Abstract
Superior mesenteric artery syndrome (SMAS) is a rare condition. The diagnosis is usually made by exclusion. A previously healthy 20-year-old woman who had recurrent SMAS is described. Diagnosis of the condition was difficult. Initially, small-bowel enteroclysis, upper gastrointestinal series and endoscopy, biopsy of gastric and duodenal mucosa, abdominal computed tomography (CT) and ultrasonography were used to make the diagnosis. Abdominal CT suggested pancreatitis causing compressive obstruction of the superior mesenteric artery. Conservative management was helpful at first, but cramping and projectile emesis recurred. Upper gastrointestinal series suggested duodenal distension and a filling defect in the region of the superior mesenteric artery. Repeat endoscopy showed a lateral pulsatile compression in the region of the distal duodenum and SMAS was diagnosed. Medical therapy was not helpful so duodenojejunostomy was carried out. The operation was successful and the patient was symptom-free for 1 year, when the syndrome recurred, with symptoms of periumbilical pain, intermittent episodes of vomiting and loose stools. At reoperation the duodenojejunal anastomosis was found to be displaced to the left of the superior mesenteric artery pedicle causing recurrent obstruction. The duodenojejunostomy was converted to a Roux-en-Y duodenojejunostomy. The patient has since remained well. A MEDLINE search of the literature for the period 1961 to October 1994 revealed that there were no reported cases of a recurrence of SMAS in an otherwise healthy adult patient. In spite of the difficulty in diagnosing this condition, heightened awareness can lead to early diagnosis and avoid unnecessary suffering for the patient.
Superior mesenteric artery syndrome (SMAS) is an uncommon condition thought to be caused by intermittent functional obstruction of the third part of the duodenum between the superior mesenteric artery and the vertebral column and aorta.1 Since the entity was first described at autopsy by von Rokitansky in 1861,2 about 400 cases have been reported in the medical literature. 3–6 However, there are no epidemiologic data to verify the true incidence.
The condition is often referred to as Wilkie’s syndrome or cast syndrome. In 1908, Laffer7 presented one of the first reviews. In 1927, Wilkie8 published the largest and most complete study of this disease, based on 75 cases. Skepticism about the existence of the condition continued until the 1960s, when new radiologic techniques provided evidence to support the existence of SMAS. Some still question whether it is a true entity9 and others believe that the syndrome is overdiagnosed.10 Currently, the reported incidence is on the rise, possibly due to increased physician awareness.11
Clinical symptoms include epigastric pain, a sensation of fullness, nausea and bilious vomiting. Weight loss is common because patients often regurgitate their food or become afraid to eat.12 Weight loss can in turn exacerbate the condition.12–14 Symptoms can be relieved by postural changes in the prone or knee–chest position, suggesting vascular compression. However, since relief with postural change occurs inconsistently, its absence does not rule out vascular compressive syndromes. 12 Moreover, peptic ulcer disease coexists with SMAS in up to 25% of cases.5,11,15,16 Whether duodenal obstruction precedes the peptic ulcer or the peptic disease contributes to the arteriomesenteric duodenal compression is uncertain.
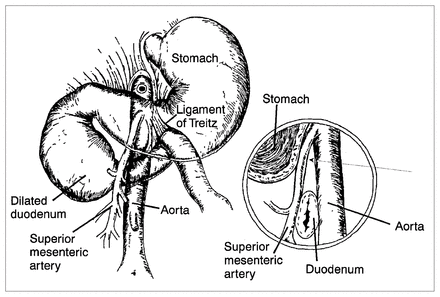
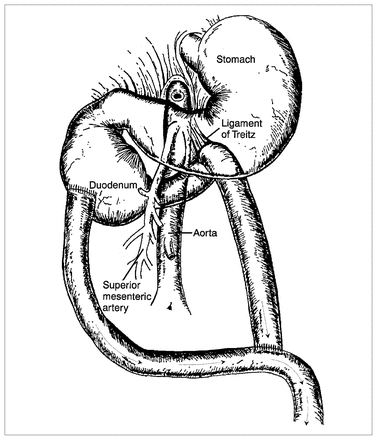
Many causes for SMAS have been suggested. These include a high insertion of the duodenum at the ligament of Treitz, a congenitally low origin of the superior mesenteric artery and compression of the duodenum caused by peritoneal adhesions, which are a result of duodenal malrotation.17,18 However, the most common cause is thought to be narrowing of the aortomesenteric angle and the distance between the aorta and the superior mesenteric artery at the level of the duodenum, with consequent extrinsic compression of the duodenum (Fig. 1).19 Some believe that because of our erect posture, the superior mesenteric artery leaves the aorta at a more acute angle than it does in quadrupeds.12 The disorders that predispose to SMAS have been divided into five categories: severe wasting diseases such as burns, cancer and endocrine diseases; severe injuries such as head trauma; spinal trauma, deformity, including the application of a body cast; dietary disorders such as anorexia nervosa or malabsorptive syndromes; and the postoperative state.20–26 Symptoms of SMAS are nonspecific, and the syndrome can be difficult to differentiate radiologically from peptic ulcer disease, cholelithiasis, chronic pancreatitis and abdominal angina.27 We describe a case of SMAS with recurrence in a previously healthy adult.
Anatomic relationship between duodenum, aorta and superior mesenteric artery in superior mesenteric artery syndrome (SMAS). Lateral view shows compression of duodenum by superior mesenteric artery. Arrow indicates abnormal, narrowed aortomesenteric angle.
Case report
A 20-year-old previously healthy, slender woman initially presented with a history of increasing abdominal cramps, nausea and vomiting for 3 days. There was no history of an eating disorder. She was not taking any medication and her personal and family histories were unremarkable. She had a low-grade fever and was extremely dehydrated. Apart from a marked tenderness and distension of the epigastrium, the findings on physical examination were normal. The leukocyte count was elevated (21.4 × 109/L), but the serum amylase level was within normal limits.
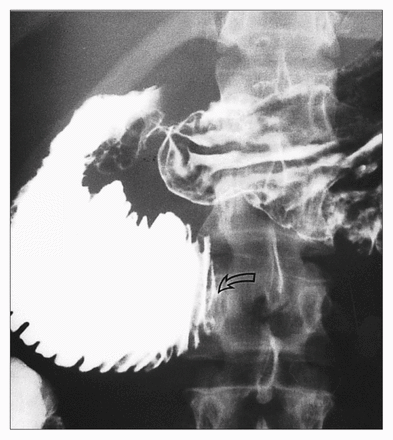
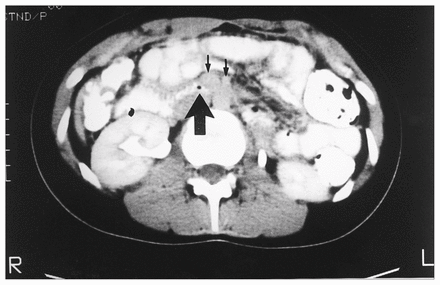
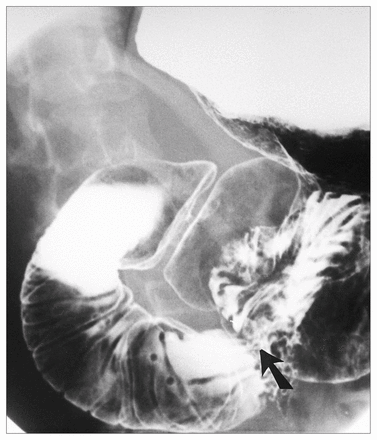
Three views of the abdomen showed a virtually gasless abdomen with an air bubble seen in the region of the duodenum. On small-bowel enteroclysis, a withdrawal duodenogram showed a relative dilatation of the third and fourth portions of the duodenum. An upper gastrointestinal series revealed thickened duodenal folds and a hold-up of contrast medium at the level of the superior mesenteric artery (Fig. 2). Two days later, upper gastrointestinal endoscopy revealed a normal stomach with no evidence of obstruction in the first or second parts of the duodenum. Biopsy specimens of both gastric and duodenal mucosa were also normal. The upper gastrointestinal series was repeated 7 days after the last study and showed only a transient hold-up of barium at the level of the superior mesenteric artery. Abdominal computed tomography (CT) 2 days later demonstrated a prominent head of the pancreas, but there was no mass lesion; however, the region of the duodenum near the superior mesenteric vessels was narrowed (Fig. 3). Despite the normal serum amylase level, a transient resolving idiopathic pancreatitis, causing temporary superior mesenteric artery compressive obstruction was diagnosed. Initially, the patient responded well to conservative management. Abdominal ultrasonography done 2 months later showed a normal abdomen.
Upper gastrointestinal series after 1 day of nasogastric suction shows high-grade obstruction at junction of third and fourth portions of duodenum (open arrow) and thickened duodenal folds proximal to obstruction.
Computed tomography scan outlines normal proximal duodenum with narrowing of duodenal lumen (large arrow) at level of superior mesenteric vessels (small arrows).
One month after the ultrasonography, the patient again had episodes of projectile emesis with occasional epigastric and lower abdominal cramps. A repeat upper gastrointestinal series showed distension of the duodenum proximal to the junction of the third and fourth portions, with a variable linear filling defect crossing the region, in keeping with the superior mesenteric vessels. There was no evidence of a duodenal web. A repeat flexible endoscopic examination with a colonoscope revealed, interestingly, lateral pulsatile compression corresponding to the peripheral pulses in the distal duodenum. This had not been observed previously with the shorter endoscope. Although the endoscope could be passed through the lateral compression, there was no doubt that the compression did contribute to obstruction. These findings were consistent with those of SMAS.
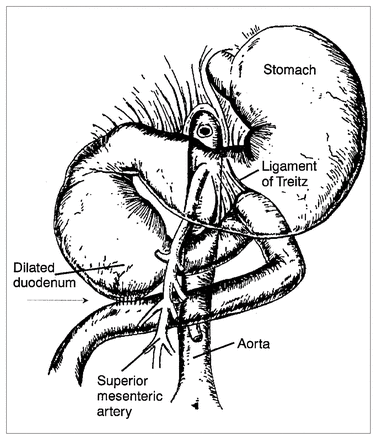
Despite adequate medical therapy, the patient continued to vomit bilious fluid and to lose weight. A duodenojejunostomy was done. Intraoperatively there was a definite change in calibre from the second part of the duodenum to the proximal jejunum, with the duodenum being considerably larger. The third part of the duodenum was mobilized and sewn side to side to the first loop of jejunum distal to the ligament of Treitz, well to the right of the aorta (Fig. 4). The patient’s recovery was uncomplicated and she was discharged home after 11 days in hospital. At the time of discharge she was tolerating small meals well.
Diagram of first surgical procedure showing side-to-side anastomosis (arrow) of third part of duodenum to first loop of jejunum, distal to ligament of Treitz and to right of aorta.
The patient remained well for over a year. Then she began to have crampy periumbilical pain, which progressed over a few months to intermittent vomiting. She also complained of loose stools with no bleeding and she lost 6.8 kg over 3 months. Another upper gastrointestinal series revealed a discrepancy in size at the junction of the third and fourth portions of the duodenum, suggesting duodenal obstruction (Fig. 5).
Repeat upper gastrointestinal series 16 months postoperatively again shows narrowing at junction of third and fourth portions of duodenum proximal to anastomosis (arrow).
The working diagnosis was stricture at the duodenojejunal anastomosis causing recurrent SMAS. Small-bowel enteroclysis done 2 weeks later showed variable compression of the duodenum in the region of the superior mesenteric vessels. The majority of the barium passed beyond this point into the jejunum, but only a small amount was seen to pass to the anastomotic site, implying a high-grade obstruction. Surgery was recommended.
Intraoperatively, the proximal duodenum and the duodenal bulb were dilated, but there was no stricture at the duodenojejunal anastomosis. The previous anastomosis had simply been displaced to the left of the superior mesenteric pedicle, explaining the recurrence. To bypass the obstruction, a decision was made to take down the previous duodenojejunostomy and convert to a Roux-en-Y duodenojejunostomy. The proximal end of the 40 cm long Roux limb was anastomosed end-to-side to the second part of the duodenum (Fig. 6). Her recovery was uncomplicated and she resumed a regular diet within a week. At follow-up 2 years later she was still well.
Diagram of second surgical procedure in which previous duodenojejunostomy was taken down and converted to Roux-en-Y duodenojejunostomy. Arrows indicate direction of gastrointestinal flow.
Discussion
Vascular compression of the duodenum can present at any age, but the majority of patients are adolescents or young adults.3,5,11,12,15,19 Females are affected more often than males.1,3,12,19 Although there are those who question the existence of this entity,9 others believe that SMAS is not rare but a matter of degree, claiming that many people with only mild compression do not present with symptoms.12 In the past 18 years, there has only been one reported death due to SMAS.12
The clinical diagnosis can be confirmed by radiologic studies obtained during an acute attack; otherwise radiologic evidence may be absent. 1,3,5,15,28–30 In reviewing 81 cases in the literature, Akin, Gray and Skandalakis12 discovered that a diagnosis of vascular compression was made radiologically in 95% of the cases. The remainder did not show conclusive evidence and the diagnosis was made only during operation. Endoscopic examination usually does not help in the diagnosis.1 The following radiologic criteria have been established for the diagnosis of SMAS: dilatation of the first and second portions of the duodenum, with or without gastric dilatation; abrupt vertical and oblique compression of the mucosal folds; antiperistaltic flow of barium proximal to the obstruction, producing to-and-fro movement; delay of 4 to 6 hours in transit through the gastroduodenal region; and relief of obstruction when the patient is placed in a position (prone or knee–chest) that diminishes the drag of the small-bowel mesentery. 10,15,28 In particular, the routine use of intravenous hyoscine butylbromide for the contrast studies may actually mask the to-and-fro peristalsis if administered at the beginning of the examination.13
Hypotonic duodenography has been shown to be of diagnostic value.30 When the duodenal loop is examined in a flaccid state, free from peristalsis, the duodenal mucosa is clearly visualized. In the study by Gustafsson and associates1 hypotonic duodenography suggested the diagnosis in 10 of 11 patients, whereas the barium meal examination was suggestive in only 1 patient. Lukes and associates30 have shown that hypotonic duodenography is more accurate in distinguishing between normal and abnormal states, especially if the barium meal studies have given negative results. Recent reports have claimed that dynamic thin-section CT with sagittal reconstruction is an excellent imaging modality.31,32
By the use of selective superior mesenteric arteriography against a barium-filled duodenum it is possible to demonstrate extrinsic compression and to measure the aortomesenteric angle and the distance from the aorta. In SMAS, the mean aortomesenteric angle is 8° (range from 6° to 15°) and the mean aortomesenteric distance is 6 mm (range from 2 to 8 mm).1 Normal values have been reported to be 25° to 60° and 10 to 28 mm respectively. 30,31 Typically, patients with SMAS have a narrow angle, in the range of 6° to 11°.11,14,17,19,29,31,32 These angles have been calculated by several investigators who used angiography, ultrasonography and CT.19,31 However, angiography did not increase diagnostic accuracy and should be reserved for questionable cases.19 Some investigators believe that the aortomesenteric distance is more crucial than the aortomesenteric angle in determining vascular compression.31
Lumbar lordosis or anterior abdominal pressure from a body cast will exacerbate the problem by altering the aortomesenteric angle.11 Lumbar lordosis is particularly pronounced in women.12 Dorph first used the term “cast syndrome” for orthopedic patients who suffered from this syndrome. They often lost weight and were kept immobile in a supine position during prolonged periods of bedrest.33 Patients with anorexia nervosa are also prone to SMAS because of the loss of periduodenal and retroperitoneal fat.11,15,16,20 Interestingly, vascular compression is virtually unknown in obese people.12 Lastly, there may be a genetic predisposition to the development of this syndrome because clustering of this disorder was reported in 1990.34
Medical treatment is usually successful in patients with a short history and a relatively minor degree of duodenal stasis shown radiologically.4 Conservative methods, including rest of the gastrointestinal tract, fluid resuscitation, total parenteral nutrition and lying in the left lateral decubitus position are generally recommended as the first line of treatment.5,10,17–19 Hutchinson and Bassett11 and Munns and colleagues35 both reported 100% success rates without any surgical intervention. However, both these series were in orthopedic patients who had an acute postoperative episode. Biopsy specimens of the duodenum and jejunum should be obtained before surgery to exclude infiltrative, infective, neurologic or malignant causes for the duodenal dilatation and chronic idiopathic intestinal pseudoobstruction. 13,31,36 Surgery is indicated if there is a long history of indigestion, progressive weight loss, pronounced dilatation and stasis of the duodenum. 4,28,37 Duodenojejunostomy from the second portion of the duodenum to the jejunum is now considered to be the surgical treatment of choice.1,3–5 Other procedures include gastrojejunostomy, Roux-en-Y duodenojejunostomy and anterior transposition of the third part of the duodenum. The last technique has apparently been shown to be an effective and permanent direct approach.38 Lysis of the ligament of Treitz has the advantage that it does not include any intestinal anastomosis as advocated by some.12 It is especially recommended in children.39
- Accepted November 7, 1995.