Abstract
The term occult pneumothorax (OP) describes a pneumothorax that is not suspected on the basis of clinical examination or plain radiography but is ultimately detected with thoracoabdominal computed tomography (CT). This situation is increasingly common in trauma care with the increased use of CT. The rate is approximately 5% in injured people presenting to hospital, with CT revealing at least twice as many pneumothoraces as suspected on plain radiography. Whereas pneumothorax is a common and treatable cause of mortality and morbidity, there is substantial disagreement regarding the appropriate treatment of OP. The greatest controversy is in patients in the critical care unit who require positive-pressure ventilation. There is little current evidence to direct the proper management of ventilated trauma patients with OP, and no studies have focussed specifically on these patients. Future randomized trials will need to consider the potential effects of OP on pulmonary mechanics and potential influences on the known risks of ventilator-induced lung injury associated with mechanical ventilation.
Thoracic injury accounts for 25% of all trauma-related deaths.1 The second most common blunt chest injury, pneumothoraces are a notable cause of preventable death, but relatively simple interventions may be lifesaving.2–4 Pneumothoraces may be dynamic, with delayed, but life-threatening manifestations arising at any time during the patient’s hospitalization. They may also cause a disproportionate degree of cardiopulmonary instability compared with chest injuries of similar anatomic severity.5,6 They are of particular concern in trauma patients requiring positive-pressure mechanical ventilation because they may progress rapidly to a tension pneumothorax. 7–10 In such cases, the intrapleural pressure rises, the mediastinum shifts and the diaphragm is depressed. These events may be associated with decreased lung capacity, anatomic shunting, hypoventilation and ventilation–perfusion mismatch of collapsed underventilated lungs.11 Cardiac output may be further embarrassed because of obstructive shock.7
Occult pneumothorax (OP) is becoming common owing to the increasing use of computed tomography (CT) as the investigation of choice for blunt thoracoabdominal trauma. OP was originally defined as a pneumothorax identified by CT of the abdomen that was not seen on conventional supine anterosuperior chest radiography (CXR).12–15 OP may be present when conventional chest radiography is clearly abnormal (Fig. 1) or relatively normal (Fig. 2). This is not surprising as CXR is the least sensitive of all the plain radiographic techniques for demonstrating a pneumothorax.5,8,16,17 Cadaver studies require up 400 mL of air in the pleural space with the patient in a supine position to confidently detect pneumothorax.18 Unfortunately, competing concerns regarding spinal injuries, hemodynamic compromise and concomitant clinical interventions make this the only practical view that can be obtained in many cases. Although careful review of the plain chest radiograph may reveal evidence of basilar hyperlucency, a deep sulcus sign or a double diaphragm; these pneumothoraces are often easily seen by studying lung windows on a thoracoabdominal CT scan. With the increasingly frequent finding of these unsuspected pneumothoraces, treating physicians must answer a series of questions. How common are they? How important are they? Do they require formal decompression? The controversy surrounding these questions is no more acute, nor important, than in the critical care unit.
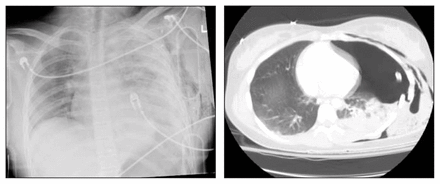
Left: anterosuperior supine chest radiograph of blunt trauma victim revealing left posterolateral rib fractures and parenchymal opacity due to pleural fluid and pulmonary contusion. There is no obvious pneumothorax. Right: computed tomography scan reveals a large occult left-sided pneumothorax.
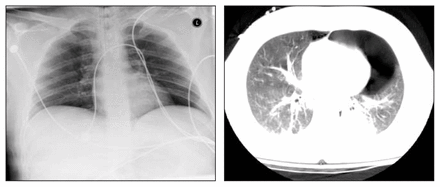
Left: anterosuperior supine chest radiograph of blunt trauma victim. There is no obvious pneumothorax. Right: computed tomography scan reveals a large occult left-sided pneumothorax.
Epidemiology
Overall, OPs are quite common and will be seen regularly in the daily practice of caring for the trauma patient. The specific rate depends on the population in question and whether it has been preselected in some way regarding injury severity or previous diagnostic tests (Table 15,8,12,13,15,18–27). In 1983, Wall and colleagues12 reported that 10 (28%) of 35 pneumothoraces detected by abdominal CT were occult, representing 2% of their 500 patients. Subsequently, a number of authors have reported a remarkably consistent incidence of OPs ranging from 5.2% to 8.0% for injured patients presenting to hospital.8,15,18,19,21,22,25 In 1989 in a series of 174 trauma patients, Rhea and colleagues18 found 8 OPs in 15 patients for a 4.6% incidence. Subsequently, Garramone and associates,19 Wolfman and colleagues,21 Brasel and associates,15 Hill and associates,22 and Neff and colleagues,8 reported rates of 5.7%, 5.4%, 5.2%, 8.0% and 5.4% in groups of 457, 664, 1669, 3121 and 2312 patients, respectively. A pediatric survey also reported a 2% incidence among 538 children.26 In these studies, the proportion of pneumothoraces that were occult compared with those that were actually seen on CXR have ranged from 29% to 72%, the majority having a rate greater than 50% (Table 1).
Incidence of Overt and Occult Pneumothorax (OP) in Trauma Patients
Although the term occult pneumothorax was first defined as a “pneumothorax not seen on CXR but detected on the abdominal CT,”8,14,15,19 chest CT is an intuitive and obvious method of diagnosing additional OPs.25,28 In critically ill people who were effectively triaged by their injury severity to undergo a thoracic CT, the proportion of OPs appears to be even higher. In a study of 25 consecutive patients with severe head trauma, a limited chest CT found 21 pneumothoraces, of which 11 (52%) were occult.13 Four studies have subsequently utilized thoracic CT for high-risk trauma patients, admitted to a critical care unit23,24 or with chest radiographs that revealed a suspicion but not a diagnosis of pneumothorax.5,27 In intubated poly-trauma patients evaluated with thoracic CT, 25 pneumothoraces were detected in 39 patients, 72% of which were not detected by CXR.23 In a Spanish critical care unit, 60% of all the pneumothoraces detected were occult.24 This is consistent with the 71% rate reported from a German unit.5 Rowan and colleagues,27 in Vancouver, examined 27 traumatized patients with thoracic CT and found 11 pneumothoraces of which 7 (64%) were occult. This group examined these same patients with a novel sonographic technique, which had a significantly higher sensitivity for detecting these OPs than CXR.27,29
Thus, the frequency with which a clinician will encounter this entity will depend on how seriously injured the patient is and where the patient is assessed (i.e., in the emergency room or at a later stage of hospitalization). Usually, OPs seem to be found unexpectedly on CT scans in about 5% of the general trauma population. CT typically reveals at least twice as many pneumothoraces as are suspected from the anteroposterior CXR. In the foreseeable future the number of OPs detected in traumatized patients will only increase. Thoracic CT is increasingly being used to investigate the mediastinum, thoracic spine and diaphragm, as well as to detect pulmonary emboli.5,30,31 Thoracic sonography, a simple test with greater sensitivity in detecting OPs, also may play an increasing role.27,32
Significance of occult pneumothorax and its management
Delayed or missed treatment of post-traumatic pneumothoraces has been reported to be a leading cause of preventable morbidity,4,6 so the traditional management of nearly all of them detected clinically or on CXR has been to place a chest tube.7 Guidelines regarding spontaneous pneumothoraces are more liberal, and typically may be treated with observation if a patient is otherwise asymptomatic and the pneumothorax is “small” (< 3 cm from the lung apex to the ipsilateral thoracic cupola33).34 With limited scientific evidence, the significance of the OP has been variously and widely interpreted by different groups of physicians. It is probable that these OPs existed and remained untreated previously but are only now apparent due to our increased use of 3-dimensional imaging. Clinical opinion supports close observation, as long as the patient is asymptomatic and is not ventilated.15,20,22,26,35 Chest tubes should be placed if the OP increases in size or if the patient becomes symptomatic. However, chest tube placement is associated with morbidity: pain, vascular injury, improper positioning of the tube, inadvertent tube removal, post-removal complications, longer hospital stays, empyema and pneumonia have been reported in up to 21% of cases.14,15,35,36
Classification of occult pneumothoraces
It seems intuitive for clinicians to consider the size of a pneumothorax, since the volume of intrapleural air may be related to both the size of the air leak and the time required for spontaneous resolution. Wolfman and colleagues21 have classified OPs as “miniscule,” “anterior” or “anterolateral” on the basis of CT findings. Miniscule pneumothoraces were no more than 1 cm thick and seen on 4 or fewer contiguous 10- mm images. Anterior pneumothoraces were thicker than 1 cm but did not extend posterior to the midthoracic coronal line whereas anterolateral pneumothoraces did; both types comprised 4 or more 10-cm slices.21 This scale has also been used by Holmes and associates.26 Others have directly estimated the size of OPs on CT by describing the maximal width and the number of 10- mm sections in which the pneumothorax appeared.15,19
Occult pneumothorax and positive-pressure ventilation
Although the intubated and ventilated patient is at the greatest risk, proper management of an OP is extremely controversial and based on little scientific evidence. Many of these patients are already compromised owing to acquired or pre-existing pulmonary conditions, and clinical respiratory distress may be masked by concomitant respiratory support and sedation. The risk of progression of a known pneumothorax to a tension pneumothorax has generally been considered a serious concern, warranting prophylactic chest tube placement for the patient who is subjected to positive-pressure ventilation.5,11–13,18,20,25 Kollef37 also reported that in ventilated patients with pneumothoraces a tension pneumothorax was statistically more likely to develop when the diagnosis was missed or delayed. The guidelines of the Advanced Trauma Life Support of the Committee on Trauma of the American College of Surgeons state that general anesthesia or positive-pressure ventilation should never be administered without a chest tube being placed in any patient who has sustained a traumatic pneumothorax or is at risk for an unexpected pneumothorax.7
Other sources have not supported these admonitions concerning OPs. The critical care unit cohort also represents a patient group in which complications of tube thoracostomy have been found to be the highest. Etoch and associates36 reported that intensive care admission and mechanical ventilation were independently associated with increased chest tube complications. This group also represents a population who will be closely observed. Garramone and associates19 retrospectively noted that size of the OP and number of rib fractures affected outcome by influencing when clinicians placed chest tubes (Table 214,15,19,21,22,35). If an OP was less than 5 mm by 80 mm, and associated with 2 or fewer rib fractures, they believed it could be managed conservatively, irrespective of the need for ventilation. Ten (59%) of 17 patients managed without chest tubes were intubated and ventilated with positive pressures. None of these patients required emergency thoracostomy secondary to respiratory failure or hemodynamic compromise; however, 3 of these 10 ultimately required a chest tube because of progression of their pneumothorax. 19 Collins and associates35 retrospectively identified 26 patients with OPs and compared the characteristics and outcomes of 13 who were observed with repeated CXR and 11 who underwent early chest tube placement. Ten of the 26 were ventilated, and 6 of the 10 were managed without a chest tube. Although 1 of these (17%) failed observation (pneumothorax progression), they concluded there was no support for the contention that an OP warranted a prophylactic chest tube prior to positive- pressure ventilation.35 Wolfman and colleagues21 observed OPs in 36 patients. Thirteen of 16 miniscule OPs and 11 of 20 moderate (anterior) pneumothoraces were managed successfully with conservative treatment, for an 11% failure rate of observation, including 1 tension pneumothorax. The 8 remaining anterolateral pneumothoraces were all treated with tube thoracostomy, whether intubated or not. Most of the patients failing observation secondary to progression of the pneumothorax had been intubated and ventilated. These authors concluded from their results that only small and moderate OPs without mechanical ventilation could be safely observed as they had not randomized the anterolateral OPs.21 Hill and associates22 noted that a total of 165 operations were performed on 127 patients with OP. Fifty-one (31%) of the group were on a mechanical ventilator, the average duration of ventilation being 12.6 days. Twenty-seven (16%) patients with OP were observed without a chest tube at some time during mechanical ventilation, although the authors did not specifically delineate the outcome of this. They found that in 5 (29%) of these 27 cases conservative treatmen failed, and the patients required a chest tube for OP progression. There were no tension pneumothoraces, however. They also observed that only the size of the pneumothorax was statistically associated with the need for a chest tube. They felt that only if a general surgeon was not involved in an operative case or if long-term high-pressure ventilation was required was a prophylactic chest tube indicated. Guerrero-Lopez and associates24 stated that OPs did not always require treatment despite mechanical ventilation if they were “small and without complications.”
Outcome Data for Management of Occult Pneumothoraces (OPs)
The best, although very limited, evidence guiding management of OPs in ventilated patients originates from 2 small prospective randomized studies. In 1993, Enderson and associates14 randomized 40 patients with OPs to tube thoracostomy (19) or observation (21), without regard for mechanical ventilation. Fifteen “observed” patients were ventilated as were 12 with early placement of chest tubes. These authors reported that 8 (53%) of the 15 had progression of their OP with positive-pressure ventilation, 3 suffering tension pneumothoraces. None of the patients with tube thoracostomy suffered any major complications while on positive-pressure ventilation, or because of chest tube placement. As a result of this morbidity, the authors strongly recommended that all patients with OP who require mechanical ventilation undergo tube thoracostomy. They also commented that the size of the initial OP was not predictive of progression or of formation of a tension pneumothorax.
Conversely, in 1999, Brasel and associates15 reported on a 24-month, prospective randomized trial in 39 blunt trauma patients with 44 OPs. Nine patients in each group were ventilated, and there was no statistical difference in the sizes of the OPs. There were no differences in complications, and no patients in either group required emergent tube thoracostomy for respiratory compromise. Two of the 9 patients on positive-pressure ventilation did receive a chest tube secondary to progression of the OP. Given the small numbers, this finding did not reach statistical significance, and there was no association between the size of the OP and failure of conservative treatment. Brasel’s group did conclude though that OPs could be safely observed in ventilated patients. It should be noted that one possible explanation for the discrepancy between studies was that the ventilatory management of patients may have varied, as most of Brasel’s patients were in the intensive care unit and most of Enderson’s were in the operative suite. Ventilation strategies in general have also changed substantially over the past decade. Decreased airway pressures and tidal volumes are now routine in the critically ill. Contemporary ventilatory management stresses careful attention to controlling peak and mean airway pressures, limiting pressures below those considered routine previously.38–40
Ventilator-induced lung injury
Any discussion of the influence of OPs in critical care needs to consider the effects of ventilator-induced lung injury. Although reported studies do not show obvious differences in the occurrence of overt cardiopulmonary disasters such as tension pneumothoraces or deaths, the influence of pneumothoraces on the pulmonary mechanics of susceptible patients remains unknown and deserves further study. In this regard, the effects of observing OPs on pulmonary mechanics is poorly described. In the ventilated patient, rises in peak and plateau pressures signal the possibility of a pneumothorax. It is also well known that when the lungs are exposed to high ventilatory volumes, tissue disruption and lung injury may result from combinations of barotrauma, volutrauma and an increased production of inflammatory mediators, or biotrauma.41–43 When ventilatory pressures are not controlled, the incidence of spontaneous pneumothoraces may reach as high as 60% of those at high risk.44 Obvious forms of “classic” barotrauma, causing respiratory collapse or resulting in persistent bronchopleural fistulas are unlikely until peak airway pressures are greater than 50 cm H2O,45 however. Experimental evidence suggests that inflammatory mediators may be produced at ventilatory pressures well below the perceived threshold for obvious barotrauma. Investigators have shown significant differences in inflammatory mediators by using a lung-protective strategy compared to conventional ventilatory management, even though the mean end-inspiratory plateau pressure was still only 31 cm H2O in the control populations.43,46 Such data have led to a general acceptance of “lung-protective ventilatory strategies” for patients at risk of ventilator-induced lung injury.
The inflection points of the inspiratory pressure–volume curves have been proposed as a means of adjusting ventilator settings to minimize ventilator-induced lung injury.47,48 The positions of these inflection points, however, are dynamic and sensitive to physiologic conditions such as edema and chest wall compliance. 47,49 We do not know whether the altered pulmonary compliance from an OP would exacerbate ventilator- induced lung injury. The beneficial results of pleural drainage of fluid in acute respiratory failure suggest that mechanics should be improved. 50 On the other hand, the need for increased ventilatory pressures due to increased pleural pressure may not directly affect the transalveolar pressure, the true effector of lung stretch.40,51 The increased concentration of inspired oxygen (Fio2) that may be administered through the ventilator might facilitate spontaneous resolution of OPs that are observed with a chest tube. It has been estimated that the volume of a simple pneumothorax decreases by 1.25% each day due to gas absorption.52 Administering 100% oxygen may increase this rate of absorption 4- to 6-fold53,54 but conflicts with the principles of reducing oxygen concentrations consistent with lung-protective ventilatory strategies.55 Current guidelines for avoiding ventilator-induced lung injury stress maintaining the Fio2 at the lowest level commensurate with adequate tissue oxygenation and certainly below 60%.41,55 Without further study these issues are speculative.
Conclusions
With respect to OP in mechanically ventilated trauma patients, there are few studies to help answer critical questions. A number of retrospective studies do comment on the incidence of this diagnosis and provide opinions for proper management, but the evidence is essentially based on 2 small prospective randomized trials in the literature, involving only 36 ventilated trauma patients who were actually randomized to the treatment of their OPs. Furthermore, significant failure rates (up to 38%) are associated with the observation of patients subjected to positive-pressure ventilation. Epidemiologic studies should be carried out to determine the incidence of OPs in the typical populations and to elucidate the natural history with and without positive-pressure ventilation. Appropriately powered, and thus presumably multicentred randomized controlled trials, between treatment and observation are needed to guide clinicians.
Footnotes
Competing interests: None declared.
- Accepted July 22, 2003.