Abstract
Duplication of the alimentary tract may affect patients of all ages. Although they are relatively rare, the importance of these congenital lesions lies in the fact that they readily mimic other surgical disease processes and may result in significant morbidity if left untreated. Prompt recognition and treatment using combined radiologic and surgical management are generally associated with an excellent outcome. Three patients who presented with intestinal duplication arising from each of the major embryologic origins are reported. Their clinical histories reveal the spectrum of presentation associated with these lesions and provide a framework for a discussion of current management strategies.
The clinical presentation of intestinal duplication in adults ranges from chronic nonspecific gastrointestinal discomfort to one of acute abdominal pain. Although several series of these lesions have been described in children,1,2 there have been few descriptions in adults. We present the cases of 3 adults who presented recently with duplication of the foregut, midgut and hindgut. These lesions are important because these duplications share patterns of presentation common to many nonspecific intra-abdominal diseases, yet are readily managed by a combined radiologic and surgical approach. The cases provide an opportunity to review the spectrum of presentations associated with enteric duplication in adults and the current literature on this topic.
Case reports
Case 1: foregut duplication
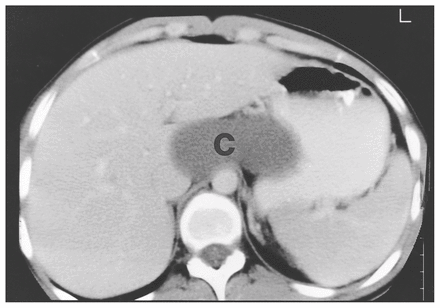
A 22-year-old woman was admitted with epigastric pain, squeezing in nature, that had been present intermittently for 2 years. Previously, chronic gastritis had been diagnosed, and she had been treated conservatively. The pain was aggravated by food and relieved by the application of pressure to the upper abdomen. The woman appeared thin, but physical examination was otherwise unremarkable. Hematologic assessment revealed a mild normocytic anemia, and ultrasonography demonstrated a cystic structure, 7 cm in dimension, in the gastrohepatic region. Contrast enhanced CT revealed a midline, fluid-filled mass between the liver and the stomach, causing indentation of the lesser curvature (Fig. 1). The patient underwent laparotomy, at which time a pear-shaped cystic mass was identified in the gastrohepatic ligament, which was intimately associated with the gastroesophageal junction. It was resected en bloc, and the point of attachment to the gastroesophageal junction was closed primarily. Pathological evaluation revealed a gastric duplication cyst. The patient’s postoperative course was uncomplicated, and at follow-up 13 months later she was asymptomatic.
Case 1. Contrast enhanced CT scan showing a midline fluid-filled mass (C) between the liver and the stomach. The lesser curve of the stomach is indented by the mass.
Case 2: midgut duplication
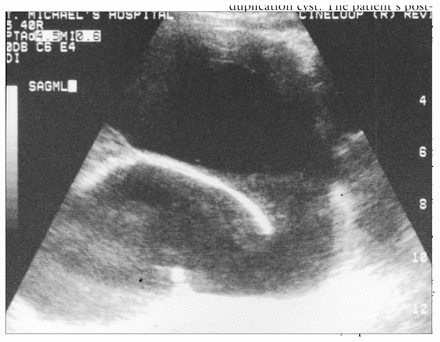
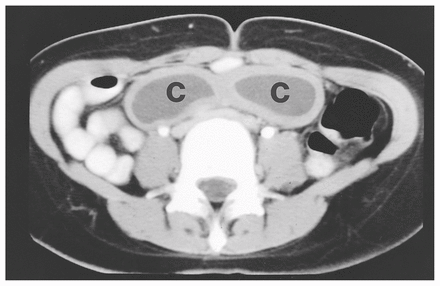
A 23-year-old woman was admitted with acute, severe periumbilical pain of 1 week’s duration. Similar but less intense pain had been present for 1 year, and she had remained systemically well. Irritable bowel syndrome had been diagnosed and treated conservatively. The pain was crampy, episodic and associated with abdominal distension and nausea. Examination revealed a tender, 10-cm mobile mass in the right lower quadrant of the abdomen. Ultrasonography revealed a fluid-filled, septate mass in that location, measuring 9 cm in maximum dimension (Fig. 2). Contrast enhanced CT demonstrated a bilobed, fluid-filled structure, straddling the patient’s midline (Fig. 3). At laparotomy, a 7-cm enteric duplication of the mid-ileum was diagnosed and was resected along with adjacent small bowel. Pathological examination of the resected specimen confirmed the diagnosis of duplication of the small bowel. The patient’s postoperative course was uncomplicated, and at follow-up 18 months later she was asymptomatic.
Case 2. Ultrasonogram in the sagittal plane showing a fluid-filled mass with a septum and solid debris layering posteriorly.
Case 2. Contrast enhanced CT scan showing 2 fluid-filled locules (C) of the mass straddling the patient’s midline.
Case 3: hindgut duplication
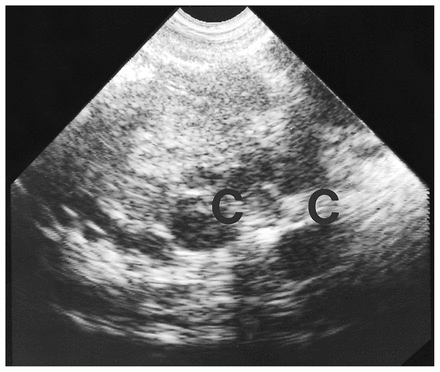
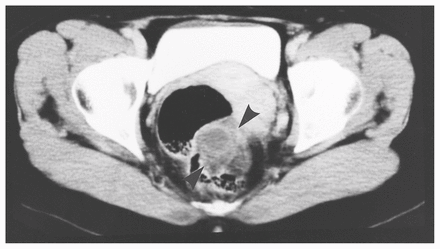
A 44-year-old woman was admitted with a 12-month history of the sensation of incomplete rectal emptying. Initially, constipation had been diagnosed and managed conservatively. The symptoms had become intolerable in the month before admission and were associated with lower back pain. There were no other symptoms. Digital rectal examination revealed a smooth, well-circumscribed cystic mass situated at the tip of the examining finger on the posterior rectal wall. Transrectal ultrasonography demonstrated a heterogeneous mass between the rectum and sacrum (Fig. 4). CT suggested that the mass had an intimate association with the rectum (Fig. 5). At operation, through a parasacral approach, the lesion was identified attached to the posterior wall of the rectum. It was resected en bloc, and the rectal mucosal defect was closed primarily. Pathological examination revealed a duplication cyst containing rectal mucosa. The patient’s postoperative course was smooth, and at follow-up 9 months later she was asymptomatic.
Case 3. Transrectal ultrasonogram, transverse view of the mass, shows 2 cystic spaces (C). In this plane the mass is mainly solid.
CT scan at another transverse plane showing the mass (arrowheads) posterior to a dilated rectum. The mass contains several hypodense areas and is mainly cystic at this level.
Discussion
The above cases provide an opportunity to review the current literature regarding the pathologic and embryologic features and management of enteric duplications in the adult.
Enteric duplications by definition comprise a group of lesions that contain a smooth-muscle wall and enteric mucosa and are found only on the mesenteric border of the intestine.1 These features differentiate intestinal duplications from the more common Meckel’s diverticulum, which is found on the antimesenteric border, and with other intra-abdominal cystic processes, which are described in Table I. Duplications may be found on intestine of foregut, midgut and hindgut derivation, although the midgut variety is the most common.2,3 In addition to an abdominal location, duplications are also seen in the chest, where they generally arise from the esophagus. In the majority of cases, some degree of communication exists between the duplication and the adjacent lumen, varying between a large opening and a thin fibrous strand.4 Twenty percent to 30% of lesions contain heterotopic mucosa, which is usually gastric.3,5 There were no associated anomalies in the current series, although vertebral and genitourinary defects may be encountered.
Differential Diagnosis of Intra-abdominal Cysts Based on Histologic and Radiologic Features
An accurate distinction can usually be made between enteric duplications and other intra-abdominal cystic processes with the combined use of ultrasonography and CT (Table I).4 An emerging diagnostic technique that may provide additional diagnostic information is endoscopic ultrasonography, which has been used in the investigation of foregut lesions.6 It is of note that before the current era of accurate cross-sectional radiologic evaluation, a correct diagnosis of enteric duplication was seldom established preoperatively. This is because neither plain radiography nor intraluminal contrast radiography accurately identifies either the communication between the duplication and the adjacent bowel or mechanical displacement of the bowel by the enteric cyst.4,7
An understanding of the embryologic development of enteric duplication may provide further insight into its clinical presentation. The “enteric bud” theory of Lewis and Thyng states that buds of intestinal epithelium protrude into the lamina propria and enlarge to establish a connection with the bowel lumen.8 The “solid lumen theory” of Bremer postulates that the rapidly enlarging embryonic gastrointestinal tract outgrows the celomic cavity and causes an accumulation of intestinal epithelial cells that then vacuolize, coalesce and form the lumen of the intestinal duplication.9 Ravitch theorized that hindgut duplication represents partial twinning,10 whereas others have postulated that an adhesion in the presomite embryo between the ectodermal and entodermal germ layers results in splitting or deviation of the notochord, which leads to alimentary duplications in association with vertebral anomalies.11 Although superficially attractive, each of these theories falls short in explaining both the presence of heterotopic mucosa and the consistent mesenteric location.
The clinical presentation of enteric duplication in adults is variable and has undergone a transition with time. In the past, patients were operated on with complications of these lesions, such as bleeding and perforation.7 Today, as described in the current study, the diagnosis is made after abdominal CT or ultrasonography for persistent, often mild, gastrointestinal symptoms. This symptom complex is dictated chiefly by the location and size of the lesion. For example, foregut duplications are most commonly associated with epigastric pain and dysphagia, the latter due to impingement of the cystic mass on the esophageal lumen (case 1).12 In foregut duplications, there have been isolated reports of heterotopic pancreatic tissue, which increases the morbidity,13 and rare instances in which these lesions communicated with the pancreatic duct or biliary tree.14,15 Duplications of the midgut are generally associated with abdominal pain; obstruction and intussusception have been reported (case 2).16 The presence of heterotopic mucosa, which is most commonly gastric, predisposes to bleeding or perforation.7,17 Duplications of the hindgut may be seen in association with intestinal obstruction or tenesmus (case 3) and may be confused with a colorectal neoplasm.
Once the diagnosis of an enteric duplication is made, surgical correction is warranted for 3 reasons. First, the majority of patients require surgical intervention for the relief of symptoms. 12 Second, surgical treatment removes the otherwise persistent risk of perforation and bleeding caused by heterotopic mucosa in the unresected cyst.5 Third, there have been reports of the development of carcinoma in the lining of cyst.18,19 Orr and Edwards18 reviewed 10 cases of neoplasia developing in duplication cysts. In this series, the most common location was the large intestine, and the most frequent cell type was adenocarcinoma.
The goals of surgery are to completely remove the lesion and associated heterotopic mucosa without jeopardizing the function of the remaining bowel. The complexity of the surgical procedure required is extremely variable and depends on both the size and location of the duplication. For instance, our foregut and midgut cases (cases 1 and 2) were amenable to complete resection and represent the most common scenario. The hindgut duplication in case 3 was managed via the parasacral route, which we felt would be less morbid than operating through the anterior abdominal wall. Various strategies have been used for lesions that are not amenable to complete resection. The classic treatment for complex duodenal duplications is cystoduodenostomy, 20 involving the formation of a large communication between the duplication and the adjacent bowel. Long ileal duplications, whose length precludes complete resection, have been treated by segmental stripping of the mucosa through a series of transverse incisions into the seromuscular wall.21 In general, such strategies are rarely required, and the commonest scenario involves a straightforward resection of the duplication along with the attached bowel.
In conclusion, through a series of case reports we have described the spectrum of disease associated with duplication of the foregut, midgut and hindgut in adults. The case histories emphasize the similarities between these relatively rare lesions and other, more common surgical disease processes. This study illustrates the importance of adequate investigation for refractory abdominal pain, and the excellent outcome that may be obtained in appropriately treated adult patients with enteric duplication.
- Accepted July 7, 1996.