Abstract
Background: Although day surgery (DS) total hip arthroplasty (THA) has good patient satisfaction and a good safety profile, accurate episode-of-care cost (EOCC) calculations for this procedure compared to standard same-day admission (SDA) THA are not well known. We determined the EOCCs for patients who underwent THA, comparing DS and SDA pathways.
Methods: We evaluated the EOCCs for consecutive patients who underwent DS or SDA THA for osteoarthritis or osteonecrosis performed by a single surgeon at 1 academic centre from July 2018 to January 2020. Patient demographic and clinical data were recorded, as were preoperative diagnosis, type of anesthesia, type of implant used, surgical time and estimated blood loss. We determined direct and indirect costs from time of arrival at the presurgical unit to hospital discharge. We determined the EOCCs using an ABC method.
Results: The study included 50 patients who underwent THA (25 DS, 25 SDA). The mean length of stay in the SDA group was 45.1 (standard deviation [SD] 21.4) hours. Differences were observed between the 2 groups in mean age, mean Charlson Comorbidity Index score, surgical technique and mean surgical time (p ≤ 0.001). The mean total EOCC for SDA THA was $10 911 (SD $706.12, range $9944.07–$12 871.95), compared to $9672 (SD $546.55, range $8838.30–$11 058.07) for DS THA, a difference of 11.4%, mostly attributable to hospital resources such as laboratory tests, radiologic studies and cost of the surgical admission.
Conclusion: Day surgery THA is cost-effective in selected patient populations. With the savings identified in this study, every 10 additional DS THA procedures would save sufficient resources to perform an additional THA operation.
Patients with end-stage hip osteoarthritis are commonly treated with total hip arthroplasty (THA). This operation has become one of the most successful and common orthopedic procedures, restoring function to more than 65 000 Canadians each year.1 In recent years, there has been a shift toward outpatient surgery and shorter hospital stays for patients undergoing THA in order to accelerate patient recovery, but also to reduce health care costs.2 A multimodal, interdisciplinary approach optimizing patient selection, preoperative planning, patient counselling, improved surgical techniques involving minimally invasive approaches, optimized perioperative analgesia and early postoperative mobilization has allowed day surgery (DS) THA to be done safely and with high patient satisfaction.3–9 In Canada, the average length of hospital stay for THA is 2.6 days.10 Four times as many joint replacement procedures were done as outpatient surgery in 2020–2021 compared to 2019–2020.10
On average, in 2019–2020 in Canada, inpatient hip replacement surgery had a mean estimated cost of $9591 (estimated with a case-mix group methodology and including inpatient physician costs and excluding rehabilitation), for a total more than $600 million spent annually on this procedure.10 Although DS has good patient satisfaction, the cost benefit of this procedure compared to standard same-day admission (SDA) is not well known. Despite the large impact of episode-of-care costs (EOCCs) for hospital stays on hospital budgets, accurate calculation of these costs remains a challenge in Canada. High-performing universal health care systems, such as those in Australia, France, Germany, the Netherlands, Sweden, Switzerland and the United Kingdom, use activity-based funding to remunerate hospitals.11,12 Current methodologies involve top-to-bottom approximation of costs through grouping systems and traditional accounting. Canadian provinces are also gradually transitioning toward more precise and tailored funding methods involving diagnostic-related groups and case-mix methodologies.13
To better describe EOCCs, activity-based costing (ABC) examines comprehensively all elements of a medical act to estimate costs and subsequent funding. The ABC methodology varies from traditional costing schemes in that calculations based on weighted resource use are utilized, rather than a top–down approximation system.12 Those methodologies advance the understanding of the economic burden of different medical conditions and the source of cost variability through elaborated clinical and administrative data analysis.11,14–16
Despite the emergence of ABC models, few investigators have successfully evaluated the EOCCs of orthopedic procedures such as THA.17–20 Considering the increasing number of prosthetic joints and the substantial economic impact of THA, it is important to understand its EOCCs to provide policy-makers with the information necessary to develop optimal financing methods based on individual patient complexity, which is paramount for the economic sustainability of our health care system.
The objective of this study was to determine the EOCCs using ABC methodology for patients who underwent THA, comparing DS and SDA (with a 2-d hospital stay) pathways.
Methods
This was a retrospective cohort study of consecutive patients treated surgically with THA (cementless arthroplasty or hybrid THA) for osteoarthritis or osteonecrosis in an academic centre. All procedures were performed by a single surgeon (M.T., a fellowship-trained arthroplasty surgeon) at 1 institution from July 2018 to January 2020. All patients requiring simple, primary THA without associated bone graft or osteotomy were included. Patients older than 80 years were excluded, as were those with severe cardiovascular or pulmonary disease, liver cirrhosis, renal failure, type 1 diabetes or narcotic dependence, and those requiring revision arthroplasty, since they were not eligible for our institutional DS enhanced recovery pathway. The study was approved by our hospital ethics board.
Demographic characteristics
Patient demographic data, including age, gender, comorbidities, body mass index, American Society of Anesthesiologists classification and Charlson Comorbidity Index score, were recorded. The patients’ charts were reviewed retrospectively to identify the preoperative diagnosis, type of anesthesia, type of implant used, surgical time and estimated blood loss. We defined postoperative length of stay as the number of days each patient spent on the wards after the surgical intervention.
Surgery and postoperative protocol
All patients had surgery under epidural or spinal anesthesia and received sedation based on their level of anxiety. Normothermia was maintained with a heated blanket. In all cases, a highly porous coated cementless acetabular implant with a highly cross-linked polyethylene liner was used. The choice of femoral implant was based on the patient’s femoral morphology and bone quality.
All patients had portable anteroposterior pelvis radiography in the postanesthesia care unit (PACU) to ensure that the hip was reduced and there were no fractures. In addition, patients in the SDA group had anteroposterior and cross-table lateral hip radiography the day after surgery, before discharge. No patients had any routine blood tests in the PACU. Patients in the SDA group routinely had a complete blood count and electrolyte analysis performed on the day after surgery.
A perioperative pathway protocol21 was used in the DS group. The preoperative medications for both groups included orally administered acetaminophen, celecoxib, ondansetron and tranexamic acid.21 In addition, the patients on the DS pathway received controlled-release oxycodone and pregabalin and a scopolamine patch 1–2 hours before surgery. Patients in both groups received 2 g of cefazolin within 60 minutes before the incision was made. Two hours after surgery, patients were given 2 g of tranexamic acid orally. Postoperative instructions included weight bearing as tolerated, diet as tolerated, restricted adduction and posterior hip precautions. Four to 6 hours after surgery, patients were evaluated for discharge with the following criteria: must eat a meal before discharge, normotensive after orthostatic challenge, pain managed adequately, distal neurovascular status intact and clearance by a physiotherapist. All patients in the DS group were seen by a physiotherapist in the PACU 2–4 hours after surgery. Patients in the SDA group did not receive any physiotherapy until the next day, on the ward.
Patients in both groups received a second dose of 2 g of cefazolin 8 hours after the preoperative dose. Patients were discharged home with a prescription for acetaminophen, 650–1000 mg every 6 hours for 14 days, hydromorphone, 0.5–1.0 mg orally every 4–6 hours as needed, celecoxib, 100 mg orally twice a day for 10 days, and rivaroxaban, 10 mg orally once a day for 5 days followed by acetylsalicylic acid, 81 mg orally for 30 days.
Costing data
We determined direct and indirect costs from time of arrival at the presurgical unit to hospital discharge. Data were extracted with the PowerPerformance Manager (PPM) system (PowerHealth), a centralized database that is secured and managed by an institutional data warehouse. The PPM system enables prompt extraction of data including itemized direct and indirect costs per episode of care, procedure codes, adverse events and precise temporal data. The demographic characteristics were extracted manually from the institution electronic medical record system, OACIS (Telus Health).
The episode of care consisted of the surgical episode from arrival at the preoperative unit to discharge from the PACU for patients in the DS group and to discharge from the surgical ward for patients in the SDA group. Costs for the 6-week postoperative visit were also included. Itemized costs were extracted for each patient’s perioperative EOCCs with the PPM software. These operational costs were classified as direct (clinician labour, equipment, implants, consumables and laboratory testing) or indirect (management, information technology, billing, logistics, sterilization, maintenance and human resources) (Box 1). The arthroplasty implants included the acetabular component, 2 acetabular screws, cross-linked polyethylene liner, femoral stem and ceramic femoral head. We calculated the costs associated with these consumables based on direct purchase price. All items, from the surgical gowns in the operating room (OR) to the cost of individual blood tests on the wards, were extracted, coded and linked to an encounter. All fixed and periodic costs of support resources necessary to supply a service or a procedure were included. Allocation of the indirect costing inputs to each surgical encounter followed an ABC framework as provided by governmental authorities. Areas of care received indirect costs following equations allocating the amount based on time spent on each activity. If time was not applicable (e.g., with consumables), we distributed indirect costs following ratios based out of use. We excluded physician remuneration from direct costs because these fees are borne by the provincial government and not the hospital.22
Direct and indirect costs for activity-based costing
Direct costing areas
Operating room
Postanesthesia care unit (recovery room)
Surgical unit
Laboratory testing
Supply, implants and consumables
Pharmacy
Imaging
Professional (occupational therapy/physiotherapy)
Durable medical equipment
Transport
Emergency department
Intensive care unit
Indirect costing areas
Billing
Information technology
Central sterile processing
Maintenance
Human resources
Hospital administration
Nonclinical salaries
Statistical analysis
The data were stored and analyzed via Prism version 9 software (GraphPad). We used unpaired Student t tests to compare outcomes between the DS and SDA groups. Statistical significance was set at p < 0.05 and confidence interval at 95%. Continuous variables were presented as mean and standard deviation (SD).
Results
The study included 50 patients treated surgically with THA, 25 in the DS group and 25 in the SDA group. Cementless arthroplasty was done in 44 patients (88%), and 6 patients (12%) had hybrid THA. Differences were observed in mean age, mean Charlson Comorbidity Index score, surgical technique and mean surgical time between the DS and SDA groups (Table 1). The mean age was 71.1 (SD 10.0) years in the SDA group and 60.6 (SD 10.4) years in the DS group (p < 0.001). The mean Charlson Comorbidity Index score was 3.2 (SD 1.3) in the SDA group and 2.0 (SD 2.0) in the DS group (p = 0.001). Six patients (24%) in the SDA group had a cemented prosthesis, compared to no patients in the DS group. The mean surgical time was greater in the SDA group than in the DS group (113.0 [SD 24.5] min v. 88.2 [SD 13.5] min) (p < 0.001). The mean length of stay in the SDA group was 45.1 (SD 21.4) hours.
Demographic and clinical characteristics of patients who underwent total hip arthroplasty with the same-day admission or day surgery pathway
Episode-of-care costs
The mean total EOCC for SDA THA was $10 911.12 (SD $706.12) (range $9944.07–$12 871.95), compared to $9672.09 (SD $546.55) (range $8838.30–$11 058.07) for DS THA, a difference of –$1239.03 (11.4%) (p < 0.001) (Table 2).
Episode-of-care costs
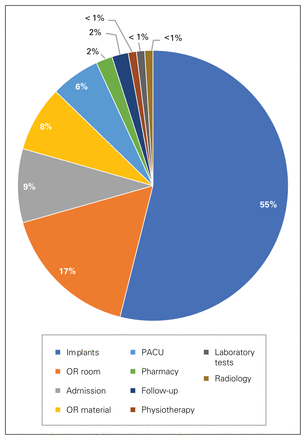
The mean cost of implants was not significantly different between the DS and SDA groups ($6011.38 [SD $343.76], range $5186.77–$7015.98 v. $6047.67 [SD $481.09], range $5128.62–$7000.81) (p = 0.8) (Table 2). The mean OR operational cost for the DS group was $1632.30 (SD $158.55) (range $1414.29–$1958.25), compared to $1860.34 (SD $358.67) (range $1396.49–$3058.25) for the SDA group (p = 0.006). There was no significant difference in the mean per-patient PACU cost between the DS and SDA groups ($712.99 [SD $297.23], range $135.42–$1776.93 v. $615.11 [SD $211.13], range $135.42–$1167.53) (p = 0.2). The additional routine postoperative blood tests and the radiographs in the SDA group resulted in an extra cost of $61.53 and $27.85 per patient, respectively (Figure 1). The costs for postoperative physiotherapy, OR materials and the 6-week follow-up visit were identical for the 2 groups. All patients in the SDA group were discharged before or at postoperative day 2, and the cost of admission to the ward averaged $946.12 (SD $426.61) (range $546.10–$2366.88), 10% more than the cost to treat the patients in the DS group.
Same-day admission episode-of-care costs. OR = operating room; PACU = postanesthesia care unit.
Discussion
This study provides an in-depth analysis of the EOCCs associated with THA protocols in a Canadian academic centre. We used an ABC methodology to compare the 6-week cost of DS versus SDA THA. This methodology provides a precise analysis of both direct and indirect costs. In this study, the EOCC of DS THA was on average $1239, or 11% less expensive than that of SDA THA.
There are multiple factors that explain the difference in cost between the DS and SDA THA protocols, but the cost of the hospital admission was the main driver of the increased cost of SDA THA. In addition to the mean daily fee of $946 of the surgical nursing unit, an admission was associated with higher costs for laboratory tests and additional radiologic studies (mean $89.30). Our DS protocol does not include any postoperative laboratory tests or additional radiographic imaging of the hip. Although these additional tests are commonly ordered as part of a routine admission protocol, it may be cost effective to consider individualizing this practice and testing only patients with suspected complications or underlying medical conditions that warrant close in-hospital follow-up.23,24 It is currently unknown what proportion of THA procedures in Canada are done as day operations. Assuming that 65 000 THA procedures are done every year, if 50% of those are done as DS procedures as opposed to all SDA procedures, this would allow savings of more than $40 million.
Despite the higher comorbidity in the SDA group, there was no significant difference in pharmacy costs between the 2 groups. We calculated the OR cost based on the time the room was used and the personnel present before, during and after the operation. One of the reasons explaining the increased OR costs in the SDA group is the difference in demographic characteristics between the 2 groups and the difference in implant fixation. It has been shown that patients with a higher Charlson Comorbidity Index score incur higher costs for THA.25,26 Patients selected for SDA were significantly older than those in the DS group and had a higher mean Charlson Comorbidity Index score, which can be associated with a more complex approach and a longer surgical procedure, and, therefore, a more expensive procedure. As well, 6 patients in the SDA group had a cemented femoral stem, compared to no patient in the DS group. Cementing and implanting a femoral stem takes more time than implanting a cementless stem, thereby contributing to the increased surgical time and, consequently, the increased OR costs in the SDA group. Although direct implant costs were not significantly different between the 2 groups, using the same implants (either all hybrid or uncemented) would tend to normalize surgical time between the groups and, by the same effect, OR cost. However, even when the implant and the other costs were equalized, admission to the ward on its own resulted in an increase of 10% in the cost of THA. The inherent differences between patients in the DS and SDA groups resulted in different medical comorbidity and implant profiles between the 2 groups; however, our cost analysis captures these differences. The surgeon and anesthetist fees for the surgery (about $1000 and $500, respectively) were not included in our cost analysis.
Other investigators have looked at the potential cost savings associated with DS THA. Venditolli and colleagues21 reported a reduction in hospital costs of $1489 with DS compared to SDA THA, but they did not include the implant costs. Petis and colleagues27 found that the cost in Ontario of a 2-day hospital stay after THA was $2727, compared to $1239 for a 1-day stay. Using an ABC methodology, DiGioia and colleagues28 estimated a mean cost of US$11 319–US$12 319 for SDA THA using an implant costing US$4000–US$5000. With a similar implant cost, and including the estimated surgeon and anesthetist fees, our estimated cost for SDA THA would be $12 411 (US$9814). Other studies have shown a reduction in total costs with outpatient THA in the United States of 16%–21%,29,30 compared to the 11% reduction in the present study.
Limitations
There are limitations of this study that can be addressed in future research. The small sample was susceptible to sampling bias and reduced the statistical power of the analysis. The patients in the SDA group were older than those in the DS group, with more comorbidities, which affected the surgical technique and choice of implant. However, not all patients are eligible for DS THA owing to their medical conditions. This study took this into account by recognizing the need to admit some patients undergoing THA, and the need to use a cemented femoral stem and perform postoperative blood tests in many of these patients. When we excluded the implants and tests, the cost of SDA THA was still 10% higher than that of DS THA. In addition, the enrolment of patients from a single hospital and single surgeon limits the generalizability of the results. However, institution-specific data should be encouraged to promote internal quality-improvement initiatives and interinstitution comparisons. Finally, the retrospective nature of the study may also be a potential source of selection bias in the composition of this cohort, although temporality was not an issue, given the nature of the primary outcome.
Conclusion
The mean total cost of SDA THA was $10 911, compared to $9672 for DS THA, a difference of 11%, mostly attributable to hospital resources such as laboratory tests, radiologic studies and cost of the surgical admission. With the savings identified in this study, every 10 additional DS THA procedures would save sufficient resources to perform an additional THA operation. The DS pathway was associated with superior postoperative functional outcomes compared to the SDA pathway. This value-based health care study provides evidence that DS THA is cost-effective when used with evidence-based perioperative interdisciplinary care pathways.
Acknowledgement
The authors thank the McGill University Health Centre cost analysts and Data Warehouse team for providing the data.
Footnotes
Competing interests: Mitchell Bernstein reports consulting fees from Smith & Nephew, Orthofix, Restor3D and NuVasive. He sits on the Executive Board of the Limb Lengthening and Reconstruction Society. No other competing interests were declared.
Contributors: M. Bernstein, J. Montreuil and A. Hart designed the study. P. Moisan and M. Tanzer analyzed the data. P. Moisan and J. Montreuil wrote the manuscript, which M. Bernstein, A. Hart and M. Tanzer critically revised. All authors gave final approval of the article to be published.
- Accepted June 13, 2022.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/