Abstract
Background: Cold renal perfusion (CRP) with 4°C crystalloid fluids has been described as a method to reduce renal injury during open surgical repair of complex aortic aneurysms (cAAs) (those requiring at least a suprarenal clamp site). We performed a meta-analysis to ascertain whether CRP improves kidney-related outcomes after open surgical cAA repair.
Methods: Patients of any age or gender who had undergone open surgical repair of cAAs were included. Primary outcomes were the presence of postoperative kidney injury, the need for dialysis and mortality related to kidney injury. We compared patients who were treated with any intraoperative CRP strategy to a control population without CRP. We used a fixed-effects model to analyze derived odds ratios (ORs) and assess heterogeneity. We performed risk of bias analysis to identify potential confounding elements.
Results: Among the 935 studies screened, 5 primary articles met the inclusion criteria. Cold renal perfusion significantly reduced postoperative acute kidney injury (OR 0.46 [95% confidence interval 0.32–0.68], Z = 3.98, p = 0.001). Only 1 study included data for the other primary outcomes. The data were considered homogeneous, with Cochrane Q = 0.23 and I2 of 0%.
Conclusion: This meta-analysis showed reduced postoperative acute kidney injury with the use of CRP during open cAA repair. A prospective randomized controlled trial to perform further subgroup analysis and research the various types of CRP solutions may be warranted to identify further possible benefits.
Patients undergoing major open surgical repair of complex aortic aneurysms (cAAs) are at heightened postoperative risk for acute kidney injury (AKI).1–9 Renal insufficiency has been reported in up to 33.6% of patients in the postoperative period,10 with a rate of new-onset dialysis of 3.3%.11 Previous observational literature in this patient population showed an all-cause mortality rate of 4.2% in the postoperative period,10 which was partly attributed to the high incidence of renal failure.12–14 Cold renal perfusion (CRP) as an adjunct intraoperative technique during open surgical repair has been a favoured approach to improve kidney-related outcomes.15–21 Championed by Crawford and colleagues,22 CRP as a renal-protection strategy has been particularly useful in patients with prolonged aortic clamp time and renal ischemic time.14,23–25 The technique consists of inserting renal artery catheters and perfusing the kidneys with 4°C crystalloid fluid during periods of aortic cross-clamping.16,18
Complex aortic aneurysms are localized, pathological dilations of the abdominal and/or thoracic aorta that may involve the renal or visceral branch vessels. They account for about 15% of all aortic aneurysms.2 We define the subset of cAA as an aneurysm requiring at least suprarenal aortic cross-clamp during open surgical repair, namely, juxtarenal, suprarenal or thoracoabdominal (extent I–V) aortic aneurysms (TAAAs). Despite advances in this era of endovascular surgery, open surgical repair remains an important treatment option of cAA, with excellent long-term outcomes and fewer secondary follow-up procedures.1,10,26 Open surgical repair of cAA is especially indicated in younger, fit patients without major medical comorbidity, patients with connective tissue disease and patients who do not meet anatomic requirements for fenestrated/branched endovascular graft repair.11,26–29
There is a paucity of data supporting the widespread use of CRP during open cAA repair. Currently, there is no established approach or protocol for the use of CRP,16,21,24,30–35 and it is left to the discretion of the individual vascular surgery service or institution to implement its use. Major vascular surgery clinical practice guidelines do not mention or endorse the use of CRP in open aortic surgery.36,37 With regard to open surgical repair of TAAAs, European guidelines state that CRP with cold crystalloid is similar in efficacy to cold blood and may be beneficial, without clear endorsement.37 Furthermore, there are conflicting primary articles reporting on the possible benefits of CRP.2
We performed a meta-analysis to ascertain whether CRP improves kidney-related outcomes after open surgical cAA repair. We hypothesized that the use of CRP for cAA would have a demonstrable benefit toward reducing postoperative AKI and the need for dialysis, and would reduce renal-related mortality. Our objective was to provide support for the ongoing use of CRP during complex aortic repair and spur review boards overseeing clinical practice guidelines to consider adopting a formalized CRP protocol.
Methods
We performed a meta-analysis of articles (prospective or retrospective) that directly compared the use of CRP during aortic cross-clamping to no renal perfusion strategy. The meta-analysis was performed in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) protocol. In keeping with PRISMA reporting standards, we obtained PROSPERO prospective registration (CRD42020166131).
Eligibility and inclusion/exclusion criteria
Studies that included patients of any age or gender who had undergone open complex aneurysm repair with concomitant use of a CRP strategy, compared with a control group of patients who did not have cold perfusion, were eligible for inclusion. The method and indication of CRP was determined by individual study authors. The use of adjunctive systemic perfusion strategies such as left heart bypass or hypothermic circulatory arrest, or methods of renal perfusion/cooling other than CRP were excluded, as was the use of CRP with cold blood. Urgent and emergent open surgical cases were included; however, hybrid or pure endovascular operations were excluded. Patients with preoperative dialysis were excluded. Articles published in English on any calendar date were included. In the case of studies without clear methods of the CRP protocol or patient data that were unavailable for extraction, we contacted the study authors directly for further information. Articles without further clarification were excluded.
Search strategy and study screening
Search terms for this study were developed with the aid of an expert study librarian and were reviewed by all study authors (terms are available upon request). A comprehensive database search was generated on Mar. 5, 2020, and confirmed on Oct. 1, 2021, with Ovid Online (OvidUI_ 04.10.01.008, Ovid Technologies). MEDLINE, Embase, the Cochrane Central Register of Controlled Trials and the Cochrane Databases of Systematic Reviews databases were searched. Potential studies for inclusion were compiled with the use of Covidence online (Covidence v2118, Alfred Hospital, Melbourne, Australia), and duplicates were removed.
Titles and abstracts of the imported studies were screened independently by 2 authors (J.G. and A.D.) using the predetermined inclusion and exclusion criteria, followed by a full-text review of all relevant studies by the same 2 authors. Discrepancies at either stage were resolved by consensus between the reviewing authors or by a third reviewer (R.M.) as needed. Forward and backward reference searching was completed on all studies included in the full-text review to identify any additional eligible articles.
Data extraction and outcomes
We extracted the study design, publication date and patient demographic characteristics, as well as primary and secondary outcomes. We defined 3 primary outcomes comparing CRP to the control arm after cAA repair during the postoperative period: presence of postoperative AKI, need for postoperative dialysis, and mortality related to kidney injury/failure. Secondary outcomes comparing CRP to the control group in the postoperative period after cAA repair that were eligible for extraction included length of hospital stay; AKI related to the aortic clamp site; AKI related to elective, urgent or emergent procedure status; and overall procedure cost, as available.
Quality assessment
We used the 2011 Oxford Centre for Evidence-Based Medicine levels of evidence table38 to determine whether studies met level 1 or level 2 evidence, as per our inclusion criteria. Risk of bias was assessed independently by 2 reviewers (J.G. and A.D.) using the Cochrane framework, including the Risk Of Bias In Non-Randomized Studies – of Interventions (ROBINS-I) tool.39 Any discrepancies were resolved by a third reviewer (R.M.).
Statistical analysis
We constructed a dichotomous contingency 2 × 2 event table from review of the provided tables and text for each article selected for data extraction. We used Rev-Man5 software (Version 5.4, The Cochrane Collaboration), Microsoft Excel (Version 16.0) and STATA software (Version 17, StataCorp) for statistical analysis. We calculated odds ratios (ORs) and standard error (SE) of the log OR for each article from extracted 2 × 2 tables. A Mantel–Haenszel method of combining results between studies and a fixed effects model were used to calculate the summary OR and SE of the summary log OR (confidence interval [CI] of 95%). We performed a test for presence of an overall intervention effect (test statistic, Z) and defined statistical significance as p < 0.05. We performed subgroup analysis when the extracted data permitted. We performed relative weighting of each included article and tests for heterogeneity of extracted data. Finally, we calculated the Cochrane Q statistic for Mantel–Haenszel meta-analysis that follows a I2 distribution with k – 1 degrees of freedom, as well as the I2 statistic.
Results
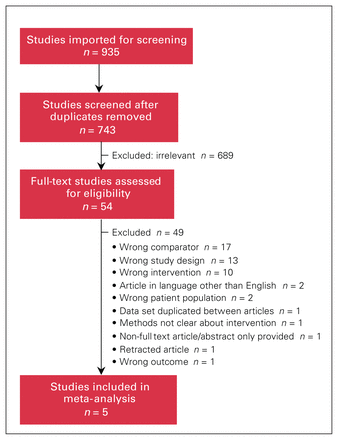
A total of 935 studies were imported through the literature search, yielding 743 studies after removal of duplicates (Figure 1). Abstract and title screening resulted in 54 primary articles for full-text review, of which 49 were excluded, primarily for 3 reasons:
Flow diagram showing study selection.
The article was a feasibility study or case series article (designated as “Wrong study design” in Figure 1). In all excluded articles, every patient received CRP for complex aortic surgery, and feasibility outcome measures of AKI, dialysis use and mortality were discussed. However, as all patients received CRP, no study included a control group of patients who did not receive CRP. Therefore, no comparisons could be made.
The study compared CRP to an alternative renal perfusion strategy that was beyond the scope of the present meta-analysis (designated as “Wrong comparator”). In these studies, outcomes of patients treated with CRP were compared to those of a study group containing patients treated with distal aortic perfusion, usually in the form of left heart bypass. A control group (i.e., a patient group that received no renal perfusion strategy) was not included in these studies.
The authors compared combined CRP and left heart bypass to a control group who received no renal perfusion strategy. As the focus of this meta-analysis was to determine the effect of CRP relative to a control group consisting of no renal perfusion given, these studies (designated as “Wrong intervention”) were excluded to eliminate possible confounding results of left heart bypass on the true effect of CRP.
Five primary articles were included after full-text review for data extraction.14,24,30,31,40 All 5 had a nonrandomized retrospective cohort study design, were from the United States and Canada, and reported on at least 1 of the primary outcomes. O’Donnell and colleagues14 reported a retrospective cohort design from prospectively collected Vascular Quality Initiative data.
Patient demographic and operative characteristics
The publication date of the included articles ranged from 1989 to 2019, with patient data collected from 1975 to 2017 in the US and Canada. The studies involved 1086 patients (mean age 66.7 yr, 371 [34.2%] female) who underwent open surgical repair. The most common comorbidity was hypertension (811 patients [74.7%]), followed by cardiac disease (428 [39.4%]) and diabetes (76 [7.0%]). The majority of patients (890 [82.0%]) had a previous history of smoking tobacco or were current smokers.
The mean aneurysm size was 6.5 cm in maximal transverse aortic diameter (Table 1). Thoracoabdominal aneurysms represented the majority of repairs (882 patients [81.2%]), with fairly equal division among extent I–IV, followed by juxtarenal (133 [12.2%]) and suprarenal (72 [6.6%]) aneurysms. Just over half of patients (569 [52.4%]) had symptoms, including the presence of back or chest pain, dissection or frank rupture. The majority of these aneurysms represented pain with preserved hemodynamic stability and were repaired urgently. Three studies included ruptured aneurysms.24,31,40 Svensson and colleagues31 did not provide numbers for ruptured aneurysms or acute dissection/dissecting aneurysms included in the CRP or control groups. These repairs were likely performed with cardiac bypass for hemostatic control before extensive repair owing to the patient’s hemodynamic instability. Thirty-nine patients requiring emergency repair (3.6% of total patients included in the current study) were included in the studies by Kashyap and colleagues40 and Allen and colleagues.24 The most common surgical approach taken for repair was a retroperitoneal approach (1052 cases [96.9%]), with most patients (954 [87.8%]) having the proximal aortic clamp placed in a supraceliac location. In the 4 studies in which renal ischemic time during aortic cross-clamping was recorded, the mean time was 35.7 minutes. Patients required renal artery reconstruction in 28.1% of cases.
Patients’ demographic and operative characteristics, with relevant inclusion and exclusion criteria
Cold renal perfusion methods
The method and indication for CRP varied among studies. Allen and colleagues24 used a 4°C heparinized (1 unit/mL) saline solution in patients with preoperative renal insufficiency or when the renal ischemic time was expected to be longer than 30 minutes. Anagnostopoulos and colleagues30 infused an iced solution of Ringer lactate containing 1000 units of heparin sodium, 25 g of mannitol, 22 mEq of sodium bicarbonate and 500 mg/L of methylprednisolone, at the surgeon’s discretion. Kashyap and colleagues40 used a 4°C solution of Ringer lactate with 25 g of mannitol and 1 g/L of methylprednisolone, whenever the renal arteries were accessible. Finally, Svensson and colleagues31 used 4°C Ringer lactate, with no indication recorded. O’Donnell and colleagues14 did not report the method or indication for CRP use.
Postoperative renal insufficiency was defined as a normal preoperative creatinine level (< 1.8 mg/dL [159.12 μmol/L]) increasing to an abnormal level (> 1.8 mg/dL [159.12 μmol/L]) or an increase in serum creatinine greater than 20% by Allen and colleagues,24 as an increase in serum creatinine level greater than 50% by Anagnostopoulos and colleagues,30 as a doubling of the serum creatinine level to more than 3.0 mg/dL (265.2 μmol/L) or a requirement of dialysis by Kashyap and colleagues,40 as a serum creatinine level greater than 2 mg/dL (176.8 μmol/L) by Svensson and colleagues,31 and as an increase in serum creatinine level greater than 0.5 mg/dL (44.2 μmol/L) or a new requirement of dialysis by O’Donnell and colleagues.14 O’Donnell and colleagues14 were the only authors to standardize AKI definitions to a standardized schema such as the RIFLE (Risk of renal dysfunction; Injury to the kidney; Failure of kidney function, Loss of kidney function and End-stage kidney disease) criteria41 as determined by Vascular Quality Initiative requirements for publication.
Primary and secondary outcomes
Cold renal perfusion was given intraoperatively in 378 patients (34.8%) (Table 2). Of note, 172 patients (15.8%) were diagnosed with preoperative chronic kidney disease (CKD), which was defined in the 5 studies as a preoperative serum creatinine level greater than 1.5 mg/dL to greater than 2.0 mg/dL (> 133 μmol/L to > 177 μmol/L) or as an estimated glomerular filtration rate of less than 60 mL/min per 1.73 m2. No study distinguished the proportion of patients with preoperative CKD or the outcomes of CRP versus no CRP in this subgroup.
Patients’ baseline characteristics and outcomes
Among the 1086 included patients, irrespective of whether CRP was given, 324 patients (29.8%) developed an AKI, 58 patients (5.3%) required the use of dialysis, and the mortality rate was 6.2% at 30–90 days postoperatively. Only data on the presence of postoperative AKI for the CRP and control groups were available for primary outcome comparison and statistical analysis. Svensson and colleagues31 were the only authors to provide CRP outcome events for dialysis and mortality, and therefore meta-analysis and statistical testing could not be performed for these primary outcomes. We calculated an OR of 0.39 in favour of CRP during cAA for preventing postoperative dialysis use. Cold renal perfusion was not associated with prevention of death, with an OR of 1.1 from this single study; however, this represented all-cause mortality. The mortality signal was highest for the subgroup of patients who required postoperative dialysis, with rates of 18.3%–63.2%.14,31,40 O’Donnell and colleagues14 reported a mortality rate of 6.2% among patients with AKI, a significant AKI hazard ratio of 1.5 (95% CI 1.0–2.1) and a significant dialysis hazard ratio of 5.8 (95% CI 3.2–10.3).
No secondary outcome data were available in any of the included articles.
Effect of cold renal perfusion on postoperative acute kidney injury
We calculated ORs showing the effect of CRP versus control on postoperative AKI for each of the 5 articles. O’Donnell and colleagues14 and Svensson and colleagues31 primarily weighted the analysis, at 22.9% and 51.6%, respectively. The OR ranged from 0.257 to 0.870, with a summary OR of 0.46 (95% CI 0.32–0.68) in favour of CRP in reducing postoperative AKI (Figure 2). A Cochrane Q statistic of 0.23 and I2 statistic of heterogeneity were calculated as 0% (χ2 = 2.37, p = 0.7), indicating homogeneity of the included studies.
Forest plot showing the effect of cold renal perfusion (CRP) versus control on postoperative acute kidney injury (AKI) events from each of the 5 included primary articles and their respective weighting on the overall summary odds ratio (OR). CI = confidence interval.
Study quality
The ROBINS-I tool showed that each of the 5 studies had at least 1 domain that was at risk of bias, mainly within the domain of intervention deviation (Table 3). The study by Kashyap and colleagues40 was specifically of low quality for not reporting outcomes for 38% of patient outcomes; however, because their study contributed only 65 patients to the meta-analysis, this accounted for a 12.4% weighting in the summary OR. Nonetheless, all studies were deemed of satisfactory quality for inclusion.
ROBINS-I tool and scoring of bias within each domain*
Discussion
In this meta-analysis, we found that CRP, and not a clamp- and-sew method, was an intraoperative strategy to mitigate postoperative AKI in open surgical repair of cAAs. This result is important to all vascular surgeons in North America, as the repair of juxtarenal and suprarenal aneurysms (which require a suprarenal aortic cross-clamp for repair) would benefit from the routine use of CRP.
In support of our initial hypothesis, we found a significant OR (0.46, 95% CI 0.32–0.68) in favour of CRP as a method of reducing postoperative AKI events after open surgical repair of cAAs. Standardized heterogeneity calculations showed low I2 and Cochrane Q score values, indicating homogeneous patient pooling among the included articles. Notably, 4 of the investigator groups concluded that the use of CRP for open cAA repair showed a benefit of reducing AKI events,14,24,30,40 whereas Svensson and colleagues31 reported unclear benefit. This likely reflected a sufficient pooling effect and appropriate effect size when the extracted primary data were combined for outcome assessment.
The study by Svensson and colleagues,31 which had the majority weighting in this meta-analysis, incorporated 3 separate renal perfusion protection strategies (CRP alone, left heart bypass alone and left heart bypass with CRP), as well as a control group. We extracted data only for the CRP perfusion group and the control group for calculating the OR for this study, which supported CRP in reducing postoperative AKI. This analysis differed from the statistical testing performed by Svensson and colleagues,31 who used a factorial analysis of variance design that included 2 left heart bypass groups, with or without concomitant use of CRP. The inclusion of these additional left heart bypass groups failed to determine benefit of CRP. In addition, owing to limited data, we could not make conclusions regarding the effect of CRP on postoperative dialysis use or mortality rates, although there was a positive trend favouring CRP (OR 0.39 in preventing postoperative dialysis). The development of acute renal failure after open repair and postoperative dialysis use were linked to elevated mortality rates of up to 63% in our meta-analysis. Further study is required to elucidate the effect of CRP on these outcomes.
The 2 studies that showed the least benefit of CRP had the highest likelihood of CRP’s being used despite the lowest rates of postoperative AKI.24,40 In the study by Kashyap and colleagues,40 78.3% of patients were in the CRP group, and the rate of postoperative AKI among all patients was 11.7%. The overall OR (0.87, 95% CI 0.30–2.55) favoured CRP; the study had a weighting of 12.4% in the meta-analysis. Sixteen (11.3%) of the 141 patients given CRP had a postoperative AKI, compared to 5 (12.8%) of the 39 patients in the control group. These 39 patients were not given CRP because the renal arteries were not accessible. Although CRP did show benefit in this study, the level of dissection around the renal arteries may not have been as extensive in the control group as in the CRP group. In the study by Allen and colleagues,24 58.5% of patients were in the CRP group, and the rate of postoperative AKI among all patients was 12.3%. The overall OR (0.68, 95% CI 0.15–2.98) favoured CRP; the study had a weighting of 6.5%. Four (10.5%) of the 38 patients given CRP had an AKI, compared to 4 (14.8%) of the 27 patients in the control group. The fact that the number of AKI events was the same in the 2 groups was likely related to the small sample. This study had the smallest study population by weighting in our meta-analysis.
Future work on the type of CRP perfusate and additives might reveal differences in AKI outcomes. A study comparing cold crystalloid enriched with histidine–tryptophan–ketoglutarate (HTK) to Ringer lactate solution showed improved CRP efficacy with the former.15 A randomized double-blind trial comparing Custodiol HTK solution to conventional crystalloid solution showed that Custodiol HTK solution was an independent predictor of decreased AKI rates.41 Small trials comparing renal perfusion with blood versus conventional crystalloid CRP showed inferiority of normothermic blood perfusion and similar efficacy of cold Ringer solution and cold blood.17,42 Regarding the optimal temperature of CRP, Svensson and colleagues31 monitored renal temperature during CRP with cooling to a mean of 17.2°C in a subset of patients. Cooling of renal parenchyma was previously found to reduce kidney oxygen consumption to 5%–15% of normal levels and provide about 2 hours of reversible ischemic time in animal models.43–45 Systemic diuretics and other additives for specific renal protection were shown to produce no improvement in renal outcomes in a Cochrane review.46 Systemic mannitol administration intraoperatively may be of benefit in producing favourable renal outcomes; however, current guidelines do not support its routine use.14,37,47,48
Regarding the duration of aortic cross-clamping and CRP, O’Donnell and colleagues14 found in a subgroup analysis that CRP tended to be of benefit in reducing AKI events when clamp duration exceeded 25 minutes. Although this has been replicated in other studies,23–25 a preoperative method for determining an expected clamp duration has not been formally devised. Technical factors during complex open surgical repair determine renal outcomes. Dubois and colleagues48 found significantly more postoperative AKI events in patients who underwent extra renal artery procedures, had frequent division of the left renal vein for aneurysm exposure, or had prolonged aortic clamp time. Although renal artery reconstruction increased the total cross-clamp time in the study population included in our meta-analysis, CRP was the most helpful in this subgroup in some of the included studies. For example, in the study by Anagnostopolous and colleagues,30 83% of the CRP group required renal artery reconstruction. The use of CRP in these patients, who were at high risk for postoperative renal dysfunction, negated the effects of prolonged renal ischemic/cross-clamp times. Our meta-analysis underlines the importance of having an intended plan or protocol to implement CRP in cases in which aortic cross-clamp times are expected to be longer than 25 minutes, renal artery reconstruction is required or the aforementioned technical factors exist. We believe the decision should be made at least before the application of the aortic cross-clamp to institute CRP. Renal ischemic time may also be minimized by ensuring that properly trained ancillary staff are present at critical moments, as well as having all required CRP equipment in a ready-to-use state in the operating room.
Regarding the assessment of bias risk, the included articles were sound for their retrospective cohort study designs. They could not be considered comparable to a well-performed randomized controlled trial. For the “patient selection” domain of the ROBINS-I tool, 2 studies30,40 enrolled patients at a single centre where CRP was used at the surgeon’s discretion, indicating clear nonrandomization and possible bias risk. Regarding the “intervention deviation” domain, given that CRP represented an intervention of unclear benefit at this time, we assigned a grade of moderate risk of bias. A small study was appraised as being at serious risk of bias with regard to missing data: Anagnostopoulos and colleagues30 reported outcome data for 62% of their study population (6.7% weighting of the current meta-analysis). Overall, each study included in the meta-analysis, despite their risk of biases, was judged to be of adequate methodologic rigour to meet inclusion as per the Oxford Centre for Evidence-Based Medicine Levels of Evidence.38
Limitations
A major limitation was the differing definitions of preoperative CKD and postoperative AKI across the included articles. Standardization of the degree of kidney damage with established criteria such as the Kidney Disease: Improving Global Outcomes guideline for CKD49 and RIFLE criteria for AKI50 would bypass this limitation and allow apt comparisons among studies in the future. None of the authors of studies in our meta-analysis that also included patients with preoperative CKD reported renal outcomes separately, so it could not be determined whether CRP specifically benefitted these patients. Older studies therefore suffered a moderate risk of inadequate methodologic quality owing to these poor definitions of AKI and CKD, as well as arbitrary start and end points. Patients with preoperative CKD represented a small proportion (15.8%) of the overall meta-analysis population. Furthermore, although elevation of the postoperative creatinine level was defined unequally between studies, postoperative AKI was defined in each study with the use of cut-off criteria that applied equally to patients within studies. Although imperfect, there were sufficient definitions of CKD and AKI, as well as standardized application of these terms within studies, to allow ample comparisons and to draw conclusions from our meta-analysis. Anagnostopoulos and colleagues30 did perform detailed univariate analyses of preoperative predictors, including CKD, on the risk of postoperative AKI. Importantly, CKD as a possible independent preoperative risk factor did not correlate with postoperative renal complications. Therefore, the individual study authors decided to include these patients in their primary article. On univariate analysis, preoperative CKD was predictive of postoperative AKI in the study population of Kashyap and colleagues.40 Patients with CKD were equally likely to have been allocated to the CRP group or the control group. The effect of preoperative CKD on the development of postoperative AKI was determined to be mixed homogeneously between groups to a sufficient degree to proceed with analysis, although this limited their overall article. One would expect that the use of CRP would benefit patients with CKD overall. The fact that Allen and colleagues24 had the majority (77%) of patients with preoperative CKD in the CRP group confirms this postulate. Their study had an OR favouring CRP (0.68), even though the CRP group was counterbalanced by a larger number of patients with preoperative CKD. The increased variability of this calculated OR may be attributable to this fact.
Another limitation pertained to the inclusion of articles in which outdated methods for open surgical repair of TAAAs were used. The included articles had patient enrolment dating back to 1975. These studies included thoracoabdominal aneurysms that were traditionally repaired with CRP or a clamp-and-sew method. Modern cardiovascular surgery practices have been updated to CRP with left heart bypass and hypothermic circulatory arrest for distal aortic perfusion for extent I–III and V TAAA.51–55 This may be a confounding factor in the included studies, as, during the initial screening process, we excluded eligible primary articles in which left heart bypass with hypothermic circulatory arrest was used. The subsets of patients with preoperative CKD and the TAAA population, although at clear risk for further renal deterioration after open surgical repair, require further study to fully elucidate the impact of CRP on minimizing renal injury.
A possible limitation from 2 included studies30,40 is centred on the inclusion of patients with urgent or emergent operative status. Addressing urgent repair status for symptomatic aneurysms (although reported in as much as half of the study population) was represented primarily by the presence of characteristic back or chest pain with preserved hemodynamic stability. Overall, these urgent repairs did not contribute to preoperative hypotension and were similar to repairs performed electively, unlike emergency repairs, where hemodynamic instability was observed. Among emergency repairs, only a minority of the meta-analysis patient population was represented by aneurysms with frank rupture or dissecting aneurysms. These patients, however, would have been at increased risk for postoperative renal complications and may have represented a distinct patient population in these 2 studies. Considering this, emergency operative status was specifically analyzed by Kashyap and colleagues40 and Anagnostopoulos and colleagues.30 Although Kashyap and colleagues40 did include patients with pathological conditions, including ruptured aneurysms and dissecting aneurysms that were repaired emergently, they performed a univariate analysis specifically of possible associated factors contributing to postoperative AKI, renal failure and death; this analysis did not implicate emergency repair status in their statistical analysis. Likewise, Anagnostopoulos and colleagues30 included patients with ruptured aneurysms with emergency repair status; these emergent repairs represented 13% of their patients. A similar univariate analysis of possible preoperative predictors of postoperative renal dysfunction, including ruptured/emergency status, likewise did not show any significant association. Those authors postulated that these patients self-selected to remain hemodynamically stable to undergo repair and were worthy of inclusion in their primary article. As emergency repair status was not found to be significantly associated with renal outcomes in multiple detailed analyses, and a minority of patients (39, representing 3.6% of the total meta-analysis population) required emergency repair, we elected to proceed with the meta-analysis.
Conclusion
We found that CRP was an effective intraoperative strategy to mitigate postoperative AKI in open surgical repair of cAAs. This finding provides evidence that CRP should be viewed as an adjunctive intraoperative technique to be incorporated in future guidelines for safe aortic surgery. Given the gaps in the literature regarding other renal outcomes (dialysis use and renal-related mortality) and patient subgroups at high risk for postoperative renal dysfunction (i.e., those with preoperative CKD and TAAA subgroups), and different types of CRP solutions, a prospective randomized controlled trial may be warranted. As only 1 of 3 primary outcomes and no secondary outcomes could be assessed in the available literature, a randomized controlled trial would further clarify the role of CRP to a broader extent and context. Identifying patients at high risk for postoperative renal failure should be a priority to improve patient outcomes. Further study is necessary to characterize underlying factors such as the presence of CKD, extensive aneurysm burden and prolonged clamp times in order to create a formalized CRP protocol and recommendations for CRP in clinical practice guidelines.
Footnotes
Competing interests: None declared.
Presented at the virtual Canadian Society for Vascular Surgery 43rd Annual Meeting on Vascular Surgery, Sept. 24–25, 2021
Contributors: A. Devrome designed the study. J. Grab and A. Devrome acquired the data, which J. Grab, H. Krzyzaniak and R. Moore analyzed. J. Grab and H. Krzyzaniak wrote the manuscript, which A. Devrome and R. Moore critically revised. All authors gave final approval of the article to be published.
Funding: This work was supported by the Division of Vascular Surgery, Department of Surgery, University of Calgary, Alberta Health Services.
- Accepted February 24, 2022.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use) and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/