibia was addressed first, followed by the femur. Augments and/or offset couplers were used where appropriate to deal with bone loss and optimize joint line height and prosthesis positioning. The implants were cemented in the metaphyseal region. In most cases, diaphyseal engaging press-fit splined titanium stems were used. Cemented stems were used in selected cases because of bone morphology issues.
Routine follow-up occurred at 6 weeks, 6 months, 1 year, 2 years and every 2 years thereafter. These appointments served as observation points in the survival analysis to confirm implant survival. Complications and revision events following HTKR implantation were identified through chart review. Revision surgery where 1 or more of the implants was altered was labelled as a failure event and the date of revision acted as the observation point for the survival curve. Prothesis survivorship rates were determined using a Kaplan–Meier plot.
Available data for patient-reported outcome measures were compiled, including the 12-item Oxford Knee Score (OKS)11 and a 5-point scale assessing patient satisfaction. Data for patient-reported outcome measures were included only for patients with a minimum of 6 months of follow-up. Change in OKS score from the preoperative to postoperative time points was evaluated using a paired Student t test. Patient satisfaction was simplified to “satisfied” (satisfied and very satisfied), neutral and “unsatisfied” (unsatisfied and very unsatisfied) categories to facilitate statistical analyses. The Fisher exact test was used to evaluate differences in patient satisfaction between follow-up periods. Pearson correlation analyses evaluated relationships between patient demographic data, satisfaction and OKS scores. SAS version 9.3 (SAS Institute Inc.) was used for all statistical analyses, and significance was set at p < 0.05. Zero-numerator 95% confidence intervals (CIs) were calculated using this formula:12 95% CI equals 1 minus the nth root of 0.05.
Results
Patients
A total of 38 patients (39 implantations) were identified as having received an HTKR prosthesis between October 2011 and March 2016. Of the 39 procedures, 27 were revision procedures and 12 were primary procedures. Owing to loss to follow-up within the group, 34 of the 39 cases contributed to survival data at 1 year and beyond, and 31 contributed to survival data at 2 years and beyond. The majority of patients were women (71.8%) with a group mean age of 69 years (range 51–92 yr). Among the 27 revision cases, the indications for revision surgery were instability (17), infection (4), aseptic loosening (4) and combined instability and aseptic loosening (2). In all cases involving primary procedures, a hinged component was selected to manage medial collateral ligament incompetence secondary to the following conditions: degenerative arthritis with severe deformity (6 knees), inflammatory arthritis (2 knees), severe degenerative arthritis with postpolio syndrome (2 knees), Charcot arthropathy (1 knee) and postinfectious arthritis (1 knee).
Complications
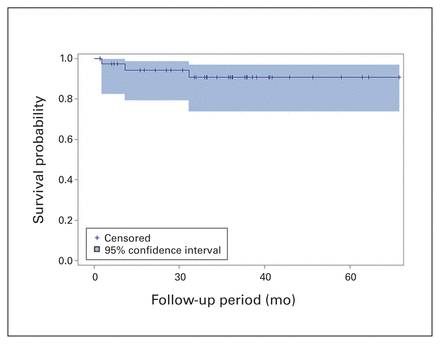
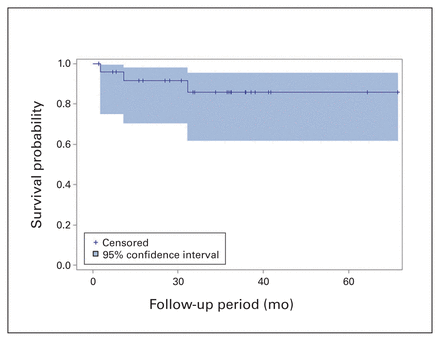
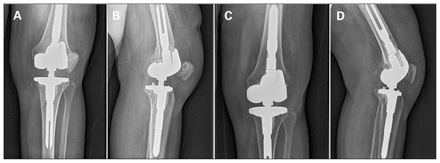
Survival rates for all HTKR protheses in this study at 1 and 2 years were 94.4% (95% CI 79.2%–98.6%) at 1 year and 90.7% (95% CI 73.6%–97.0%) at 2 years (Fig. 1). Three patients in this cohort experienced failure of their HTKR prosthesis; all of these patients had received the prosthesis in a revision procedure. Accordingly, survival for the revision HTKR protheses was 91.6% (95% CI 70.5%–97.9%) and 85.9% (95% CI 61.8%–95.3%) at 1 and 2 years, respectively (Fig. 2), whereas survival for the primary HTKR protheses was 100% (95% CI 76.1%–100%) at 1 year and 100% (95% CI 71.7%–100%) at 2 years. The first HTKR failure resulted from implantation of an undersized femoral component because the appropriate size was not available at the time of surgery (Fig. 3A, Fig. 3B). The patient’s joint line was subsequently elevated. Combined with an inadequate lateral trochlear ridge height, this resulted in recurrent patellar dislocations and functional deficit. Revision was performed 7 months after the index procedure (Fig. 3C, Fig. 3D). The second failure was due to a periprosthetic joint infection 23 months after surgery, probably as a result of a massive soft tissue defect to the anterior leg from an acute traumatic event. The third failure was at 22 months after surgery following a mechanical fall. This caused loosening of the hinge locking bolt, and the patient experienced patellar subluxation and end extension impingement.
Kaplan–Meier survival curve for all hinged total knee replacement prostheses up to the maximum follow-up of 71 months.
Kaplan–Meier survival curve for revision hinged total knee replacement protheses up to the maximum follow-up of 71 months.
(A, B) Patient with an undersized femoral component for the hinged total knee replacement prosthesis and subluxating patella. (C, D) Revised femoral component for the hinged total knee replacement prosthesis.
Complications occurred in 7 patients (18%), 3 of whom were considered to have had failed implantations because they underwent revision surgery as previously described. The patient who underwent revision surgery for the undersized femoral component also underwent a medial capsule repair 2 months after the index procedure. No components were revised during this procedure. Two additional patients sustained disruption of their extensor mechanism and required further surgery for repair. One patient experienced a periprosthetic fracture of the proximal tibia after implantation. This was treated by open reduction and internal fixation with a proximal tibial locking plate 2 weeks after the index procedure.
Patient-reported outcome measures
Data for patient-reported outcome measures at a minimum of 6 months follow-up were compiled for 30 patients. The data for the 3 patients who experienced failures were excluded, and 5 patients did not return to the clinic for follow-up. The mean follow-up duration for these 30 patients was 29.1 months (range 1.4–62.9 mo). Group mean OKS scores improved significantly from the preoperative time point to the 6-month, 1-year and 2-year postoperative time points (Table 1). The majority of patients were either satisfied or very satisfied with their HTKR prosthesis at 6 months (63%), 1 year (50%) and 2 years (52%) (Table 2). No significant difference in satisfaction was detected between 6 months and 1 year or 2 years (p = 0.5 and 0.8, respectively).
Scores on the Oxford-12 Knee Score instrument for patients who received a guided-motion hinged total knee replacement prothesis
Patient-reported satisfaction following implantation of a guided-motion hinged total knee replacement prothesis
Preoperative OKS scores were strongly correlated with 6-month, 1-year and 2-year postoperative scores (r = 0.82, p < 0.001; r = 0.65, p = 0.012; r = 0.62, p = 0.024, respectively), calculated using Pearson correlation analysis, indicating that patients with higher preoperative function typically regained higher function following HTKR implantation.13 Postoperative OKS scores were correlated with patient satisfaction at 6 months, 1 year and 2 years postoperatively (r = 0.77, p < 0.001; r = 0.68, p = 0.002; r = 0.57, p = 0.028, respectively), indicating that patients with good function following surgery were generally satisfied with their joint replacement. There were no differences in OKS scores between patients who underwent the procedure in a primary versus a revision setting at any point in follow-up (p > 0.39). In addition, there was no correlation between OKS scores and age, body mass index and sex. Mean flexion arc was 98° (standard deviation [SD] 22°) preoperatively, with means of 109° (SD 15°), 107° (SD 13°) and 110° (SD 16°) at 6, 12 and 24 months postoperatively, respectively.
Discussion
The HTKR system with guided-motion inserts used in this study is a modern hinged knee replacement option for patients with significant soft tissue dysfunction at the knee. The overall revision rate in this study was 7.7% (3 of 39 implantations) with a 2-year survival rate (for nonrevision implants) of 90.7% in the 31 patients with more than 2 years of data. These results are comparable with the findings of other studies of HTKR protheses. One recent study looked at mixed primary and revision hinge use and reported a revision rate of 11.5% at a mean of 46 months.14 Bistolfi and colleagues reported 93% implant survival at 2 years and 79% at 5 years.15 Guenoun and colleagues reported 89% implant survival at a mean of 36 months.16 Similarly, the complication rate reported in this study of 18% (7 of 39, including 3 failures) is within the range of rates reported in the literature of 9.2% at 2 years,8 28% at 36 months and 36% at 5 years.15
No revisions of primary HTKR cases were performed during the study period. This high success rate aligns well with survival rates reported in the literature for use of hinged TKRs in primary procedures: 96.8% survival at 5 years3 and 95% survival at 6 years.17 Although there has been no statistical difference in the failure rates of hinged TKR done in primary versus revision settings, there appears to be a trend toward increased failure among patients who undergo revision procedures.18 This is probably secondary to the generally increased complexity of the revision group as these patients tend to have reduced bone stock and compromised soft tissues that have resulted in previous failures of prostheses.
This study demonstrated a significant improvement in patient-reported function after surgery. There was a difference of 14.0 points on the OKS scale at 1 year after surgery. In keeping with other studies, we found that a patient’s preoperative OKS score strongly predicted their postoperative score. Improvement in function was associated with postoperative patient satisfaction. The majority of our patients were either satisfied or very satisfied with their HTKR prosthesis at all follow-up time points. We were unable to determine if the guided motion of the insert design had a discernible impact on the clinical outcomes achieved.
Of the 3 revisions that occurred in our study sample, 2 involved an element of patellar instability. The guidedmotion design of the protheses we used is intended to better replicate the internal rotation of the tibia as the knee flexes. In both cases, there were mechanical alterations that predisposed the knee to patellar instability (undersized femoral component and component impingement/altered alignment, respectively). Unlike most modern hinged knee designs with almost unlimited rotation, the studied design is linked and has a maximum of 18° of rotation. Although highly rotationally constrained devices are at increased risk of failure, there is no consensus on the degree of rotational freedom required for a hinged design.1 It is unclear if the rotational freedom of the hinged design could have contributed to the failure mechanism in these 2 cases.
Limitations
There are a number of limitations to this study. The relatively short duration of follow-up prevented us from commenting on prothesis performance beyond the initial 2 years after implantation. It has, however, been documented that infection, aseptic loosening and mechanical failure occur most frequently within 2 years after implantation. 19–21 The second limitation is the retrospective nature of the study. Although the data were collected prospectively as part of a larger clinical research database, there is a greater potential for missing data and loss to follow-up in this type of study than in a dedicated prospective trial. This may have contributed to underreporting of complications, resulting in the study having a relatively lower complication rate than reported by other authors. Additionally, some patients declined to provide responses to portions of the patient-reported outcome measures, such as questions relating to satisfaction. This could have led to overestimation of the satisfaction data if less satisfied patients were more likely to withhold their responses. Moreover, the data set for this study is relatively small at 38 patients (39 cases). The limitations of the small sample size are exacerbated in the subgroup analysis of revision and primary surgeries. Given the relative infrequency with which hinged protheses are used and the relatively short time that this product has been available, these issues were unavoidable. Nonetheless, this sample is similar in size to the samples in several other studies assessing HTKR.14,22,23 In addition, there is a risk that 1 or more of the 5 patients lost to follow-up could have undergone a revision at another centre. Although this would have resulted in an overestimate of the survival rate, the scenario is unlikely given the relative geographic isolation of the tertiary care arthroplasty centre where the study was completed. The selection of revision of 1 or more protheses as the definition of failure of survival is in keeping with other reported studies.3 It does, however, potentially lead to underreporting of failure owing to patients unable or unwilling to undergo further surgery in the face of a clinical failure. Although we are not aware of any clinical failures for which the patients have not gone on to surgery, this methodology has the potential to create bias in the data. Because some patient-reported outcome data were not available, we have reported improvement in these scores as the difference between the group mean at preoperative and postoperative time points instead of calculating improvement for each patient and taking the mean of these values. Comparison of group means is not ideal, but it provides a reasonable measure for overall group function, which in our study demonstrates improvement from the preoperative state. The missing patient-reported outcome data limit the extrapolation that can be done for the clinical outcome measures reported.
Conclusion
To our knowledge, this is the first clinical study reporting the survival and clinical outcomes of a modern HTKR system with guided-motion articulation. The system appears to be a suitable alternative for ligamentous, soft tissue and bony deficiencies in both primary and revision surgery. This study shows no immediate concerns with its use; it was not possible to assess longer term performance because this system has only recently become available. The findings for the HTKR system studied in this report were consistent with published survivorship and functional improvement rates for other HTKR designs. It is unclear what role the guidedmotion design of the bearing surface plays in the outcome, and further study is recommended.
Footnotes
Funding: This study was supported by Smith & Nephew. Smith & Nephew did not provide any input into the study design; the collection, analysis or interpretation of the data; the preparation of the manuscript; or the decision to submit the article for publication.
Competing interests: This research was financially supported by Smith & Nephew through institutional funding. T. Turgeon has acted as an unpaid consultant for Smith & Nephew, outside the submitted work. Institutional research funding was also received from DePuy Synthes, Zimmer Biomet and Hip Innovation Technology, outside the submitted work. No other competing interests were declared.
Contributors: Both authors designed the study. T. Turgeon acquired the data, which both authors analyzed. Both authors wrote the article, which T. Turgeon critically reviewed. Both authors approved the article for publication.
- Accepted September 17, 2019.