Abstract
Traumatic injury remains the leading cause of potentially preventable death in Canadians under age 40 years. Although only a minority of patients present with hemodynamic instability, these patients have a significant chance of dying. The causes of instability must be recognized and corrected quickly by using a systematic approach. To allow key supportive interventions to be undertaken swiftly, it is more important to identify and prioritize systemic compromise than to confirm specific diagnoses. Most potentially preventable trauma death relates to airway obstruction, hemopneumothorax, intracranial hemorrhage and intracavitary bleeding. Definitive airway control should be assured as a first priority. Hemopneumothoraces are typically addressed by chest tube insertion, although thoracic exploration will occasionally be urgently required. Hemorrhage control is much more important than fluid resuscitation and mandates the earliest possible definitive management. Unstable patients nearing physiological exhaustion require abbreviated or “damage-control” surgical tactics. This should be recognized early in the resuscitation rather than late in an operative procedure. The management of expanding intracranial hemorrhage requires optimization of oxygenation, ventilation and circulatory support while urgent CT and expert neurosurgical care are provided. Polytrauma presenting with head injury challenges the most developed of trauma systems, necessitating thoughtful prioritization of care and taking into consideration local capabilities. Bedside trauma sonography is an evolving tool that complements the physical examination during an initial survey. Future breakthroughs in trauma resuscitation may involve procoagulant medications, imaging technology, circulatory assist techniques and the use of inflammatory modulators. The greatest future challenge in trauma care, though, will be the provision of basic organized resuscitative care to the global community.
Traumatic death remains a major public health issue. Injury occurs unexpectedly and often affects the most productive segment of society. In the developed countries, injury constitutes the leading causes of death among people aged 15–44 years and is increasingly a leading cause of death in the low- and middle-income countries, where death due to infection is also high.1 Overall, it is estimated that 16% of the world’s burden of disease can be attributed to injury.1 The concept of the “golden hour” is now more than 2 decades old, yet most preventable trauma deaths still occur early in hospitalization. Studies from the last decade from North America, Europe and Australia reveal that up to 48% of traumatic deaths are potentially preventable.2–4 Many of these fatalities have been observed to occur early in the hospitalization5,6 and involve management of the airway,6,7 thoracic injuries2,6 and control of shock or hemorrhage, or both.2,5,8
Organized trauma systems based on designated centres with strong multidisciplinary collaboration are critical to maximize the benefit of specific interventions.9 All critically ill patients should ideally be transported to a dedicated trauma centre providing the highest level of care available.10,11 With the geographic realities of Canada, this requires an organized and inclusive trauma system. Each facility that may receive such patients needs to have prearranged response teams, organized resuscitative equipment and transfer agreements to higher levels of care if appropriate. While recognizing the importance of the system, this manuscript focuses on the bedside resuscitation of the individual injured patient with unstable vital signs. Further, because trauma is a surgical disease, the capacity to perform invasive surgical procedures is assumed, as such care is necessary to save the most critically injured.
What is an unstable trauma patient? An overview of the pathophysiology of critical injury
The subset of patients presenting with deranged vital signs has a high likelihood of requiring operative interventions for severe chest and abdominal injuries and an increased risk of dying.12,13 The terms unstable and stable are frequently used in critical care, yet they are often not defined well. In this review, “unstable” refers to the severely injured patient who manifests abnormal vital signs during resuscitation and is at high risk of depleting physiological reserves. The clinician should remain aware, though, that many “stable” patients have serious, life-threatening injuries and are also at a high risk of dying. Nonetheless, because this terminology is ubiquitous in practice, we will consider the unstable patient to be one who should be attended to immediately to prevent a catastrophic outcome.
With the exception of head injuries, inadequate oxygen delivery is the final common pathway that results in death. Hypoxemia secondary to airway obstruction and inadequate ventilation can be fatal within minutes. Shock is a state of oxygen delivery insufficient to sustain normal tissue and cellular function.14 Regardless of its etiology, it represents an advanced derangement of cellular and organ bioenergetics. Although it is not the only cause of posttraumatic shock, hemorrhage is the most prevalent. Hemorrhagic shock marked by hypotension is rarely seen in patients with less than 30% of a blood volume deficit and reflects an advanced state of physiological exhaustion.15 Thus, from a practical bedside standpoint, all trauma patients with overt or occult shock are presumed to be suffering from hemorrhagic shock until proven otherwise, although it must be recognized that this assumption in some cases leads to excessive volume resuscitation, the consequence of which remains an important unanswered question. There are other causes for shock that must also be considered, especially if the patient does not respond to initial measures. Obstructive shock is related to mechanical impediment of the venous return to the heart that prevents cardiac filling. Etiologies include tension pneumothoraces and cardiac tamponade. Neurogenic shock results from the loss of vasomotor tone to peripheral vascular beds and is almost always associated with acute spinal cord injury. 16,17 Cardiogenic shock is a direct result of circulatory pump failure, a condition that may antedate (especially as our population ages) or follow the injury. Septic shock is unusual unless the patient presents late, as might be seen in a developing-country or operational setting. Rarely, endocrinologic emergencies such as adrenal insufficiency, diabetic ketoacidosis or myxedema coma might be associated with traumatic injury.18 Finally, “traumatic shock” has been conceptualized as an independent postinflammatory state secondary to immune activation14 superimposed on traumatic injury, although this phenomenon is likely indistinguishable from organ dysfunction that often develops after resuscitation from severe injury or illness.19 A recent review reported the specific etiology of shock following blunt trauma injury to be 59% hypovolemic, 16% due to isolated head injuries, 13% due to “other” causes (predominantly obstructive), 7% neurogenic and 5% due to injuries too complex to adequately classify.20
Overall approach to the unstable patient: physician education
Arriving at the bedside of an unstable dying patient will be overwhelming if an organized approach to treatment is not adhered to. Given the complexity of decisions that must be made in the crucial initial minutes of assessment, several teaching resources have been created, all of which are highly recommended if not mandated, for frontline clinicians. The Advanced Trauma Life Support Course (ATLS) offers a tested algorithmic approach to the injured patient that conceptualizes the assessment and immediate treatment of physiological abnormalities in an order prioritized by the immediacy of the threat to life. Emphasizing the principles of surgical decision making, operative techniques and damage control, the Definitive Surgical Trauma Care course is a relatively new educational tool, conceived and promulgated by the International Association of Trauma Surgery and Intensive Care in a quest to fill a gap in surgical education, experience and trauma operating skills.21 This course goes beyond the valuable technical lessons of the Advanced Trauma Operative Management course to incorporate the complexities of decision making when one is faced with a multisystem unstable patient.22
Overall approach to the unstable patient: resuscitation and damage control
As ATLS principles provide a framework to approach the patient, the A, B, C, D, E system-based evaluation guides the initial resuscitation. With the unstable patient, though, the clinician must remain particularly aware of local resources and the patient’s physiology. Many advances have resulted in the improved survival of trauma patients. Damage-control surgery is perceived to be one such advance.23,24 In recent years, operative strategy for the most critically injured has evolved to emphasize abbreviated or damage-control strategies to rapidly reverse shock and restore physiological stability, with reconstructive procedures delayed until homeostasis has been restored.25,26 An “abbreviated” laparotomy constitutes hemorrhage control, prevention of enteric spillage and a ready acceptance of planned repeat operations. 25–27 Although the traditional markers indicating damage-control procedures have been hypothermia (temperature < 34°C), acidosis (pH < 7.2 or base deficit ≥ 8 Meq/L) and diffuse coagulopathy, the need for such an approach should be recognized before profound physiological decompensation has occurred.28 This point has been referred to as the ground zero of damage control to emphasize the critical need for preemptive implementation in the prehospital and early resuscitative phases.24 During the initial assessment, key factors should alert caregivers. Although most injured patients seen in daily practice will not require damage control, penetrating trauma with hypotension, high-velocity gunshot or blast injuries, multisystem trauma with thoracoabdominal injury, and limited surgical resources should all readily prompt such a treatment strategy.28,29
The ultrasonography-augmented physical examination
Over the last decade, focused assessment with sonography for trauma (FAST) has become an indispensable tool,30–33 supported by a level I recommendation for use34 that adds another domain to the physical examination. In the unstable patient, the goal of clinician-performed sonography is to quickly identify or confirm obvious pathology so that invasive interventions can be directed with greater accuracy.35–37 With continued training and with the simplification of the technology, ever-expanding indications and techniques are becoming available to examine new organ systems or anatomic regions. It is important that nonexperts maintain a high index of suspicion and not rely on a negative examination as a definitive test.
The prioritized assessment and treatment of the physiologically unstable patient
The unstable patient should be examined systematically, with treatment directed at correcting life-threatening abnormalities as soon as these problems are detected.
Airway
Relieving an obstructed airway is the single greatest priority in any injured person.9,15 An obstructed airway results in inadequate oxygenation and ventilation and causes severe hypoxemia that results in death within minutes. A full review of airway management in trauma is beyond the scope of this manuscript but is discussed concisely in other publications. 9,38 In general, airway patency is expediently assessed by noting the patient’s ability to talk and ventilate spontaneously, in the absence of stridor or accessory muscle use. Any compromise must be dealt with immediately. Even without airway injury, the hemodynamically unstable patient should have early definitive airway control. This helps prevent aspiration, reduces the oxygen debt of breathing, maximizes systemic oxygen delivery, corrects acid base disturbances and allows the clinician to focus on other life-threatening issues.14 In the appropriate patient, a rapid sequence induction can quickly facilitate intubation. However, careful consideration must be given to the patient with a closed head injury and raised intracerebral pressure, coincident airway or cervical spine trauma, contraindications to succinylcholine or anatomic markers of a difficult intubation. It is essential that the intubationist be skilled in airway management and that both an auxiliary plan and personnel are available should the intubation attempt fail. A needle or surgical cricothyroidotomy may be the most efficient backup technique among the array of potential adjunctive airway capture devices available. Spinal precautions must be maintained throughout airway assessment and management because cervical spine injuries are present in 4%–20% of seriously traumatized patients.39 Further, in both cardiac tamponade and tension pneumothoraces, removing the thoracic pump and instituting positive pressure ventilation may further impede venous return and exacerbate cardiovascular collapse.40
After successful intubation, it is crucial that correct endotracheal tube position be determined because emergency intubation doubles the risk of tube malpositions.41 Confirmation via physical examination is limited because 60% of right main stem intubations occur with equal breath sounds documented and 70% despite symmetric chest excursion, and bilateral breath sounds are often heard by anesthetists despite fatal esophageal intubation.42,43 Adequate ventilation should thus be confirmed with the use of capnography.44 Capnography will not detect a right mainstem intubation, however, and carbon dioxide delivery may be absent in cases of circulatory arrest.45 Thoracic sonography is another valuable technology capable of immediately screening the chest to ensure that the left visceral pleura is “sliding” upon the parietal, thus ruling out a right mainstem or esophageal intubation.46,47 Arterial blood gases and a chest radiograph should also be obtained to confirm adequacy of ventilation, oxygenation, perfusion and tube placement.
Breathing: thoracic injuries with ventilatory or oxygenation inadequacy
After securing the airway, the resuscitating clinician must quickly address breathing and respond to life-threatening thoracic injuries. These are responsible for 25% of all trauma deaths.15,48 Rib fractures are the most common serious thoracic injury, and pneumothoraces are the most common intrathoracic injury following blunt trauma.15,48 Although most of these thoracic injuries can be treated by placement of a chest tube,15 the patient in physiological distress may need an emergency thoracotomy for control of massive air leaks or hemorrhage, especially after penetrating injuries.49 Life-threatening thoracic injuries that should be detected during a primary survey include tension pneumothoraces, massive hemothoraces, cardiac tamponade and flail chest.15 Standard examination includes observing for external injuries such as open wounds, leaking air and chest wall deformity. This is followed by palpation for crepitus, deformity and asymmetric respiratory motion. Auscultation is carried out in both the apical and lateral chest areas. We subsequently follow auscultation with a sonographic examination of the same areas in an examination termed the extended FAST exam.50,51
Pneumothoraces are a preventable cause of posttraumatic death. They have a 1 in 5 incidence in victims of major trauma found alive and may cause catastrophic cardiopulmonary derangements when compared with other thoracic injuries of comparable anatomic severity.52 Esposito and colleagues documented a 13% preventable death rate that was predominately related to the management of airway and chest injuries in the emergency department.6 The most concerning type of pneumothorax is the tension pneumothorax; delays in diagnosis and treatment may result in respiratory and circulatory collapse. In the unstable patient, pneumothoraces should be diagnosed clinically by noting diminished breath sounds, hyperresonance to percussion, increased jugular venous pressure and mediastinal shift. When instability is present, treatment should not wait for chest radiographic (CXR) confirmation. In reality, in a patient with coincident hypovolemia and delayed radiographs, diminished breath sounds may be the only appreciable sign in a noisy resuscitation environment. Treatment is ideally needle decompression of the pleural space followed by chest tube insertion. If there is no response to needle decompression, formal finger or tube thoracostomy is required to assure that pneumothoraces are not being missed.53 When there is no time for a CXR, ultrasonography can diagnose and record the presence of pneumothorax and also document the successful evacuation of air from the pleural space.50,54,55
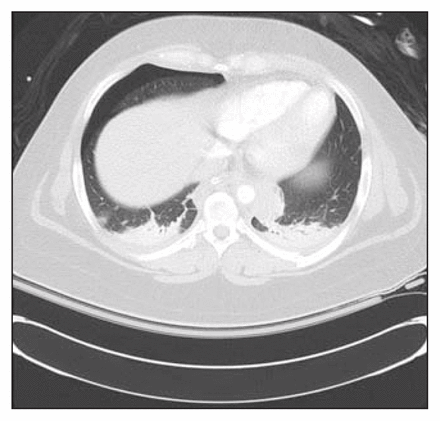
A feature of posttraumatic pneumothoraces that is only now being appreciated as a result of the frequent use of CT is the insensitivity of the anteroposterior supine chest radiograph (AP CXR) to detection of pneumothoraces. 56–58 Remarkably large occult pneumothoraces may be present without an obvious anterolateral pleural stripe on an AP CXR. (Fig. 1).50,56 Despite not being evident on CXR, all pneumothoraces are dynamic and evolve over time, potentially increasing if a flap valve is present. Both the clinical importance and optimal management of these posttraumatic pneumothoraces remain unknown because there has been little prospective study.59,60 Thoracic sonography is a potential method of detecting posttraumatic pneumothoraces during the primary survey, with greater sensitivity for detecting occult posttraumatic pneumothoraces and without the delay of image processing.50,55,57,61 The technique is conceptually simple and focuses on identifying a sonographic sliding movement at the pleural interface. If they are separated by even a small amount of air, these signals are lost. Although we feel this technique has great promise, until it is shown to be generalizeable, the safest approach is to liberally drain the chest.
An occult pneumothorax seen on a CT scan that was not detected on a plain anteroposterior supine chest radiograph.
In addition to ventilatory insufficiency, thoracic trauma can also induce circulatory insufficiency or shock. The most common cause of a massive hemothorax is bleeding from lacerated intercostal arteries, although hemodynamically unstable patients may have injuries to the heart, lungs or great vessels. These may require urgent control in the resuscitative suite. Owing to the pressure sensitivity of the cardiopulmonary system, damage-control techniques involving tight-packing are less applicable in the case of thoracic injuries, although the approach is congruous in the need for faster, simpler definitive repairs that minimize hypoxemia, blood loss and resuscitation.49 Intercostal sources of bleeding often accumulate relatively slowly and may be tamponaded by timely reexpansion of the lung through chest tube insertion. The thoracic site of exsanguination is discussed with the circulatory assessment, although it may be detected and treated by the clinician who is focusing on improving ventilation.
Circulatory instability
When faced with an unstable trauma patient, treatment should begin according to a presumptive diagnosis of hypovolemia even before a specific diagnosis is confirmed. Preventing shock by arresting hemorrhage remains the most important task of the trauma surgeon and the major focus of this review. The physical examination and simple bedside diagnostic adjuncts, notably portable supine CXR, pelvic radiographs and portable ultrasonography, typically suggest the etiology and, most importantly, the potential anatomic site of origin62 (Box 1). The seriously injured patient is dynamic and may illustrate more than 1 cause with different time courses. Thus the clinician must maintain a high index of suspicion and continually reassess the patient.
Bedside assessment of the 5 cardinal sites of massive adult hemorrhage
| Anatomic site | Modality | Reliability |
|---|---|---|
| Intraperitoneal | FAST | Positive test is diagnostic; negative test is suspect |
| DPL | Gross positive is diagnostic; technically positive only is suspect | |
| Retroperitoneal | Pelvic radiograph | Unstable fracture pattern is suggestive |
| Thoracic (pleural) | Chest radiograph | Positive test is usually diagnostic |
| Multiple long-bone fractures | Physical examination | US and radiograph confirmatory but do not quantify blood loss |
| External bleeding | Physical examination | Positive findings diagnosic but do not quantify blood loss |
FAST = focused assessment with sonography for trauma; DPL = diagnostic peritoneal lavage; US = ultrasonography.
Hypovolemic shock
After an expedited assessment of the thorax, attention focuses on the circulatory system, recognizing that circulatory insufficiency may have already become apparent during the chest examination. The patient is again observed and spoken to, and pulses are assessed manually. Worrisome findings include diminished consciousness, pallor, profound tachycardia and weak or imperceptible pulses. Obtaining circulatory stability in unstable patients usually requires identifying an injury after initiating resuscitation; otherwise, the patient is likely to die. It has recently been reported that, during the first 90 minutes in the emergency department, patients who were hypotensive with major abdominal injuries requiring laparotomy sustained an additional 1% increase in mortality for every 3 minutes spent in resuscitation before surgery.63
Fluid resuscitation
Whether to administer fluid to the injured patient, what type to use and how much is controversial. Whether to administer any fluid at all is the initial question. The clinician must balance judicious fluid resuscitation to attempt to maintain a critical level of perfusion to the vital organs with avoiding overresuscitation that might reverse vasoconstriction of injured vessels, dislodge early clot, dilute coagulation factors, cool the patient and induce visceral swelling.64–67 A prospective trial of penetrating torso injuries noted lower mortality in those in whom fluids were restricted until arrival in the operating room,68 although this study has not been replicated.69 These findings and their biological plausibility have advanced hypotensive resuscitation as a strategy for minimizing aggressive fluid resuscitation in situations where expeditious surgical treatment is possible. 70 Further, delaying hemorrhage control and concurrently continuing aggressive fluid resuscitation may induce abdominal compartment syndrome. 71,72–74 High crystalloid/packed red blood cell (PRBC) ratios appear to be particularly prevalent in patients who later develop secondary abdominal compartment syndrome during interventional procedures.72
These decisions are further complicated by controversy as to what actually constitutes hypotension. While a systolic blood pressure of 90 mm Hg has been long considered a marker of hypotension, systolic blood pressures below 110 mm Hg have actually been noted to be associated with a notably increased mortality in a large administrative database. Traditional resuscitative end points including blood pressure and even urine output are less sensitive indicators of tissue oxygen delivery. 69,75–77 Occult shock may persist despite normalization of these indices, resulting in an ongoing oxygen debt and transition to anaerobic metabolism. Animal data demonstrate that the cumulative wholebody oxygen debt accurately predicts mortality in hemorrhagic shock.78,79 Irreversible tissue damage, and even death, may occur despite adequate but delayed resuscitation.80 Thus it should be noted that a systolic blood pressure of 90 mm Hg may represent not just the beginning of circulatory failure; it may be a clinical manifestation of severe physiological decompensation. 81 The presence of a concomitant head injury (see the section below on the severely head-injured unstable patient) and delays to treatment are also often realities of trauma care that will temper enthusiasm for hypotensive resuscitative strategies.
If fluid is to be administered, there is further controversy as to which fluid should be used. Ongoing debates include the merits of crystalloid, compared with colloid, solutions. A controversial Cochrane meta-analysis claimed no benefit, and even a disadvantage, to using colloid fluids in critical care,82 although a recent, well-performed Australian study could not confirm these concerns.83 Newer synthetic colloids have logistical advantages; however, they have not been fully evaluated.84 Although studies of hypertonic saline that were carried out in the past decade failed to demonstrate clear statistical superiority to crystalloids, subgroup analyses demonstrated benefit in severely head-injured and hypotensive patients requiring surgery.85–87 Hypertonic saline also has a newly envisioned role as a potential combined resuscitative and immunomodulating agent.88,89 Hemoglobinbased oxygen carriers (HBOCs) also have great potential, although the first randomized trauma trial in this area showed a worse outcome in the HBOC group.90 Additionally, each particular formulation must be considered as a separate agent when evaluated. Neither hypertonic saline nor HBOCs can be currently considered as proven therapies.
Unfortunately, a sound resolution to these fundamental questions regarding the most appropriate type of resuscitation fluid and preoperative resuscitation goals is presently both unavailable and beyond the scope of this paper. We therefore recommend that standard ATLS principles be employed as subsequently described, with emphasis on the highest priority that localizing and arresting hemorrhage warrants.91 If an obvious source of hemorrhage is detected with a plan to proceed for immediate operative intervention, we initiate intravenous access but do not aggressively resuscitate unless there is clinical evidence of inadequate brain perfusion. Otherwise, standard fluid management remains infusion of 2 L of crystalloid fluid.15 We prefer 1 of these lines to be a large-gauge central catheter in a supradiaphragmatic location, usually the subclavian vein.92 This provides the most direct access to the central circulation and is potentially more accessible to the anesthesia team. The procedure requries an experienced team who can place these lines expediently. If not, 2 large-bore (16-gauge or larger) catheters placed peripherally may be safer.
Practically, fluid infusion can be conceptualized as a test for hemodynamic stability that stratifies patients into management categories. Responders to intravenous fluid challenge generally have no ongoing bleeding and do not typically require emergency operative intervention. Transient responders have initial improvement with subsequent deterioration and will often require intervention but may be further investigated with caution. Nonresponders have persistent shock despite aggressive resuscitation, typically have ongoing high-volume bleeding and require urgent intervention.
Blood should be urgently administered early to those with postinjury shock; however, there should be awareness that it is a complex biological compound associated with immune suppression and multiple organ dysfunction syndrome.93 The current caution to target a lower hemoglobin transfusion threshold in the critically ill94,95 does not apply to the acutely unstable trauma patient. Without time for hemodilution, hematocrits will be normal in acute bleeding. Animal data have suggested that anemia may exacerbate the oxygen debt beyond that induced by the shock state alone in uncontrolled hemorrhage and delayed blood administration.96,97 Although the initial crystalloid volume challenge is important in defining patient status, to continue large-volume infusion in the setting of major hemorrhage sets the stage for hypothermia, acidosis and coagulopathy. 98 Recent experience with massive hemorrhage in both civilian and military settings advocates early resuscitation with generous plasma at a ratio of 1:1 and using PRBCs or even plasma as the primary resuscitation fluid.99–101 The conflict in Southwest Asia has also generated a formal concept of damage-control resuscitation that emphasizes the earliest correction of acidosis and the liberal use of blood products, including plasma and fresh whole blood (when available), to target inherent coagulopathies while keeping the systolic blood pressure at approximately 90 mm Hg.99,100
Localization
To render hemorrhage control effective, the primary source of bleeding must be detected quickly. There are 5 anatomic locations in the adult where enough blood can be lost to result in shock. These are as follows: external; in the soft tissues related to multiple long-bone fractures (a closed fractured femur can account for 2 or 3 units of blood); in the pleural cavities; in the abdominopelvic cavity; intraperitoneal; and retroperitoneal (Box 1).62,102 Unfortunately, patients may have injuries involving 1 or more of these locations. Injured patients should be completely disrobed and externally inspected to detect external bleeding, which is best controlled with direct pressure or, exceptionally, with a tactical tourniquet for short periods of time.103 This is followed by definitive surgical control.104 Fractured long bones should be reduced and splinted as soon as practical to decrease ongoing blood loss, restore distal perfusion and reduce pain, but this should not distract attention when the source of instability is unknown. Massive hemothoraces may be detected clinically by noting reduced air entry on auscultation. They may be distinguished from pneumothoraces through percussion or emergency sonography,15,105 although this distinction is not necessary because an expedient tube thoracostomy will treat both. If not detected clinically, the diagnosis should be made on the AP CXR. Massive hemothoraces may require thoracotomy, determined by the initial volume of blood recovered, hemodynamic consequences and rate of ongoing bleeding. Cell-saving equipment should be used to reduce the need for allogeneic blood transfusion. Although complete exposure of the patient is required for inspection, it is critical to prevent heat loss and hypothermia.106
Resuscitative thoracotomy
Patients with penetrating injury who have lost vital signs for no longer than 15 minutes should undergo resuscitative thoracotomy through a left anterolateral incision.107,108 The goals are to release cardiac tamponade; control intrathoracic bleeding, massive air embolism or air leak; or to increase perfusion to the brain, heart and lungs through descending aortic cross-clamping and open cardiac massage. 49,109 In experienced centres, significant survival rates can be expected with appropriate patient selection. Survival is highest in victims of isolated penetrating cardiac injury who present to the emergency department with signs of life; up to 57% survive. 110,111 By contrast, the utility of resuscitative thoracotomy is greatly reduced in victims of blunt trauma, where only 1%–2% are salvaged.109 Absent signs of life (detectable blood pressure, respiratory effort, cardiac electrical activity or pupillary responses) either at the scene or in the emergency department, absent cardiac activity, and persistent hypotension (systolic blood pressure < 70 mm Hg) have been noted to be markers of a dismal outcome.109 Although many feel the procedure does not have a role in traumatic cardiopulmonary arrest, 108 we have personal experience with survivors of the procedure performed after blunt posttraumatic cardiac arrest. Select indications include patients with electrical cardiac activity who suffer a cardiac arrest within 5 minutes of arrival or who demonstrate a pericardial effusion on FAST examination.107 It is implicit that the procedure be performed by those with experience and imperative that responsive patients have expeditious definitive hemorrhage control rather than a just prolonged resuscitative process.
The distended abdomen
Clinical examination of the abdomen is not a reliable means of diagnosing hemorrhage amenable to surgical intervention. Thus an expedient bedside diagnostic test is required to determine the presence of blood. Although diagnostic peritoneal lavage is performed less commonly in larger trauma centres, it remains a useful test when ultrasonography is unavailable or indeterminate.112 It is in this setting that the FAST examination may be extremely efficient, detecting the presence of intraperitoneal fluid within seconds and thus directing the search for hemorrhage to the peritoneal cavity.32,113 Although CT offers the most accurate diagnostic information after trauma, the unstable patient should never be transported to a CT scanner remote from a resuscitative area. When a large hemoperitoneum is detected, the patient should be transported immediately to an operating theatre and should undergo a laparotomy without delay. If a damage-control approach has been used owing to instability, the abdomen is typically left open to prevent abdominal compartment syndrome.
Retroperitoneal bleeding and severe pelvic fractures
If a pelvic fracture involves the posterior elements or is structurally unstable on examination, without obvious intraperitoneal blood on FAST scan, there is a high likelihood that the source of hemodynamic instability is retroperitoneal bleeding from the pelvis.62 Controlling the pelvic volume with a sheet tied around the pelvis is simple and can be done early for fractures with rotational instability. 114 Angiography is a valuable adjunct in controlling inaccessible bleeding from the solid viscera and inaccessible retroperitoneal bleeding from pelvic fractures. Interventional radiologic techniques have evolved from exceptional second-line procedures to standard adjuncts in damage-control surgery.23,115 We easily envision the near future wherein a standard resuscitative theatre would be capable of supporting both operative and interventional radiologic procedures. It has been traditionally felt that arterial injury accounts for hemodynamic instability in only 10%–20% of patients, with venous bleeding considered most commonly responsible.116 However, it has recently been reported that hemorrhage was predominantly from a pelvic source in hypotensive patients with unstable pelvic fractures, with 59% of these patients having a positive angiogram.117 Closure of the pelvic volume with either anterior external fixation or the use of the posterior pelvic C-clamp may greatly facilitate hemostasis in some fracture patterns.118–120 Some authors also favour pelvic packing as the initial approach to patients in extremis from severe pelvic fractures.116,121 Regardless of which technique is deemed appropriate, experienced orthopedic consultants should be involved early as part of the multidisciplinary approach. 114 Delay of hemorrhage control with ongoing fluid resuscitation should be rigorously avoided because this appears especially associated with the development of abdominal compartment syndrome.72
The clinician must always be suspicious of causes for shock other than hypovolemia. If the patient does not respond to fluid resuscitation and a site of hemorrhage cannot be localized, evaluating this possibility is even more critical.
Circulation: obstructive shock
Pneumothorax
Ideally, a tension pneumothorax should have been identified and alleviated when breathing was assessed. Pneumothoraces may be dynamic, though, developing quickly and unexpectedly, and thus their presence should be re-evaluated frequently, as discussed above.
Cardiac tamponade
Cardiac tamponade is another form of obstructive shock whose clinical presentation can vary from subtle to catastrophic. Classic signs such as tachycardia, muffled heart sounds and an elevated jugular venous pressure should be identified but are easily missed.14 Although penetrating wounds to the precordium are typically obvious, a high index of suspicion is required in blunt trauma where no external injury may be apparent. The FAST examination may quickly identify pericardial fluid, allowing for immediate bedside interventions or transport to an operating room.37,122 Obstructive shock is a special circumstance that presents increased risks following intubation. Both the myocardial depression of anesthetic agents and diminished venous return associated with positive pressure ventilation may precipitate cardiovascular collapse.40 Therapeutic options include subxiphoid window, pericardiotomy through an emergency anterolateral thoracotomy or formal median sternotomy, depending on circumstances and resources.
Blunt cardiac injury and cardiogenic shock
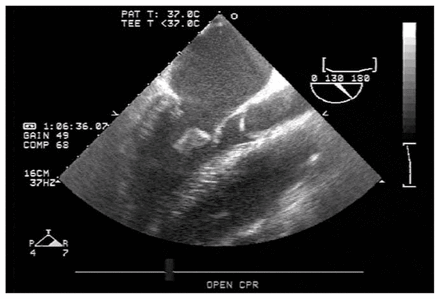
Cardiogenic shock results from inadequate pump function. It may be due to primary cardiac pathology that antedates traumatic injury, or it may be a result of acquired injuries. Blunt cardiac injury refers to a spectrum of injuries ranging from simple electrocardiographic changes to free wall rupture.123 Cardiogenic shock from blunt cardiac injury is uncommon in survivors to hospital, although severe cardiac injuries are common in autopsy series.14 Both the diagnostic criteria and the treatment of these injuries remains highly controversial. Possible etiologies include myocardial, valvular or coronary artery injuries.124,125 Patients with severe blunt cardiac injury usually develop pump dysfunction within the first hours postinjury.14 This presents an exceedingly difficult diagnostic challenge that may only be resolved with formal echocardiography124–126 (Fig. 2). Although detecting intrapericardial fluid is well within the capability of any interested clinician, evaluating cardiac function requires dedicated training and practice. This diagnostic modality would thus ideally be available early in any resuscitation.
Acutely flail mitral valve in a middle-aged man with cardiovascular collapse after blunt chest trauma seen at emergency transesophageal echocardiography.
Vasomotor shock
The term vasomotor shock includes several conditions that are related by abnormalities in the tone of the vasculature. Specific subcategories include neurogenic, septic, induced or pharmacologic and anaphylactic shock.
Neurogenic shock
Neurogenic shock results from the loss of vasomotor tone in the peripheral arterial beds that leads to an increased capacitance, diminished venous return and reduced cardiac output.17,127 Relative bradycardia often accompanies the hypotension, providing a key to diagnosis. Mixed pictures are common, though, and neurogenic shock should be a diagnosis of exclusion. Shock in patients with a spinal cord injury with penetrating trauma was found to be hypovolemic in 74% of patients and neurogenic in only 7%.128 The converse was true after blunt injury, with 70% of the cases being neurogenic.16,128 To treat neurogenic shock, intravenous fluid should be given to increase cardiac preload. Vasoconstrictors are permissible if hypovolemia is excluded and will increase vascular tone, decrease capacitance and potentially minimize secondary ischemia to the penumbra of injury.127
Septic shock
Septic shock is unusual in the acute setting. It might be seen in a third world or military setting where there is a substantial delay to definitive care. When unexpected septic shock is identified early in the hospitalization, missed hollow viscous injury should be considered.
Pharmacologically induced hypotension
The administration of analgesia or sedatives may result in reduced blood pressure through either a primary effect or secondarily by reducing the patient’s own sympathetic tone. Thus, although clinicians should always strive to reduce pain and suffering, the unstable patient should only be administered judicious dosages of short-acting medications, with close monitoring and after the initial injuries have been assessed.113
Anaphylactic shock
Anaphylaxis describes the loss of vasomotor tone following the massive outpouring of vasoactive mediators resulting from the degranulation of mast cells and basophils.129 This is distinctly uncommon in the acute trauma setting but might be suspected if temporally related to drug administration such as antibiotics. The increasing incidence of latex allergies in the Western world may also make this an increasing concern, especially in patients with multiple previous surgical exposures, atopy and food allergies.130,131
Traumatic shock
It is now postulated that trauma itself, even without sepsis, can induce the inflammatory cascade that results in hemodynamic instability.14,19 In this setting, there will be a loss of vasomotor tone for which vasopressors are appropriate. Correction of traumatic shock requires attention to the primary causes and prompt hemorrhage control, repayment of the oxygen debt, débridement of necrotic tissue, stabilization of bony injuries and attention to soft tissue wounds as appropriate.
Double jeopardy: the unstable patient with severe head injury
Associated brain injuries are present in up to 60% of patients with severe blunt trauma,132 which frequently presents conflicts in management. It is anathema to take a hypotensive patient to a CT scanner, yet this is the most important imaging modality to direct neurosurgical care. Following up on the discussion of the rationale for hypotensive resuscitation, hypotension occurs in about one-third of patients with severe traumatic brain injury and is one of the most important predictors of outcome. A single episode of hypotension (systolic blood pressure ≤ 90 mm Hg) occurring during the period between injury and resuscitation increases morbidity and doubles mortality.133 Although it is generally perceived that hypotension related to trauma is not caused by head injury, this has been questioned. Mahoney and colleagues20 reported that isolated head injury was the only identified etiology in 13% of a blunt trauma group with shock. Another study134 reported that 8.5% of patients with severe traumatic brain injury had no discernable extracranial causes for their hypotension. The majority (76%) of these patients received early mannitol or furosemide, which may have induced hypovolemia. Thus it appears that, if brain injury does cause hypotension, it is relatively rare and should be a diagnosis of exclusion.
Given the strong association between hypotension and poor outcome in severe traumatic brain injury, hypotension should be scrupulously avoided. It is recognized that aggressive resuscitation before definitive surgical management may cause hemodilution, disrupt hemostatic clots and worsen blood loss.68 Notably, however, patients with head injuries were excluded from Bickell and colleagues’ oft-cited delayed resuscitation trial.68 Although no prospective trial has been performed, owing to ethical concerns, there is a suggestion from the post hoc analysis of a prospective trial that improvement in blood pressure in hypotensive patients with head injuries improves outcome.87 In addition, concern about the exacerbation of cerebral edema and intracranial hypertension with overaggressive fluid resuscitation has been addressed in study of polytrauma with closed head injury. No correlation was found between intracranial pressure and the amount of fluid or blood infused.135
Mannitol and hypertonic saline have been suggested as small-volume resuscitation fluids in patients with severe traumatic brain injury because both have beneficial effects on intracranial pressure. However, mannitol has been associated with relative hypotension 2 hours after hospital admission, compared with saline, when given in the prehospital setting. 136 Unlike mannitol, hypertonic saline is not associated with an osmotic diuresis and subsequent risk of hypovolemia. Further, the aforementioned post hoc analysis suggested an improved survival for patients with severe traumatic brain injury who received hypertonic saline in the prehospital setting.87 Unfortunately, a recent prospective randomized controlled study failed to corroborate these results.137
Finally, as noted, the threshold for hypotension of systolic blood pressure of 90 mm Hg has not been defined well with a physiological rationale. For patients with intracranial hypertension, maintenance of systolic blood pressure at 90 mm Hg will typically result in a cerebral perfusion pressure of ≤ 50 mm Hg. Therefore, intracranial pathology must be taken into account when identifying initial hemodynamic goals. These goals should be reassessed once intracranial pressure measurement is established. The optimal resolution between these conflicting needs may be best resolved by expediting the assessment and resuscitation as well as by close multidisciplinary cooperation.
Future directions
Future population benefits will likely be accrued from the expansion of organized trauma care to the many people both nationally and internationally who are currently without these services, which constitutes a global challenge for this century.138 Recent advances in patient-specific trauma care, such as the acceptance of damage-control procedures, early trauma portable sonography and the appreciation of abdominal compartment syndromes, can only hope to minimize the “biological first hit” caused by injury.23,98 Even after seemingly successful resuscitation, critically injured patients develop sequential organ failure known as the multiple organ dysfunction syndrome. Future breakthroughs in trauma resuscitation may involve bringing now-distant modalities such as CT scanning and angiography into the resuscitative suite, greater use of procoagulants, increased familiarity with circulatory assist techniques for thermal control and vital organ support and, most importantly, inflammatory modulators targeting the posttraumatic proinflammatory cascade.23,98,139,140 Recombinant Factor VIIa acts specifically at the local site of injury where tissue factor and phospholipids are exposed, leading to a millionfold amplification of localized coagulation. 141–144 Remarkable but anecdotal cases of rescue from refractory coagulopathic bleeding when all other measures failed have been reported.145–148 This requires confirmation with prospectively randomized, multi-institutional trauma trials. In future, if wounds are beyond current resuscitative capabilities, heparin-bonded extracorporeal circulatory pumps offer potential heroic last-ditch options to either ameliorate end-stage shock to maintain perfusion of vital organs or to accept vascular collapse and induce temporary suspended animation.149 Suspended animation preserves the viability of the entire organism from ischemia during no-flow or very low flow (shock) states to permit definitive surgical care to be provided.150,151 At present, ultraprofound hypothermia allows up to 2 hours of pulseless viability in large animal models.151–153 Current efforts in this field include the development of fully synthetic solutions that completely replace the circulating blood volume after total body washout.152 What is certain, though, is that the surgeon’s presence will be required at the bedside to acutely diagnose and provide effective hemorrhage control, either directly or by coordinating available hospital resources.
Acknowledgements
We thank Dr. Rosa Chun, Department of Anesthesia, and Dr. John Kortbeek, Chief of Surgery, Foothills Medical Centre, Calgary, Alberta, for reviewing the manuscript. et al. Isolated brain injury as a cause of hypotension in the blunt trauma patient. J Trauma 2003;55:1065–9.
Footnotes
Competing interests: A.W. Kirkpatrick served on a multinational research advisory board concerning the use of Recombinant Factor VIIa in traumatic injury.