Abstract
Objective: Evaluation of the safety and potential cost savings of a computerized, laboratory-based program to manage inpatient warfarin thromboprophylaxis after major joint arthroplasty.
Design: A consecutive-case study of adults.
Setting: A tertiary care orthopedic institution.
Patients: Patients requiring joint arthroplasty who had no recent episodes of thromboembolic disease, no mechanical heart valve, atrial fibrillation, severe liver disease or baseline international normalized ratio [INR] greater than 1.3 admitted over a 54-month period (July 1994–December 1998). All patients received a standard regimen of warfarin beginning on the evening after the operation. Four hundred and thirty randomly selected patients managed by the program were followed up by telephone survey 3 months after discharge. Patients exhibiting erratic responses to warfarin were withdrawn from the program and managed individually thereafter.
Intervention: Major joint arthroplasty with warfarin therapy administered through the computerized program.
Main outcome measures: Test results maintained within the desired therapeutic range (INR 2.0–3.0), clinically severe bleeding episodes, readmission rates, clinically symptomatic and venographically proven episodes of venous thrombosis or pulmonary embolism.
Results: Over the study period 5629 patients underwent joint arthroplasty; 5372 patients were considered for the program; 332 patients were ineligible and were managed individually; 311 entered patients did not complete the program. This left 4729 patients who completed the program. In 2932 (62%) patients test results were maintained in the desired therapeutic range. The major bleeding rate was less than 0.5%, the readmission rate was 3.8%, the deep venous thrombosis rate was 3.7% and the pulmonary embolism rate was 0.2% with no thromboembolic related deaths in the small sample cohort.
Conclusions: The majority of patients requiring warfarin thromboprophylaxis can be safely and effectively managed by this laboratory-based computerized program while in hospital. Significant potential cost savings in nursing time could be achieved.
There is general agreement in North America that hip and knee joint arthroplasties place the patient at high risk for the development of venous thromboembolism and that some form of chemical or mechanical prophylaxis is required.1,2 Despite the growing use of low-molecular-weight heparin, warfarin sodium, a coumarin derivative, remains widely utilized in major orthopedic surgery.3,4 The use of warfarin is associated with a number of outstanding clinical and laboratory issues including the need for regular laboratory monitoring.5,6
The Orthopaedic & Arthritic Institute of Sunnybrook and Women’s College Health Sciences Centre in Toronto performs approximately 1200 hip and knee arthroplasties annually and uses a standardized warfarin thromboprophylaxis protocol during the patient’s in-hospital stay, beginning in the evening after the operation. Therapy is monitored by means of serial prothrombin times, expressed as the international normalized ratio (INR) (defined as the observed prothrombin time ratio raised to the power of the International Sensitivity Index of the instrument-specific thromboplastin used in the laboratory).7 A laboratory-based computerized program using an internally developed algorithm and software was introduced in July 1994. The program provides a daily dose recommendation and a cumulative graphic representation of the patient’s results and warfarin dose record for the ward staff. The therapeutic range (2.0–3.0) follows published guidelines.4
The objective of this study was to evaluate the safety and effectiveness of this program as well as selected cost-related issues. We considered the proportion of test results in the therapeutic range, the number of clinically severe bleeding episodes, hospital readmission rates, clinically symptomatic and venographically proven episodes of venous thrombosis and pulmonary embolism. Financial measures considered were pharmacy and comparative nursing care costs associated with the program. Patients who underwent similar procedures in the 18-month period before the program was introduced served as historical controls. These patients received the identical loading doses of warfarin and were individually managed by staff surgeons or internists. No other changes in patient mix or surgical practice were made, although the length of stay in the study period declined relative to the historical control period from 10 days to 8.7 days.
Methods
The program is based on an oral anticoagulant management algorithm developed by one of the authors (W.C.) and used in a traditional manual format by medical and nursing staff at the hospital. The program incorporates minimal patient data apart from the age of the patient and estimated weekly alcohol intake. The program is written in BASIC language and uses a laboratory based personal computer networked to a HUMMINGBIRD laboratory information system version 9.14 (Laboratory Consulting Inc., Madison, Wisc.). The program has built-in safety features including a maximum recommended dose and is capable of storing 50 000 test results online. Coagulation testing is performed on an ACL 1 000 instrument (Instrumentation Laboratory, Lexington, Mass.), with use of a sensitive Instrumentation Laboratory reagent (PT Fibrinogen HS Plus, sensitivity index of 1.12) for the prothrombin time. Reporting of all prothrombin time results is the INR with a normal reference range of 0.9 to 1.1. Control of anticoagulant therapy was assessed by the proportion of test results in the recommended therapeutic range (2.0–3.0) over the total number of patient-days in hospital.
All consecutive patients scheduled for major joint arthroplasty surgery (primary and revision hip and knee procedures) over the period from July 1994 to December 1998 were considered for the study. Patients who met the standardized entry criteria — no recent episodes of thromboembolic disease and no mechanical heart valve, atrial fibrillation, severe liver disease or baseline INR greater than 1.3 — were entered on the computer program.
The historical control group comprised 279 patients from whom 2889 test results were obtained for comparison with the 4729 patients who completed the program.
Program protocol
Patients receive a standard dose of 5 or 10 mg warfarin on the evening after the procedure, based on their age and history of weekly alcohol intake. Patients older than 75 years or those with a significant weekly alcohol intake receive 5 mg as a loading dose. The laboratory-based program then assumes responsibility for the daily dose determination of warfarin for the patient. Using the daily INR result from the laboratory information system, the program calculates the recommended dose based on the parameters of the system and issues a daily report for the ward staff (Fig. 1). The patient’s degree of control while on the system is incorporated into the program once 3 sets of results have been entered.
Example of a daily report showing the international normalized ratios (INR results) and dose recommendations,
The nursing staff administers the recommended dose and has the option to notify the attending physician of any concern related to the patient. The responsible physician can modify the dose or withdraw the patient from the program and provide individualized management thereafter, based on clinical judgement. Patients who do not meet the entry criteria are placed in a separate clinical pathway and managed individually by the attending medical staff.
If a test result exceeds the recommended maximum (INR >3.0) at any stage, a dose recommendation of 0 mg of warfarin is printed on the result sheet. The patient is placed in an “on hold” status until the daily test result returns to the therapeutic range whereupon the recommended dose is printed again. The program incorporates this information into subsequent dose calculations and recommendations. The length of hospital stay determines the duration of warfarin therapy, with the majority of patients being discharged either to a rehabilitation hospital or home approximately 9 days after surgery.
Measures for complications
Bleeding, which was indicated by wound swelling, obvious blood loss or a subsequent fall in the first day postoperative hemoglobin value of greater than 20 g/L, was monitored by the medical and nursing staff. Surveillance venography and ventilation-perfusion studies were not performed prospectively but only on clinical suspicion of a possible deep venous thrombosis or pulmonary embolism. Hospital and radiology records for 2 18-month periods, one before and one immediately after introduction of the program were reviewed as to the number of readmissions, the reason for the readmission and any positive venograms performed during these periods. This sample included 1655 patients managed on the program.
Follow-up
Five hundred patients from the initial study period were randomly selected in mid-1996 and their hospital records reviewed. Hospital staff then attempted to contact by telephone the 430 patients identified as having been managed on the program. Those patients successfully contacted were requested to participate in a telephone interview and were placed into 3 groups based on their duration of warfarin therapy (Table 1).
Duration of Warfarin Therapy in 430 Patients Randomly Selected for Follow-up
Analysis of the findings
An external information systems expert was contracted to fully validate the program including the safety features. Statistical analysis of readmission rates was by χ2 analysis.
Costs for warfarin and one low-molecular-weight heparin preparation were calculated from pharmacy data and a literature source.8 The total comparable costs were calculated on a once daily dose of each agent for all the patients over the study period. Nursing costs were assessed by a limited, internal time-work study of the process involved in dose determination before and after initiation of the program and were calculated at a salary cost with benefits of $30.00/h (all figures in Canadian dollars).
Results
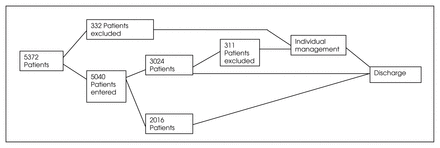
During the 54-month study period 5372 patients underwent primary and revision hip and knee joint arthroplasty and received warfarin prophylaxis (Fig. 2). Of these, 5040 patients (94%) met the entry criteria and were admitted to the program; the other 332 patients (6%) were excluded for clinical or laboratory reasons. Of the 5040 patients in the program, 4729 (94%) completed the program. At some time during their therapy, 3024 (60%) were placed “on hold” with a recommended dose of 0 mg warfarin; 28% for 1 day only and 72% for more than 1 day, either sequentially or more frequently non-sequentially; the remainder, not placed “on hold” continued to receive their daily dose based on the program’s recommendations. Three hundred and eleven (6%) of the 5040 patients entered were removed from the program permanently. These patients exhibited erratic responses to warfarin and were withdrawn by their attending physician or surgeon. Patients were discharged after a mean hospital stay of 8.7 days after unilateral primary hip arthroplasty and 9.0 days after primary knee arthroplasty.
Flow diagram of the study results.
The 4729 patients produced 51 810 test results, which were compared to the historical control group of 279 patients individually managed who produced 2889 test results obtained using the same test system and available in the computer system: 62% of the test results were maintained in the therapeutic range (95% confidence interval [CI] 60.6%–63.4%); 13% were above (95% CI 12%–14%) and 24% were below (95% CI 22.8–25.2%). These compared with rates of 52%, 32% and 16%, respectively, obtained from the historical controls. (Comparison of the patient groups after introduction of the program was not considered valid as the individually managed group was biased toward more difficult to manage patients.)
Less than 0.5% of patients suffered major and minor bleeding episodes, a rate similar to that in the historical control group. Readmission rates for suspected thrombotic disease were unchanged for the 18 months before and after introduction of the program in the sample cohort whose charts were reviewed (1655 patients) (2.96% v. 3.75%, p = 0.089).
Of the 500 randomly selected patients reviewed, 430 were successfully questioned. Nineteen (4%) patients had a suspected deep venous thrombosis, 3 were treated on clinical suspicion and excluded from the analysis, and 16 (3.7%, 95% CI 1.9%–5.5%) had a venographically proven deep venous thrombosis. One patient had a pulmonary embolus on postoperative day 42 for a total morbidity of 4.7% (95% CI 2.7%–6.7%). There were no deaths from thromboembolic disease in this sample. When stratified by duration of warfarin therapy, the single case of pulmonary embolism occurred in the early discharge group of 127 patients (group I, Table 1), 4 of the 16 positive cases of deep venous thrombosis occurred in group II patients (6.6%, 95% CI 4.2%–9.0%) and 12 of the 16 positive cases of deep venous thrombosis occurred in group III patients (5.0%, 95% CI 2.8%–7.2%).
The once-daily cost of warfarin administration (34¢) with the specimen collection and laboratory testing costs of $10.50/patient daily was calculated to be $10.84. The cost of a low-molecular-weight heparin preparation ($13.80/d) with nursing administration costs of $5.00/d was calculated to be $18.80, which would be a daily variance of $8.46 for each patient. Potential nursing administration cost savings in determining the daily warfarin dose were based on a time differential of 15 minutes ($7.50/patient daily) before introduction of the program, based on obtaining the test result, referencing the algorithm and determining the required oral dose. This was reduced to 4 minutes ($2.00/patient daily) after program introduction, based on reading the recommended dose from the result form and supplying the tablet to the patient during established medication rounds. The time saving is based on a salary of $30.00/h for one full-time nurse a year ($58 612, 1950 h/yr). The potential savings per patient would thus be 11 minutes of nursing time or $5.50/patient daily for a total annual figure, based on 10 152 patient-days/yr of $55 836 (all figures in Canadian dollars).
Discussion
Successful oral anticoagulation and the degree of therapeutic control achieved is a function of the patient population, place of care and caregiver, as well as the manner of laboratory monitoring, and, in general, requires significant administrative and nursing time. The use of specialized consultants, clinics or computerized programs in the management of patients on oral anticoagulation has evolved mainly in Europe and the United Kingdom.9–15 Control of oral anticoagulant therapy can be assessed in a number of ways; the number of INR results within the target therapeutic range expressed as a percentage of the total number obtained, the proportion of time within the therapeutic INR range (according to the Rosendaal method or the proportion of all patients in the INR target ratio at a certain point in time.16
This study used the proportion of INR results within the therapeutic range since all patients underwent daily testing, thereby negating any possible patient selection bias and varying testing intervals inherent to outpatient testing systems. This method also allowed direct comparison with the internal control group and the results reported utilizing a similar format.14 Direct literature comparisons with patients on relatively stable doses of warfarin are difficult because of the expected degree of instability during induction of anticoagulation.
Surveys of outpatient anticoagulation in the UK have found that patients on long-term therapy are only adequately anticoagulated 50% to 85% of the time.14 A review of 4 computerized programs in the UK showed that therapeutic test values were maintained in between 51.5% and 59.4% of visits.14 In a multicentre, comparative study of traditional control versus computerized control in an outpatient setting, only 45% of patients in the traditional arm were in therapeutic control during the first 3 weeks of the stabilization period.15 Literature on the use of computerized programs in the inpatient setting during the induction or pre-stabilization period is also limited, and physicians may be reluctant to trust a computerized program to care for their patients during this phase of treatment.15
The computer program tended to be more conservative in dosing recommendations and the proportion of patients with high INR test results decreased from 32% in the historical control group to 13% using the program. This was associated with an increase in the undercoagulated group from 16% to 24%. The proportion of INR results in our study that were below the therapeutic range (24%) was similar to those reported in the UK review of 5 sites in both the computer dose and traditional groups (29% and 36% respectively).15 The percentage of high INRs at 13% also compared favourably to those reported in both computer dose and traditional arms at 14% and 16% respectively. These results were similar to those reported from an Italian study in which the percentages of low and high INRs were 26% and 6% respectively. 17 These reviews have concluded that computerized programs were at least as effective as traditional methods and that for patients requiring a higher INR therapeutic range, the computer programs showed a significant benefit.14,15
We also elected to begin monitoring the tests on day 2 of therapy, although the warfarin effect may take several days to manifest depending on the size of the loading dose. Reported studies indicate that despite careful monitoring, 25% to 30% of values will fall outside the therapeutic range whether the patient is managed individually or by a computerized protocol.14,15 It has been postulated that considerable changes in serial test results may be caused by random fluctuation alone.18
The percentage of patients requiring automatic dose adjustments by being placed “on hold” was considered too high, and some minor modifications of the software were attempted in 1996 to reduce the sensitivity of the program to minimal changes in the daily INR result. Poller and colleagues15 showed that 65% of patients required dose changes in the traditional arm during the first 3 weeks of therapy, indicating the relative instability of the patient’s individual response to the effect of the drug. The duration of the “on hold” order was 1 dosing period or 1 day in 28% of those patients placed “on hold,” whereas 72% were “held” for more than 1 day by the program. Comparative data are difficult to obtain for patients managed by manual methods. However, this statistic remained unacceptably high, and the software was rewritten in an attempt to smooth out the initiation phase, and this update was introduced in January 1999. Studies are underway to examine the effect of this change as well as possibly reducing the 10-mg loading dose of warfarin to 5 mg for all patients entered into the program. The majority of the patients are elderly, and the initiation of therapy may be particularly difficult in these patients, often interacting with dietary and medication changes as well as postoperative hemodynamic changes as a result of blood loss.
The deep venous thrombosis rate of 3.8% in the followed-up patient sample was not based on prospective venography surveillance and reflects only clinically suspected and venographically proven cases. Venous thromboembolism is most often clinically silent and thus underreported in this study.5 The rate of thromboembolic events compares to previously reported rates of 0.8% to 3.4% in patients receiving warfarin prophylaxis. 3,19,20 The sample size is small and must be considered when reviewing the significance of these results. Additional recruitment was attempted but proved impossible for logistical reasons. The cost estimates and potential cost savings are speculative and are meant to be illustrative and not conclusive in nature. Accurate workflow analysis and costing would be integral to the assessment of the overall cost-effectiveness of the program and the amelioration of the frequently expressed “hassle factor” and administrative time associated with the traditional model of oral anticoagulation.
Summary and conclusions
We have reported the largest experience in the literature with a computerized decision-support system for the management of inpatient oral anticoagulation. The program proved to be as safe and effective as manual control for the most patients receiving warfarin as thromboprophylaxis for major orthopedic surgery. It provides significant time for those patients requiring individualized attention. The memory capacity of the program makes data collection simple and will facilitate the comparison of clinical trials. The software is also portable so that patients discharged from hospital to other institutions can be maintained on the program during their rehabilitation provided the laboratory testing systems and testing periods are comparable. This may become more relevant as the period of postoperative anticoagulation increases. The software is being rewritten to allow for variable testing periods suitable for an outpatient setting, and studies are ongoing in attempts to reduce the number of patients requiring dose changes during the initiation period by decreasing the sensitivity of the program to minimal changes in the INR result.
Acknowledgements
Joseph Tang, BSc, MLT, of the laboratory staff wrote parts of the computer program. The program is the property of the Orthopaedic & Arthritic Institute. Dr. Roger Brighton, Department of Surgery and Ms. Lisa Kwok, BScPhm, Department of Pharmacy performed the follow-up chart audit and telephone survey. Mrs. Tina Serpe provided secretarial assistance.
- Accepted February 20, 2001.