Abstract
Background: Breast cancer is the most common cancer affecting females in Canada, and about half of females with breast cancer are treated with mastectomy. We sought to evaluate geographic variation in breast reconstruction surgery in Alberta, Canada.
Methods: Using linked population-based administrative databases, we extracted data on all Alberta females aged 18 years and older who were diagnosed with breast cancer and treated with mastectomy during 2004–2017. Analyses included regression modelling of odds of reconstruction at 1 year and a spatial scan to identify geographic clusters of lower numbers of reconstruction.
Results: A total of 16 198 females diagnosed with breast cancer were treated with a mastectomy, and 1932 (11.9%) had reconstruction within 1 year postmastectomy. Those with reconstruction were more likely to be younger (adjusted odds ratio [OR] 16.7, 95% confidence interval [CI] 13.7–20.3; aged 21–44 yr v. ≥ 65 yr) and were less likely to be from lower-income neighbourhoods. They were more likely to have at least 1 comorbidity and were more likely to have advanced stages of cancer and to require chemotherapy (adjusted OR 0.55, 95% CI 0.47–0.65) or radiotherapy after mastectomy (adjusted OR 0.59, 95% CI 0.39–0.87) than females without reconstruction. We identified rural northern and southeastern clusters with frequencies of reconstruction that were 69.6% and 41.6% of what was expected, respectively.
Conclusion: We found an overall postmastectomy rate of breast reconstruction of 11.9%, and we identified geographic variation. Predictors of reconstruction in Alberta were similar to those previously described in the literature, specifically with patients in rural communities having lower rates of reconstruction than their urban counterparts. These results suggest that further interventions are required to identify the specific barriers to reconstruction within rural communities and to create strategies to ensure equitable access to all residents.
Breast cancer is the most common cancer affecting females in Canada, accounting for 25% of all new malignancies.1 Among this patient population, approximately half are treated with mastectomy.2 Despite a trend toward breast-conserving surgery for females with breast cancer, rates of postmastectomy breast reconstruction surgery continue to rise owing to improved screening resulting in earlier detection, as well as increased patient and physician awareness of, and accessibility to, reconstructive options after mastectomy. A person’s motivation for seeking reconstruction is highly individualized and multifactorial.3
The benefits of breast reconstruction surgery are well recognized. A 2002 statement from the National Institute for Health and Clinical Excellence recommended that “reconstruction should be available [to all females with breast cancer] at the initial surgical operation” (https://www.nice.org.uk/guidance/csg1/resources/improving-outcomes-in-breast-cancer-update-pdf-773371117). The statement was further reinforced in 2009, when the institute specifically noted that all patients, regardless of geographic location, should be offered the option of reconstruction (www.nice.org.uk/nicemedia/pdf/CG80NICEGuideline.pdf). Nevertheless, despite a growing body of evidence correlating breast reconstruction with improvements in psychological, functional, and sexual well-being, this elective procedure remains underused. Factors theorized to influence these low statistics include age, socioeconomic background, cancer stage, and living rurally.4
In this study, we describe rates of breast reconstruction by administrative health geographic zones over time for females in Alberta, Canada, diagnosed with breast cancer and treated with a mastectomy. Further, we identify geographic areas with lower-than-expected rates of breast reconstruction at 1 year postmastectomy, and identify geographic areas with longer-than-expected times to delayed breast reconstruction. Finally, we examine the key patient and tumour factors associated with the identified geographic areas of reconstruction underrepresentation. We hypothesized that rural and remote areas would have lower rates of breast reconstructions at 1 year postmastectomy, with longer times to reconstruction.
Methods
This retrospective population-based cohort study is based on linkage of large administrative health databases from Alberta.
Study population
The study population consisted of all females aged 18 years and older, residing within the province of Alberta, diagnosed with breast cancer, and treated with a mastectomy between Jan. 1, 2004, and Dec. 31, 2017. Inclusion criteria were a pathology diagnosis of in situ or invasive breast cancer, and treatment with mastectomy. We extracted cases from the Alberta Cancer Registry in 2020. Patients undergoing mastectomy for prophylactic reasons were excluded, as these patients are not currently traced in Alberta databases.
Alberta is a province in western Canada with a uniform, single-payer health care system that provides medically needed treatments at no cost to residents. Alberta Health Services (AHS) is the provincial agency charged with the delivery of health services and has access to the administrative databases maintained by Alberta Health, the provincial health ministry. Alberta has a land mass of 640 330 km2 and had a population of 4 067 175 in 2016.5
Data sources and variables
In this study, we used linked records from population-based, administrative health data sources: Alberta Cancer Registry, Discharge Abstract Database,6 and National Ambulatory Care Reporting System (NACRS).7
Alberta has a population-based cancer registry that has been certified by the North American Association of Central Cancer Registries.8 The Alberta Cancer Registry was established in 1942 and is legally mandated by the government to capture information on residents diagnosed with cancer. Information collected includes patient identifiers (e.g., surname and postal code), demographic characteristics of the patient (e.g., sex and age), features of the tumour (e.g., date of diagnosis, malignancy number, morphology, cell type behaviour, and collaborative stage), and treatments (status of chemotherapy and radiotherapy initiation). We calculated the Charlson Comorbidity Index9 within 6 months of breast cancer diagnosis and ignored the scores for cancer and metastasis. An indicator of 1 or more comorbidities was created. The postal code data in conjunction with the Postal Code Conversion File10 also allowed for neighbourhood income quintile and community size (< 10 000, 10 000–99 999, and ≥ 100 000) to be obtained.
We linked data for females who underwent mastectomy with the inpatient data (Discharge Abstract Database) and outpatient data (NACRS) to determine breast reconstruction dates. For each patient, the data after the diagnosis date and up to Dec. 31, 2018, were extracted. We chose this cut-off date to have the most currently available data that provide at least 1 year of data after mastectomy. The data extracted included dates and procedure codes based on the Canadian Classification of Health Interventions.11,12
For the delivery of health services, AHS divides the province into 5 geographic zones from north to south of the province:13 North, Edmonton, Central, Calgary, and South had populations of 477 558, 1 345 999, 472 304, 1 599 624, and 300 432 in 2016, respectively.14 The Edmonton Zone is in the geographic centre of the province and has the capital city of Edmonton along with multiple nearby urban communities (e.g., Sherwood Park and St. Albert). The other major metropolitan area is the Calgary Zone, with Calgary, Airdrie, and Banff as notable communities. The Central Zone has Red Deer and is more rural, along with the South Zone (e.g., Lethbridge and Medicine Hat). The North Zone has the largest geographic area and is the most rural, with some notable communities such as Fort McMurray and Grand Prairie. Smaller geographic units called sub–regional health authorities were introduced in 2003 and correspond to smaller geographic units than the zones (Appendix 1, Figure 1, available at www.canjsurg.ca/lookup/doi/10.1503/cjs.003823/tab-related-content). The residence at the time of diagnosis for each patient is geocoded to sub–regional health authorities and zone based on the 2003 boundaries. In addition, AHS provided geographic population-based centroids (latitude/longitude) for each of the sub–regional health authorities.
Case and outcome definition
We defined a case as a female in Alberta diagnosed with an in situ or an invasive breast cancer at age 18 years or older who had a mastectomy and breast reconstruction within 1 year of mastectomy. Mastectomies were defined by surgical modality for breast tumour (extended total mastectomy, modified radical mastectomy, and total mastectomy) by the Alberta Cancer Registry. The earliest date of one of these surgical modalities formed the date of mastectomy. Breast reconstruction was defined with the codes 1.YM.80 (repair breast), 1.YM.88 (excision partial with reconstruction, breast), 1.YM.90 and 1.YM.92 (excision total with reconstruction, breast and excision radical with reconstruction, breast), 1.YM.79 (repair, by increasing size), and 1.YM.78 (repair, by decreasing size). The earliest date of one of these codes formed the date of breast reconstruction. The time to breast reconstruction was the date of reconstruction minus the date of mastectomy. If the time was 365 days or fewer, the patient was considered to have had a reconstruction within 1 year.
Statistical analysis
Numerical summaries (e.g., frequency, percentage, mean, and standard deviation [SD]) describe patients with mastectomy who have breast reconstruction within 1 year (cases) or do not have breast reconstruction within 1 year. Crude rates and age-group directly standardized rates are provided for cases over time and zone (along with 95% confidence intervals [CIs]). The population of females with mastectomy in Alberta in 2004 formed the reference population. We analyzed data using R.15 A multivariable logistic regression model was fit to determine the relation between odds of breast reconstruction at 1 year postmastectomy and predictor variables (age group at diagnosis, year of diagnosis, neighbourhood income quintile, community size, zone, cancer stage, chemotherapy, and radiotherapy). Similarly, a Cox proportional hazards regression model was fit on the time to breast reconstruction from mastectomy (censoring on Dec. 31, 2018, or death date before Dec. 31, 2018) with the same predictor variables. We report adjusted odds, hazard ratios, and associated 95% CIs. We used multivariate imputations for the 1% of cancer stage data that were missing using the mice package.16
In general, statistical cluster detection tests identify unexpected aggregations of cases in geographic area. We used 2 types of spatial scan statistics to identify sub–regional health authorities that were clusters of lower-than-expected numbers of females with breast reconstruction at 1 year postmastectomy (spatial scan for count data17) and clusters of lower-than-expected times to breast reconstruction (spatial scan for time-to-event data with an exponential distribution18). Spatial scan statistics, first proposed by Kulldorff and Nagarwalla,17 have become a popular method for identifying spatial clusters because it easily accounts for areas with diverse population sizes, identifies clusters of different sizes, identifies cluster locations, and tests the tendency to cluster.19 Heuristically, the method creates all combinations of neighbouring areas and calculates parameters inside and outside the combined area. The spatial scan identifies a combination of areas that forms a primary cluster (the most likely cluster that has the highest maximum likelihood ratio and rejects the null hypothesis of no clustering). The combination of multiple small geographic areas allows for greater identification of small geographic clusters.
The spatial scans were conducted for all years in the study period combined, with a scan window of up to 30% of the population used (i.e., the population in each sub–regional health authority is the number of females diagnosed with breast cancer who have had mastectomies). Thirty patients were dropped from the analyses because of missing sub–regional health authorities. Maps prepared with ggmap20 show the identified clusters, and Kaplan–Meier curves display the time to breast reconstruction for patients who reside inside and outside the clusters. SaTScan21 was used for the cluster detection tests through the R package rsatscan.22 A p value less than 0.05 was considered statistically significant.
With clusters identified, we used multivariable logistic regression analyses to determine whether there were characteristics of patients (other than geographic location) and their treatment that increased the odds of being in an area identified as a cluster. Predictors in the model were age group at diagnosis, neighbourhood income quintile, community size, cancer stage, chemotherapy, and radiotherapy.
Ethics approval
The Health Research Ethics Board of the University of Alberta (Pro00092044) approved this study, and patient consent was not required.
Results
Geographic and temporal trends
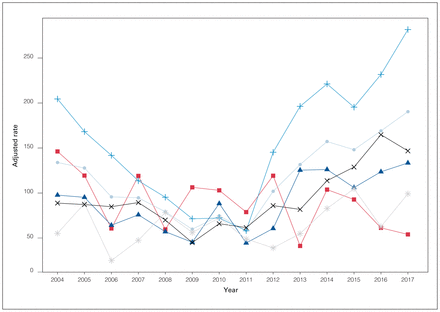
During the study period, 16 198 females in Alberta diagnosed with breast cancer were treated with a mastectomy. A total of 1932 (11.9%) of these patients underwent breast reconstruction within 1 year of their mastectomy, with zone proportions ranging from 6.9% in North to 18.0% in Calgary. Of the patients who underwent reconstruction, 1402 (72.6%) did so immediately, at the same time as their mastectomy. Patients with reconstruction were younger (mean age at diagnosis 49.9 yr) than those without reconstruction (61.4 yr, p < 0.001, Table 1). Females with reconstruction had fewer comorbidities (p < 0.001), were diagnosed at an earlier cancer stage (p < 0.001), and were less likely to require chemotherapy (p < 0.001) or radiotherapy after mastectomy (p < 0.001). Rates of reconstruction at 1 year rose for most zones after 2011, with sizable increases in the Calgary Zone (Figure 1 and Appendix 1, Table 2). In total, about 25% (4052) of females had reconstruction recorded in the data set, independent of the 1-year time frame (i.e., 2120 had reconstruction later than 1 yr after mastectomy).
Age-group directly standardized rates of 1-year breast reconstruction surgery per 1000 population of females with mastectomies by fiscal year and zones All (●), North (*), Edmonton (x), Central (▲), Calgary (+), and South (■).
Characteristics of females diagnosed with breast cancer who had mastectomies in Alberta during 2004 to 2017 and those who did and did not have breast reconstruction within 1 year
Multivariable regression models for the 2 outcomes, odds of reconstruction at 1 year, and time from mastectomy to reconstruction appear in Appendix 1, Table 3. For reconstruction at 1 year, younger patients were more likely to have reconstruction (adjusted odds ratio [OR] 16.7, 95% confidence interval [CI] 13.7–20.3 for those aged 21–44 v. ≥ 65 yr), and patients were less likely to have reconstruction if they were from zones other than Calgary, were from lower neighbourhood income quintiles, had at least 1 comorbidity (OR 0.80, 95% CI 0.66–0.95), had more advanced stages of cancer, and required chemotherapy (OR 0.55, 95% CI 0.47–0.65) or radiotherapy after mastectomy (OR 0.59, 95% CI 0.39–0.87). Analogous results were seen for the time from mastectomy to reconstruction outcome, with the exception that chemotherapy after mastectomy was no longer a statistically significant predictor. Results varied for the different years of diagnosis for the 2 outcomes.
Geographic clustering of low numbers of patients with breast reconstruction at 1 year
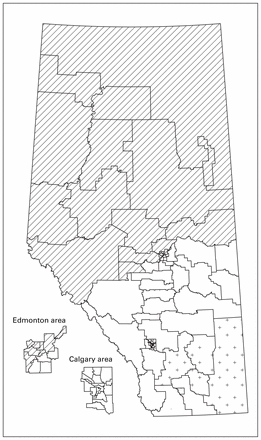
The spatial scan identified 2 statistically significant clusters at the 5% level: 1 in the north and 1 in the southeast (Figure 2). The north cluster had 387 patients with reconstruction, whereas 556.2 cases were expected given the population size of 4664 (p < 0.001). These northern areas are primarily rural. The southeast cluster had 40 patients with reconstruction, whereas 96.1 cases were expected given the population size of 806 (p < 0.001).
Clusters of lower-than-expected numbers of breast reconstruction surgery at 1 year (primary cluster = diagonal lines, secondary cluster = + symbols).
Females residing inside the clusters differed on some characteristics from those residing outside the clusters (Table 2). When multivariable logistic regression was considered, females in the clusters generally were from lower neighbourhood income quintiles and smaller communities, had more advanced cancer stages, were more likely to have a comorbidity (OR 1.50, 95% CI 1.38–1.63), and were more likely to have had radiotherapy after mastectomy (OR 1.35, 95% CI 1.13–1.63) than those residing outside the clusters.
Multivariable logistic regression models for the odds of being in the clusters versus being outside the clusters of low counts of breast reconstruction at 1 year
Geographic clustering of longer times to breast reconstruction
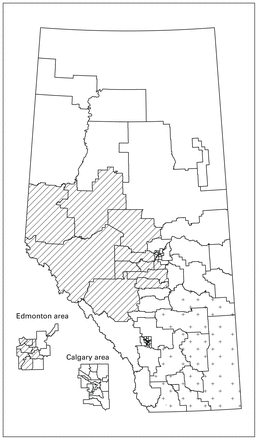
The exponential spatial scan identified 2 statistically significant clusters at the 5% level: 1 in the west and 1 in the southeast. Some parts of these clusters were parts of clusters from the analyses above based on low numbers of cases of breast cancer reconstruction at 1 year. The west cluster (p = 0.001) (Figure 3) had 969 females who had breast reconstruction from a population of 4815. Females in the west cluster had longer times to reconstruction than those living outside the clusters (Appendix 1, Figure 2). The southeast cluster (p = 0.001) had 315 breast reconstructions from 1664 patients. These females also had longer times to reconstruction than those living outside the clusters.
Clusters of longer-than-expected time to breast reconstruction surgery (primary cluster = diagonal lines, secondary cluster = + symbols).
Females residing inside the clusters differed on some characteristics from those residing outside the clusters (Table 3). When multivariable logistic regression was considered, patients in the clusters were older, from smaller communities, more likely to have a comorbidity (OR 1.30, 95% CI 1.19–1.41), and more likely to have stage I, II, or III cancers (compared with stage 0) than those residing outside the clusters.
Multivariable logistic regression models for the odds of being in the clusters versus being outside the clusters of longer times to breast reconstruction
Discussion
In this study, we used administrative health databases over a 14-year period within the province of Alberta to identify 16 198 females who underwent mastectomy for breast cancer, 1932 of whom underwent subsequent breast reconstruction within the first year postmastectomy. Overall, age-group directly standardized rates of breast reconstruction at 1 year declined during the period from 2004 to 2011 and then increased to 190.4 per 1000 by 2017. Regional variation was seen, with patients in the Calgary Zone generally having the highest rates of reconstruction and patients in the North and South Zones having the lowest rates. When smaller geographic areas were examined for clustering, parts of the southeast of the province were identified as having lower-than-expected numbers of breast reconstruction at 1 year and longer time to breast reconstruction. For each outcome, neighbourhood income quintile, community size, comorbidities, and cancer stage were important factors that differed for patients residing in the clusters associated with higher rates of reconstruction compared with patients outside the clusters. For analyses based on breast reconstruction within 1 year, patients in the cluster associated with low counts of breast reconstruction were more likely to have radiotherapy after mastectomy. For analyses based on time to reconstruction, patients in the cluster associated with longer times to reconstruction were less likely to be from younger age groups.
Our findings are consistent with previous results identifying predictors for undergoing breast reconstruction, including age, socioeconomic status, and geographic location.4
Multiple variables contribute to the decision for a patient to undergo postmastectomy breast reconstruction. Considerations including the need for adjuvant therapy, patients’ overall health, donor site availability (autologous reconstruction), acceptability of a prosthesis (alloplastic reconstruction), and patient preference together play a role in determining whether reconstruction is the right decision for the individual. Given the highly personalized nature of this decision, it follows that there is no specific ideal rate of postmastectomy breast reconstruction. Rather, the focus is on ensuring equitable availability to all patients, which can be achieved only by identifying extrinsic barriers to reconstruction that may preclude access to these procedures.
To date, breast reconstruction rates in Canada have been reported in only 2 provinces: 3.8% in Nova Scotia (1991–2001)23 and 7.9% in Ontario (1994–1995).24 These studies, however, report data that are dated by nearly 3 decades, and thus are likely not representative of current trends in Canada. To our knowledge, our data are the first report of reconstruction rates in Alberta and the only recent description of trends within Canada. Our results indicate that, with an overall reconstruction rate of 12%, more reconstruction is being performed than previously described in the country. This is consistent with international literature suggesting that rates of breast reconstruction are increasing. Historically, breast cancer reconstruction rates among patients with mastectomy have been reported as low, including in Australia (10%), Denmark (14%), and England (16.5%), with China reporting a 3.1% reconstruction rate among all patients with breast cancer.25–28 Recent reports, however, suggest substantial increases internationally. Within the United States, rates of breast reconstruction are reported to be increasing dramatically,29,30 with major cancer institutes reporting rates of reconstruction increasing from 46% to more than 60%.31 This increase has been at least in part attributed to the implementation of the Women’s Health and Cancer Rights Act, which mandated that insurance companies cover the costs associated with breast reconstruction. Data from Korea suggest that since its National Health Insurance Service began covering breast reconstruction, rates of postmastectomy reconstruction have risen to 53% (70% alloplastic and 30% autologous).32 Similarly, data from the UK (23.3%)33 and Australia (18%)34 suggest that reconstruction rates within these countries are increasing; together, these rates are substantially higher than the rates in Alberta.
In keeping with worldwide trends, the number of patients undergoing breast reconstruction is increasing in Alberta. We identified a prominent increase in the rates of reconstruction after 2011, particularly in the Calgary Zone. These findings are supported by recent literature out of the University of Calgary reporting a reconstruction rate of 23% between 2016 and 2018.35 These trends are likely attributable to improved patient and surgical oncology awareness, detection at an earlier stage, and an increased divisional, institutional, and provincial impetus on improving patient access. Despite these drivers, however, substantial improvements in reconstruction rates were limited to the Calgary regions. Although the cost of health care–related expenses is not a deterrent for patients in Canada given the national health care program, the costs associated with travel to urban centres may be a barrier to reconstruction for patients from a rural community. This is supported by observations that increasing trends in reconstruction rates are largely limited to urban centres. Identification of barriers to reconstruction is of importance to procure resources and generate interest in strategy development toward addressing this gap in patient care. One notable barrier is the perceived time sensitivity of service delivery. Breast reconstruction is generally termed “elective” surgery. As such, access in the setting of limited resources is challenging as it is not considered a priority by governing bodies. One such example was during the COVID-19 pandemic, when rates of breast reconstruction nationally were decreased by 50%–90%.36 While the need to prioritize patients affected by the pandemic is indisputable, as a result, the wait-list for reconstruction has increased substantially, further limiting patient access across the country.
Consistent with the findings of our study, younger patient age, higher socioeconomic status, higher degree of education, shorter distance from the treating hospital, and the patient having private insurance have been previously described as influencers on the rate of breast reconstruction.37,38 The identification of similar barriers among Albertans as have been described both within and beyond Canada suggests that the province may benefit from strategies that have already been developed by other centres for addressing these barriers. Further, specific identification of regions within the province in which there is disparity will allow for collaboration with both local physicians and patients to identify needs and reallocate resources to address the specific barriers within the individual communities.
Limitations
Limitations for the study include that the coding of cancers may have varied over the years as well as the diagnostic methods. We have assumed that patients have not left the province to obtain reconstructive surgery that would not be recorded in administrative databases at Alberta Health Services.
Conclusion
We report an overall postmastectomy rate of breast reconstruction of 11.9% within the province of Alberta. Our results suggest that predictors of breast reconstruction in Alberta are similar to those previously described in the literature, specifically with patients in rural communities having lower rates of reconstruction than their urban counterparts. These results suggest that further interventions are required to identify the specific barriers to reconstruction within rural communities, and to create strategies to ensure equitable access to all residents.
Acknowledgement
The authors thank Truong-Minh Pham of Surveillance & Reporting at Cancer Control, Alberta Health Services for working with them to extract the data set.
Footnotes
Competing interests: None declared.
Contributors: Yifu Huang performed statistical analyses, provided an initial draft of the manuscript, and critically reviewed the manuscript for important intellectual content. Jenna-Lynn Senger obtained funding, interpreted results, and reviewed and revised the manuscript. Lisa Korus contributed to study conceptualization, obtained funding, interpreted results, and critically reviewed the manuscript for important intellectual content. Rhonda Rosychuk conceptualized and designed the study, coordinated and supervised data extraction, obtained funding, directed statistical analyses, interpreted results, and reviewed and revised the manuscript. All authors approved the final version to be published and agree to be accountable for all aspects of the work.
Funding: This research has been funded by the Alberta Women’s Health Foundation through the Women and Children’s Health Research Institute with an Innovation Grant. The funding source had no direct involvement in the study design, analysis, interpretation, or decision to submit this work.
- Accepted February 12, 2024.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/