Abstract
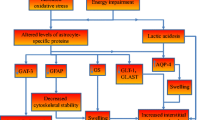
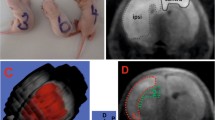
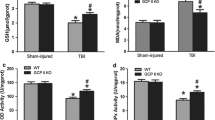
Wernicke's Encephalopathy (WE) is a serious neurological disorder resulting from thiamine deficiency, encountered in chronic alcoholics and in patients with grossly impaired nutritional status. Neuropathologic studies as well as Magnetic Resonance Imaging reveal selective diencephalic and brainstem lesions in patients with WE. The last decade has witnessed major advances in the understanding of pathophysiologic mechanisms linking thiamine deficiency to the selective brain lesions characteristic of WE. Activities of the thiamine-dependent enzyme α-ketoglutarate dehydrogenase, a rate-limiting tricarboxylic acid cycle enzyme are significantly reduced in autopsied brain tissue from patients with WE and from rats treated with the central thiamine antagonist, pyrithiamine. In the animal studies, evidence suggests that such enzyme deficits result in focal lactic acidosis, cerebral energy impairment and depolarization resulting from increased release of glutamate in vulnerable brain structures. It has been proposed that this depolarization may result in N-Methyl-D-Aspartate receptor-mediated excitotoxicity as well as increased expression of immediate early genes such as c-fos and c-jun resulting in apoptotic cell death. Other mechanisms involved in thiamine deficiency-induced cell loss may involve free radicals and alterations of the blood-brain barrier. Additional studies are still required to identify the site of the initial cellular insult and to explain the predilection of diencephalic and brainstem structures due to thiamine deficiency.
Similar content being viewed by others
REFERENCES
Aikawa, H., Watanabe, I.S., Furuse, T., Iwasaki, Y., Satoyoshi, E., Sumi, T., and Moroji, T. (1984). Low energy levels in thiamine-deficient encephalopathy. J. Neuropath. Exp. Neurol. 43:276–287.
Armstrong-James, M., Ross, D.T., Chen, F., and Ebner, F.F. (1988). The effect of thiamine deficiency on the structure and physiology of the rat forebrain. Metab. brain Dis. 3:91–124.
Au, A.M., Chan, P.H., and Fishman, R.A. (1985). Stimulation of phospholipase A2 activity by oxygen-derived free radicals in isolated brain capillaries. J. Cell Biochem. 27:449–453.
Avdulov, N.A., Chochina, S.V., Igbavboa, U., O'Hare, E.O., Schroeder, F., Cleary, J.P., and Wood, W.G. (1997). Amyloid β-peptides increase annular and bulk fluidity and induce lipid peroxidation in brain synaptic plasma membranes. Neurochem. 68:2086–2091.
Bartel, D.P., Sheng, M., Lau, L.F., and Greenberg, M.E. (1989). Growth factors and membrane depolarization activate distinct programs of early response gene expression: dissociation of fos and jun induction. Genes Dev. 3:304–313.
Benavides, J., Fage, D., Carter, C., and Scatton, B. (1987). Peripheral-type benzodiazepine binding sites are a sensitive index of neuronal damage. Brain Res. 421:167–172.
Bettendorff, L., Sluse, F., Goessens, G., Wins, P., and Grisar, T. (1995). Thiamine deficiency-induced partial necrosis and mitochondrial uncoupling in neurobalstoma cells are rapidly reversed by addition of thiamine. J. Neurochem. 65:2178–2184.
Blanc, E.M., Toborek, M., Mark, R.J., Hennig, B., and Mattson, M.P. (1997). Amyloid β-peptide induces cell monolayer albumin permeability, impairs glucose transport, and induces apoptosis in vascular endothelial cells. J. Neurochem. 68:1870–1881.
Brightman, M.W. (1977). Morphology of blood-brain interfaces. Exp. Eye Res. (Suppl) 25:1–25.
Butterworth, R.F. (1982). Neurotransmitter function in thiamine-deficiency encephalopathy. Neurochem Int 4:449–464.
Butterworth, R.F. and Héroux, M. (1989). Effect of pyrithiamine treatment and subsequent thiamine rehabilitation on regional cerebral amino acids and thiamine-dependent enzymes. J. Neurochem. 52:1079–1084.
Butterworth, R.F., Giguère, J.F., and Besnard, A.M. (1985). Activities of thiamine-dependent enzymes in two experimental models of thiamine-deficiency encephalopathy. 1. The pyruvate dehydrogenase complex. Neurochem. Res. 10:1417–1428.
Butterworth, R.F., Giguère, J.F., and Besnard, A.M. (1986). Activities of thiamine-dependent enzymes in two experimental models of thiamine-deficiency encephalopathy. 2. αketoglutarate dehydrogenase. Neurochem. Res. 11:567–577.
Butterworth, R.F., Gaudreau, C., Vincelette, J., Bourgault, A.M., Lamothe, F., and Nutini, A.M. (1991). Thiamine deficiency and Wernicke's encephalopathy in AIDS. Metab. Brain Dis. 6:207–212.
Butterworth, R.F., Kril, J.J., and Harper, C.G. (1993). Thiamine-dependent enzyme changes in the brains of alcoholics: relationship to the Wernicke-Korsakoff syndrome. Alcohol.Clin. Exp. Res. 17:1084–1088.
Calingasan, N., Baker, H., Sheu, K-F.R., Gibson, G.E. (1995a). Blood-brain abnormalities in vulnerable brain regions during thiamine deficiency. Exp. Neurol. 134: 64–72.
Calingasan, N.Y., Gandy, S.E., Baker, H., Sheu, K-F.R., Kim, K-S., Wisniewski, H.M., and Gibson, G.E. (1995b). Accumulation of amyloid precursor protein-like immunoreactivity in rat brain in response to thiamine deficiency. Brain Res. 677:50–60.
Calingasan, N.Y., Gandy, S.E., Baker, H., Sheu, K-F.R., Smith, J.D., Lamb, B.T., et al. (1996). Novel neuritic clusters with accumulations of amyloid precursor protein and amyloid precursor-likeprotein 2 immunoreactivity in brain regions damaged by thiamine deficiency. Am. J. Pathol. 149:1063–1071.
Calingasan, N., Park, L.C.H., Calo, L.L., Trifiletti, R.R., Gandy, S.E., and Gibson, G.E. (1998). Induction of nitric oxide synthase and microglial responses precede selective cell death induced by chronic impairment of oxidative metabolism. Am. J. Path., in press.
Chan, P.H., Schmidley, J.W., Fishman, R.A., and Longar, S.M. (1984). Brain injury, edema, and vascular permeability changes induced by oxygen-derived free radicals. Neurology 34:315–320.
Charness, M.E. and Delapaz, R.L. (1987). Mammillary body atrophy in Wernicke's encephalopathy: antemortem identification using magnetic resonance imaging. Ann. Neurol. 22:595–600.
Cheney, D.L., Gubler, C.J., and Jaussi, A.W. (1969) Production of acetylcholine in rat brain following thiamine deprivation and treatment with thiamine antagonists. J. Neurochem. 16:1283–1291.
Collins, G.H. (1967). Glial cell changes in the brainstem of thiamine-deficient rats. Am. J. Pathol. 50: 91–814.
Colotta, F., Polentarutti, N., Sironi, M., and Mantovani, A. (1992). Expression of c-fos and c-jun protooncogenes in programmed cell death induced by growth factor deprivation in lymphoid cell lines. J. Biol. Chem. 267:18278–18283.
Cooper, J.R. (1968). The role of thiamine in nervous tissue: The mechanism of action of pyrithiamine. Biochim. Biophys. Acta 156: 368–373.
De Caro, L., Rindi, G., and De Guiseppe, L. (1961). Contents in rat tissue of thiamine and its phosphates during dietary thiamine deficiency. Int. Z. Vitaminforsch. 31: 333–340.
Del Maestro, R.F., Bjork, J., and Arfors, K-E. (1981). Increase in microvascular permeability induced by enzymatically generated free radicals. Microvasc. Res. 22:255–270.
Demmel, U., Hock, A., Feinendegen, L.E., and Sebek, P. (1984). Trace elements in brains of patients with alcohol abuse, endogenous pyschosis and schizophrenia. Sci. Total Environ. 38:69–77.
Diorio, D., Welner, S.A., Butterworth, R.F., Meaney, M.J., and Suranyl-Cadotte, B.E. (1991). Peripheral benzodiazepine binding sites in Alzheimer's disease frontal and temporal cortex. Neurobiol. Aging 12:255–258.
Dodd, P.R., Kril, J.J., Thomas, G.J., Watson, W.E.J., Johnston, G.A.R., and Harper, C.G. (1996a). Receptor binding sites and uptake activities mediating GABA neurotransmission in chronic alcoholics with Wernicke encephalopathy. Brain Res. 710:215–228.
Dodd, P.R., Thomas, G.J., McCloskey, A., Crane, D.I., and Smith, I.D. (1996b). The neurochemical pathology of thiamine deficiency: GABAA and glutamateNMDA receptor binding sites in a goat model. Metab. Brain Dis. 11:39–54.
Donnal, J.F., Heinz, E.R., and Burger, P.C. (1990). MR of reversible thalamic lesions in Wernicke syndrome. Am. J. Neuroradiol. 11:893–894.
Dragunow, M., Young, D., Hughes, P., MacGibbon, G., Lawlor, P., Singleton, K., et al. (1993). Is c-jun involved in nerve cell death following status epilepticus and hypoxic-ischemic injury? Mol. Brain Res. 18: 347–352.
Dreyfus, P.M. (1962). Clinical application of blood transketolase determinations. N. Engl. J. Med. 267: 596–598.
Dreyfus, P.M. (1967). Thiamine Deficiency: Biochemical Lesions and their Clinical Significance. In Wolstenholme, G.E.W. and O'Connor, M. (eds.), Ciba Foundation Study Group No. 28., J. & A. Churchill, London, pp. 103–111.
Elnageh, K.M. and Gaitonde, M.K. (1988). Effect of a deficiency of thiamine on brain pyruvate dehydrogenase: Enzyme assay by three different methods. J. Neurochem. 51:1482–1489.
Estus, S., Zaks, W.J., Freeman, R.S., Gruda, M., Bravo, R., and Johnson, E.M., Jr (1994). Altered gene expression in neurons during programmed cell death: Identification of c-jun as necessary for neuronal apoptosis. J. Cell. Biol. 127:1717–1727.
Farber, J.L., Chien, K.R., and Mittnacht, R.B. Jr. (1981) The pathogenesis of irreversible cell injury in ischemia. Am. J. Pathol. 102: 71–281.
Farde, L., Pauli, S., Litton, J.-E., Halldin, C., Neiman, J., and Sedvall, G. (1994). PET-determination of benzodiazepine receptor binding in studies on alcoholism. Experientia 71:143–153.
Forloni, G., Chiesa, R., Smiroldo, S., Verga, L., Salmona, M., Tagliavini, F., and Angeretti, N. (1993). Apoptosis mediated neurotoxicity induced by chronic application of β amyloid fragment 25–35. Neuroreport 4:523–526.
Gaitonde, M.D., Fayein, N.A., and Johnson, A.L. (1975). Decreased metabolism in vivo of glucose into amino acids of the brain of thiamine-deficient rats after treatment with pyrithiamine. J. Neurochem. 24:1215–1223.
Gallucci, M., Bozzao, A., Splendiani, A., Masciocchi, C., and Passariello, R. (1990). Wernicke encephalopathy: MR findings in five patients. Am. J. Neuroradiol. 11:887–892.
Gibson, G., Barclay, L., and Blass, J. (1982) The role of the cholinergic system in thiamin deficiency. Ann. N.Y. Acad. Sci. 378: 382–403.
Gibson, G.E., Ksiezak-Reding, H., Sheu, K.F.R., Mykytyn, V., and Blass, J.P. (1984). Correlation of enzymatic, metabolic and behavioural deficits in thiamine deficiency and its reversal. Neurochem. Res. 9:803–814.
Giguère, J.F. and Butterworth, R.F. (1987). Activities of thiamine-dependent enzymes in two experimental models of thiamine-deficiency encephalopathy. 3. Transketolase. Neurochem. Res. 12:305–310.
González-Martín, C., de Deigo, I., Crespo, D., and Fairen, A. (1992). Transient c-fos expression accompanies naturally occurring cell death in the developing interhemispheric cortex of the rat. Dev. Brain Res. 68:83–95.
Gubler, C.J. (1968). Enzyme studies in thiamin deficiency. Int. J. Vit. Res. 38:287–303.
Gubler, C.J., Adams, B.L., Hammond, B., Yuan, E.C., Guo, S.M., and Bennion, M. (1974). Effect of thiamine deprivation and thiamine antagonists on the level of γ-aminobutyric acid and on 2-oxoglutarate metabolism in rat brain. J. Neurochem. 22:831–836.
Haas, R.H. (1988). Thiamin and the brain. Ann. Rev. Nutr. 8:483–515.
Hakim, A.M. (1984). The induction and reversibility of cerebral acidosis in thiamine deficiency. Ann. Neurol. 16:673–679.
Hakim, A.M. and Pappius, H.M. (1983). Sequence of metabolic, clinical, and histological events in experimental thiamine deficiency. Ann. Neurol. 13:365–375.
Hakim, A.M., Carpenter, S., and Pappius, H.M. (1983). Metabolic and histological reversibility of thiamine deficiency. J. Cereb. Blood Flow Metab. 3:468–477.
Hakim, A.M. and Hogan, M.J. (1991). In-vivo binding of nimodipine in brain: 1. The effect of focal cerebral ischemia. J. Cereb. Blood Flow Metab. 11:762–770.
Hamilton, W.J., Boyd, J.D., and Mossman, H.W. (1952). Human Embryology. Prenatal Development of Form and Function. 2nd Ed. W. Heffer and Sons, Cambridge.
Harata, N. and Iwasaki, Y. (1995). Evidence for early blood-brain barrier breakdown in experimental thiamine deficiency in the mouse. Metab. Brain Dis. 10:565–576.
Harper, C. (1979). Wernicke's encephalopathy: a more common disease than realised. J. Neurol. Neurosurg. Psychiat. 42:226–231.
Harper, C. and Kril, J. (1991). If you drink your brain will shrink. Neuropathological considerations. Alcohol Alcohol. (Suppl.) 1: 375–380.
Harper, C., Giles, M., and Finlay-Jones, R. (1986). Clinical signs in the Wernicke-Korsakoff complex: a retrospective analysis of 131 cases diagnosed at necropsy. J. Neurol. Neurosurg. Psychiat. 49: 341–345.
Hazell, A.S. and Hakim, A.M. (1994). Increase in extracellular glutamate concentration is a Ca2+-independent process in the thalamus of the thiamine deficient rat. J. Neurochem. (Suppl) 62: S104.
Hazell, A.S., Butterworth, R.F., and Hakim A.M. (1993). Cerebral vulnerability is associated with selective increase in extracellular glutamate concentration in experimental thiamine deficiency. J.Neurochem. 61:1155–1158.
Hazell, A.S. and Butterworth, R.F. (1997). Early alterations in blood-brain barrier permeability to α-aminoisobutyric acid (AIB) during experimental thiamine deficiency. J. Neurochem. (Suppl.) 69:S282.
Hazell, A.S., Hakim, A.M., Senterman, M.K., and Hogan, M.J. (1998a). Regional activation of L-type voltage-sensitive calcium channels in experimental thiamine deficiency. J. Neurosci. Res. 52:742–749.
Hazell, A.S., McGahan, L., Tetzlaff, W., Bedard, A.M., Robertson, G.S., Nakabeppu, Y., and Hakim, A.M. (1998b). Immediate-early gene expression in the brain of the thiamine deficient rat. J. Molec. Neurosci. 10:1–15.
Heinrich, C.P., Stadler, H., and Weiser, H. (1973). The effect of thiamin deficiency on the acetylcoenzyme-A and acetylcholine levels in the rat brain. J. Neurochem. 21:1273–1281.
Héroux, M. and Butterworth, R.F. (1988). Reversible alterations of cerebral γ-aminobutyric acid in pyrithiamine-treated rats: Implications for the cerebral pathogenesis of Wernicke's Encephalopathy. J. Neurochem. 51:1221–1226.
Héroux, M. and Butterworth, R.F. (1995). Regional alterations of thiamine phosphate esters and of thiamine diphosphate-dependent enzymes in relation to function in experimental Wernicke's encephalopathy. Neurochem. Res. 20: 87–93.
Hirano, A., Kawanami, T., and Liena, J. (1994). Electron microscopy of the blood-brain barrier in disease. Microvasc. Res. Tech. 27:543–556.
Hofmann, E., Friedburg, H., Rasenack, J., Ott, D., and Wimmer, B. (1988). Die Wernicke-Enzephalopathie im CT and MR. Fortschr. Röntgenstr. 148:97–98.
Holowach, J., Kauffman, F., Ikossi, M.G., Thomas, C., and McDougal, D.B. Jr. (1968). The effects of a thiamine antagonist, pyrithiamine, on levels of selected metabolic intermediates and on activities of thiamine-dependent enzymes in brain and liver. J. Neurochem. 15: 621–631.
Holthoff, V.A., Koeppe, R.A., Frey, K.A., Penney, J.B., Markel, D.S., Kuhl, D.W., and Young, A.B. (1993). Positron emission tomography measures of benzodiazepine receptors in Huntington's disease. Ann. Neurol. 34:76–81.
Iadecola, C. (1997). Bright and dark sides of nitric oxide in ischemic brain injury. Trends Neurosci. 20: 132–139.
Jenkins, L.W., Becher, D.P., and Coburn, T.H. (1984). A quantitative analysis of glial swelling and ischemic neuronal injury following complete cerebral ischemia. In Go, T.G. and Baethmann, A. (eds.), Recent Progress in the Study and Therapy of Brain Edema, Plenum, New York, pp. 523–537.
Johnson, L.R. and Gubler, C.J. (1968). Studies on the physiological functions of thiamine. III. The phosphorylation of thiamine in brain. Biochim. Biophys. Acta 156:85–96.
Joó, F. (1971). Increased production of coated vesicles in the brain capillaries during enhanced permeability of the blood-brain barrier. Br. J. Exp. Pathol. 52:646–649.
Junck, L., Olson, J.M.M., Ciliax, B.S., Koeppe. R.A., Watkins, G.L., Jewett, D.M., et al. (1989). PET imaging of human gliomas with ligands for the peripheral benzodiazepine binding site. Ann. Neurol. 26:752–758.
Kalimo, H., Rehncrona, S., Söderfeldt, B., Olsson, Y., and Siesjö, B.K. (1981). Brain lactic acidosis and ischemic cell damage: 2. Histopathology. J. Cereb. Blood Flow Metab. 1: 313–327.
Kessler, R.M., Clark, C.M., Parker, E.S., Martin, P., Sokoloff, L., Ebert, M.H., and Mishkin, M. (1984). Regional cerebral glucose use in patients with alcoholic Korsakoff's syndrome. Clin. Nucl. Med. (Abstr) 9:65.
Kimelberg, H.K., Rutledge, E., Goderie, S., and Charniga, C. (1995). Astrocytic swelling due to hypotonic or high K+ medium causes inhibition of glutamate and aspartate uptake and increases their release. J. Cereb. Blood Flow Metab. 15:409–416.
Kinnersley, H.W. and Peters, R.A. (1930). Brain localization of lactic acidosis in avitaminosis B1 and its relation to the origin of symptoms. Biochem. J. 24: 711–722.
Koh, J.-Y., Yang, L.L., and Cotman, C.W. (1990). β-Amyloid protein increases the vulnerability of cultured cortical neurons to excitotoxic damage. Brain Res. 533:315–320.
Kril, J.J., Halliday, G.M., Svoboda, M.D., and Cartwright, H. (1997). The cerebral cortex is damaged in chronic alcoholics. Neuroscience 79:983–998.
Langlais, P.J. and Mair, R.G. (1990). Protective effects of the glutamate antagonist MKB801 on pyrithiamine-induced lesions and amino acid changes in rat brain. J. Neurosci. 10:1664–1674.
Langlais, P.J. and Zhang, S.X. (1993). Extracellular glutamate is increased in thalamus during thiamine deficiency-induced lesions and is blocked by MK-801. J. Neurochem. 61: 2175–2182.
Langlais, P.J., Anderson, G., Guo, S.X., and Bondy, S.C. (1997). Increased cerebral free radical production during thiamine deficiency. Metab. Brain Dis. 12:137–143.
Lê, O., Héroux, M., and Butterworth, R.F. (1991). Pyrithiamine-induced thiamine deficiency results in decreased Ca2+-dependent release of glutamate from rat hippocampal slices. Metab. Brain Dis. 6:125–132.
Leong, D. and Butterworth, R.F. (1994). Neuronal cell death in Wernicke's encephalopathy: pathophysiologic mechanisms and implications for PET imaging. Metab. Brain Dis. 11: 71–79.
Leong, D., Lê, O., Oliva, L., and Butterworth, R.F. (1994). Increased densities of binding sites for the "peripheral-type" benzodiazepine receptor ligand [3H]PK11195 in vulnerable regions of the rat brain in thiamine deficiency encephalopathy. J. Cereb. Blood Flow Metab. 14:100–105.
Leong, D.K. and Butterworth, R.F. (1996). Neuronal cell death in Wernicke's encephalopathy: pathophysiologic mechanisms and implications for PET imaging. Metab. Brain Dis. 11:71–79.
Lindboe, C.F. and Loberg, E.M. (1989). Wernicke's encephalopathy in non-alcoholics. An autopsy study. J. Neurol. Sci. 90:125–129.
Loo, D.T., Copani, A., Pike, C.J., Whittemore, E.R., Walencewicz, A.J., and Cotman, C.W. (1993). Apoptosis is induced by beta-amyloid in cultured central nervous system neurons. Proc. Natl. Acad. Sci. USA 90:7951–7955.
Lissak, K., Kovacs, T., and Nagy, E.K. (1943). Acetylcholin-und cholinesterasegehalt von organen B1-avitaminotischer und normaler ratten. Pflugers Arch. Ges. Physiol. 247:124–131.
Mann, P.J.G. and Quastel, J.H. (1940). Vitamin B1 and acetylcholine formation in isolated brain. Nature 145:856–857.
Manz, H.J. and Robertson, D.M. (1972). Vascular permeability to horseradish peroxidase in brainstem lesions of thiamine-deficient rats. Am. J. Pathol. 66:565–576.
Mattson, M.P., Cheng, B., Davis, D., Bryant, K., Lieberburg, I., and Rydel, R.E. (1992). β-Amyloid peptide destabilized calcium homeostasis and render human cortical neurons vulnerable to excitotoxicity. J. Neurosci. 12:376–389.
Matsushima, K., MacManus, P., and Hakim, A.M. (1997). Apoptosis is restricted to the thalamus in thiamine-deficient rats. NeuroReport 8:867–870.
McCandless, D.W. (1982). Energy metabolism in the lateral vestibular nucleus in pyrithiamin induced thiamin deficiency. Ann. NY Acad. Sci. 378:355–364.
McCandless, D.W. and Schenker, S. (1968). Encephalopathy of thiamine deficiency: Studies of intracerebral mechanisms. J. Clin. Invest. 47:2268–2280.
McCandless, D.W., Curley, A.D., and Cassidy, C.E. (1976). Thiamine deficiency and the pentose phosphate cycle in rats: Intracerebral mechanisms. J. Nutr. 106:1144–1151.
McDowell, J.E. and LeBlanc, H.J. (1984). Computed tomographic findings in Wernicke-Korsakoff syndrome. Arch. Neurol. 41:453–454.
Mensing, J.W.A., Hoogland, P.H., and Sloof, J.L. (1984). Computed tomography in the diagnosis of Wernicke's encephalopathy: a radiological-neuropathological correlation. Ann.Neurol. 16:363–365.
Merrill, J. and Murphy, S. (1996). Nitric oxide, in The Role of Glia in Neurotoxicity (Aschner, M. and Kimelberg, H.K., eds.), pp 263–281. CRC Press, Boca Raton.
Miyajima, Y., Fukuda, M., Kojima, S., Matsuyama, T., Shylaja, N., and Aso, K. (1993). Wernicke's encephalopathy in a child with acute lymphoblastic leukemia. Am. J. Ped. Hemat/Onc. 15:331–334.
Moncada, S., Palmer, R.M., and Higgs, E.A. (1991). Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol. Rev. 43:109–142.
Morgan, J.I. and Curran, T. (1986). Role of ion flux in the control of c-fos expression. Nature 322:552–555.
Mosseau, D.D., Rao, V.L.R., and Butterworth, R.F. (1996). Alterations in serotonin parameters in brain of thiamine-deficient rats are evident prior to the appearance of neurological symptoms. J. Neurochem. 67:1113–1123.
Munujos, P., Vendrell, M., and Ferrer, I. (1993). Proto-oncogene c-fos induction in thiamine-deficient encephalopathy. Protective effects of nicardipine on pyrithiamine-induced lesions. J.Neurol. Sci. 118:175–180.
Murdock, D.S. and Gubler, C.J. (1973). Effects of thiamine deficiency and treatment with the antagonists, oxythiamine and pyrithiamine, on the levels and distribution of thiamine derivatives in rat brain. J. Nutr. Sci. Vitaminol. 19: 237–249.
Murphy, S., Minor, R.L., Welk, G., and Harrison, D.G. (1990). Evidence for an astrocyte-derived vasorelaxing factor with properties similar to nitric oxide. J. Neurochem. 55:349–351.
Murphy, T.H., Worley, P.F., and Baraban, J.M. (1991). L-type voltage-sensitive calcium channels mediate synaptic activation of immediate-early genes. Neuron 7:625–635.
Mutch, W.A.C. and Hansen, A.J. (1984). Extracellular pH changes during spreading depression and cerebral ischemia: mechanisms of brain pH regulation. J. Cereb. Blood Flow Metab. 4:17–27.
Myers, R.E. (1979a). Lactic acid accumulation as cause of brain edema and cerebral necrosis resulting from oxygen deprivation. In Korobkin, R. and Guilleminault, G (eds.), Advances in Perinatal Neurology, Spectrum, New York, pp. 85–114.
Myers, R.E. (1979b). A unitary theory of causation of anoxic and hypoxic brain pathology. Adv. Neurol. 26:195–213.
Myers, R., Manjil, L.G., Cullen, B.M., Price, G.W., Frackowiak, S.J. and Cremer, J.E. (1991). Macrophage and astrocyte populations in relation to 3H-PK11195 binding in rat cerebral cortex following a local ischaemic lesion. J. Cereb. Blood Flow Metab. 11:314–322.
Myers, R. (1993). Mitochondrial benzodiazepine receptor ligands as indicators of damage in the CNS: their application in positron emission tomography. In (E. Giesen-Crouse, ed.) Peripheral Benzodiazepine Receptors. San Diego, Academic Press. pp 235–273.
Nose, Y., Iwashima, A., and Nishino, H. (1976). Thiamine uptake by rat brain slices. In Gubler, C.J., Fujiwara, M., and Dreyfus, P.M. (eds.), Thiamine, John Wiley & Sons, New York, pp. 157–168.
Novelli, A., Reilly, J.A., Lysko, P.G., Henneberry, R.C. (1988). Glutamate becomes neurotoxic via the N-methyl-D-aspartate receptor when intracellular energy levels are reduced. Brain Res. 451:205–212.
Ohkoshi, N., Ishii, A., and Shoji, S. (1994). Wernicke's encephalopathy induced by hyperemesis gravidarum, associated with bilateral caudate lesions on computed tomography and magnetic resonance imaging. Eur. Neurol. 34:177–180.
Olney, J.W., Oi Lan, H., and Rhee, V. (1971). Cytotoxic effects of acidic and sulphur containing amino acids on the infant mouse central nervous system. Exp. Brain Res. 14:61–76.
Olney, J.W. (1978). Neurotoxicity of excitatory amino acids. In (McGeer, E.G., Olney, J.W., and McGeer, P.L. eds.), Kainic Acid as a Tool in Neurobiology, Raven Press, New York, pp. 95–121.
Owen, F., Poulter, M., Waddington, J.L., Mashal, R.D., and Crow, T.J. (1983). 3H-Ro5-4864 and 3H-flunitrazepam binding in kainate-lesioned rat striatum and in temporal cortex of brains from patients with senile dementia of the Alzheimer type. Brain Res. 278:373–375.
Papadopoulos, V. (1993). Peripheral-type benzodiazepine diazepam binding inhibitor receptor: biological role in steroidogenic cell function. Endocr. Rev. 14:222–240.
Peters, R.A. (1936). The biochemical lesion in vitamin B1 deficiency. Lancet 1: 1161–1165.
Peters, R.A. (1969). The biochemical lesion and its historical development. Br. Med. Bull. 25:223–226.
Peterson, C., Héroux, M., Lavoie, J., and Butterworth, R.F. (1995). Loss of [3H]kainate and of NMDA-displaceable [3H]glutamate binding sites in brain in thiamine deficiency: results of a quantitative autoradiographic study. Neurochem. Res. 20:1155–1160.
Phillips, S.C. and Cragg, B.G. (1984). Blood-brain barrier dysfunction in thiamine-deficient, alcohol-treated rats. Acta Neuropathol. (Berl) 62:235–241.
Pitella, J.E.H. and Castro, L.P.F. (1990). Wernicke's encephalopathy manifested as Korsakoff's syndrome in a patient with promyelocytic leukemia. South Med. J. 5:570–573.
Plum, F. (1983). What causes infarction in ischemic brain? Neurology 33: 222–233.
Pulsinelli, W.A., Waldman, S., Rawlinson, D., and Plum, F. (1982). Moderate hyperglycemia augments ischemic brain damage: a neuropathologic study in the rat. Neurology 32:1239–1246.
Rao, V.L.R., Mousseau, D.D., and Butterworth, R.F. (1995). A quantitative autoradiographic study of muscarinic cholinergic receptor subtypes in the brains of pyrithiamine-treated rats. Neurochem Res. 20:907–914.
Rao, V.L.R., Mousseau, D.D., and Butterworth, R.F. (1996). Nitric oxide synthase activities are selectively decreased in vulnerable brain regions in thiamine deficiency. Neurosci. Lett. 208:17–20.
Raichle, M.E. (1983). The pathophysiology of brain ischemia. Ann. Neurol. 13:2–10.
Reese, T.S. and Karnovsky, M.J. (1967). Fine structural localization of a blood-brain barrier to exogenous peroxidase. J. Cell. Biol. 34:207–217.
Reynolds, S.F. and Blass, J.P. (1975). Normal levels of acetyl coenzyme A and of acetylcholine in the brain of thiamin-deficient rats. J. Neurochem. 24: 185–186.
Rindi, G. and Perri, V. (1961). Uptake of pyrithiamine by tissue of rats. Biochem. J. 80:214–216.
Rindi, G., De Guiseppe, L., and Ventura, U. (1963). Distribution and phosphorylation of oxythiamine in rat tissues. J. Nutr. 81: 147–154.
Robertson, D.M., Wasan, S.M., and Skinner, D.B. (1968). Ultrastructural features of early brainstem lesions of thiamine-deficient rats. Am. J. Pathol. 52:1081–1097.
Robertson, D.M. and Manz, H.J. (1971). Effect of thiamine deficiency on the competence of the blood-brain barrier to albumin labeled with fluorescent dyes. Am. J. Pathol. 63:393–402.
Roche, S.W., Lane, R.J. and Wade, J.P.H. (1988). Thalamic hemorrhages in Wernicke-Korsakoff syndrome demonstrated by computed tomography. Ann. Neurol. 23:312.
Sacks, T., Moldow, C.F., Craddock, P.R., Bowers, T.K., and Jacob, H.S. (1978). Oxygen radicals mediate endothelial cell damage by complement-stimulated granulocytes. An in vitro model of immune vascular damage. J. Clin. Invest. 61:1161–1167.
Savic, I., Ingvar, M., and Stone-Elander, S. (1993). Comparison of [11C]-flumazenil and [18F]-FDG as PET markers of epileptic foci. J. Neurol. Neurosurg. Psychiat. 56:615–621.
Schanne, F.A.X., Kane, A.B., Young, E.E., and Farber, J.L. (1979). Calcium dependence of toxic cell death: A final common pathway. Science 206:701–702.
Schoemaker, H., Morelli, M., Deshmukh, P., and Yamamura, H.I. (1982). 3H-Ro5-4864 benzodiazepine binding in the kainate lesioned striatum and Huntington's diseased basal ganglia. Brain Res. 248:396–401.
Scholz, W. (1949). Histologische und topische Veräanderungen und vulnerabilitätsverhältnisse im menschlichen Gehirn bei Sauerstoffmangel, Odem und plasmatischen Infiltrationen. I. Problemstellung und feingewebliche Situation. Arch. Psychiat. Nervenkr. 181:621–665.
Schroth, G., Grodd, W., Guhl, L., Grauer, M., Klose, U., and Niendorf, H.-P. (1987). Magnetic resonance imaging in small lesions of the central nervous system. Improvement by gadolinium-DTPA. Acta Radiol. 28:667–672.
Schroth, G., Wichmann, W., and Valavanis, A. (1991). Blood-brain barrier disruption in acute Wernicke encephalopathy: MR findings. J. Comp. Assist. Tomog. 15:1059–1061.
Shah, N. and Wolff, J.A. (1973). Thiamine deficiency: probable Wernicke's encephalopathy successfully treated in a child with acute lymphocytic leukemia. Pediatrics 43:750–751.
Sheng, M., McFadden, G., and Greenberg, M.E. (1990). Membrane depolarization and calcium induce c-fos transcription via phosphorylation of transcription factor CREB. Neuron 4:477–485.
Sheu, K-F.R., Calingasan, N.Y., Dienel, G.A., Baker, H., Jung, E-H., Kim, K-S., Paoletti, F., and Gibson, G.E. (1996). Regional reductions of transketolase in thiamine-deficient rat brain. J. Neurochem. 67:684–691.
Shimamura, A.P., Jernigan, T.L., and Squire, L.R. (1988). Korsakoff's syndrome: radiological (CT) findings and neuropsychological correlates. J. Neurosci. 8: 4400–4410.
Siesjö, B.K. (1985). Acid-base homeostasis in the brain: physiology, chemistry, and neurochemical pathology. Prog. Brain Res. 63:121–154.
Siesjö, B.K. and Bengtsson, F. (1989). Calcium, calcium antagonists and calcium-related pathology in brain ischemia, hypoglycemia and spreading depression: A unifying hypothesis. J. Cereb. Blood Flow Metab. 9:127–141.
Soffer, D., Zirkin, H., Alkan, M., and Berginer, V.M. (1989). Wernicke's encephalopathy in acquired immune deficiency syndrome (AIDS): a case report. Clin. Neuropathol. 8:192–194.
Summers, J.A., Pullan, P.T., Kril, J.J., and Harper, C.G. (1991). Increased central immunoreactiove beta-endorphin content in patients with Wernicke-Korsakoff syndrome and in alcoholics. J. Clin. Pathol. 44:126–129.
Szatkowski, M., Barbour, B., and Attwell, D. (1990). Non-vesicular release of glutamate from glial cells by reversed electrogenic glutamate uptake. Nature 348:443–446.
Thomas, G.J., Harper, C.G., and Dodd, P.R. (1998). Expression of GABAA receptor isoform genes in the cerebral cortex of cirrhotic and alcoholic cases assessed by S1 nuclease protection assays. Neurochem. Int. 32:375–385.
Thompson, S.D. and McGeer, E.G. (1985). GABA-transaminase and glutamic acid decarboxylase changes in the brain of rats treated with pyrithiamine. Neurochem. Res. 10:1653–1660.
Todd, K.G. and Butterworth, R.F. (1997). Immunohistochemical evidence that superoxide dismutase is upregulated in experimental thiamine deficiency. Soc. Neurosci. Abst. 23:829.
Todd, K.G. and Butterworth, R.F. (1998a). Evaluation of the role of NMDA-mediated excitotoxicity in the selective neuronal loss in experimental Wernicke encephalopathy. Exp. Neurol. 149:130–138.
Todd, K.G. and Butterworth, R.F. (1998b). Increased neuronal cell survival after L-deprenyl treatment in experimental thiamine deficiency. J. Neurosci. Res., 52:240–246.
Todd, K.G. and Butterworth, R.F. (1998c). Microglial activation: the initial cellular response in experimental thiamine deficiency. J. Neurochem. (Suppl.) 70: S64.
Torvik, A. (1985). Two types of brain lesions in Wernicke's encephalopathy. Neuropath. Appl. Neurobiol. 11:179–190.
Troncoso, J.C., Johnston, M.V., Hess, K.M., Griffin, J.W., and Price, D.L. (1981). Model of Wernicke's encephalopathy. Arch. Neurol. 38:350–354.
Victor, M., Adams, R.D., and Collins, G.H. (1989). The Wernicke-Korsakoff Syndrome and Related Neurologic Disorders due to Alcoholism and Malnutrition. F.A. Davies, Philadelphia.
Vorhees, C.V., Schmidt, D.E., Barrett, R.J., and Schenker, S. (1977). Effect of thiamin deficiency on acetylcholine levels and utilization in vivo in rat brain. J. Nutr. 107:1902–1908.
Vortmeyer, A.O. and Colmant, H.J. (1988). Differentiation between brain lesions in experimental thiamine deficiency. Virchows. Archiv. A. Pathol. Anat. 414:61–67.
Warnock, L.G. and Burkhalter, V.J. (1968.) Evidence of malfunctioning blood-brain barrier in experimental thiamine deficiency in rats. J. Nutr. 94:256–260.
Watanabe, I. (1978). Pyrithiamine-induced acute thiamine-deficient encephalopathy in the mouse. Exp. Mol.Pathol. 28:381–394.
Watanabe, I. and Kanabe, S. (1978). Early edematous lesion of pyrithiamine induced acute thiamine deficient encephalopathy in the mouse. J. Neuropathol. Exp. Neurol. 37: 01–413.
Watanabe, I., Tomita, T., Hung, K-S., and Iwasaki, Y. (1981). Edematous necrosis in thiamine-deficient encephalopathy of the mouse. J. Neuropathol. Exp. Neurol. 40:454–471.
Wernicke, C. (1881). Lehrbuch der Gehirnkrankheiten fur Aerzte und Studirende, Vol. 2, Theodor Fischer, Kassel, pp. 229–242.
Westergaard, E. and Brightman, M.W. (1973). Transport of proteins across cerebral arterioles. J. Comp. Neurol. 152:17–44.
Witt, E.D. (1985). Neuroanatomical consequences of thiamine deficiency: A comparative analysis. Alcohol Alcoholism 20:201–222.
Yankner, B.A., Duffy, L.K., and Kirschner, D.A. (1990). Neurotrophic and neurotoxic effects of amyloid β protein: reversal by tachykinin neuropeptides. Science 250:279–282.
Yokote, K., Miyagi, K., Kuzuhara, S., Yamanouchi, H., and Yamada, H. (1991). Wernicke encephalopathy: follow-up study by CT and MR. J. Comput. Assist. Tomogr. 15:835–838.
Yoshida, S., Busto, R., Martinez, E., Scheinberg, P., and Ginsberg, M.D. (1985). Regional brain energy metabolism after complete versus incomplete ischemia in the rat in the absence of severe lactic acidosis. J. Cereb. Blood Flow Metab. 5:490–501.
Zafra, F., Hengerer, B., Leibrock, J., Thoenen, H., and Lindholm, D. (1990). Activity dependent regulation of BDNF and NGF mRNAs in the rat hippocampus is mediated by non-NMDA glutamate receptors. EMBO J. 9:3545–3550.
Zhang, S.X., Weilersbacher, G.S., Henderson, S.W., Corso, T., Olney, J.W., and Langlais, P.J. (1995). Excitotoxic cytopathology, progression, and reversibility of thiamine deficiency-induced diencephalic lesions. J. Neuropathol. Exp. Neurol. 54:255–267.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hazell, A.S., Todd, K.G. & Butterworth, R.F. Mechanisms of Neuronal Cell Death in Wernicke's Encephalopathy. Metab Brain Dis 13, 97–122 (1998). https://doi.org/10.1023/A:1020657129593
Issue Date:
DOI: https://doi.org/10.1023/A:1020657129593