Abstract
Because the clinical signs and symptoms of cardiac hydatid cyst are nonspecific and highly variable, this disease may be difficult to diagnose. In this report, the cases of 3 patients with cardiac hydatid cysts located in the subepicardium are presented.
In 2 of the patients the diagnosis was erroneous, and they were operated on for pulmonary hydatid cyst and acute abdomen in provincial hospitals before transfer to our institution. One patient who had an intrapericardially ruptured cardiac hydatid cyst presented with the clinical features of an acute abdomen. A patient with 4 subepicardial cysts had undergone surgery for a cardiac hydatid cyst 10 years before presenting at our hospital. The perforated cyst in this patient was excised while the heart was beating; the 2 other patients were operated on with extracorporeal circulation.
The epidemiologic and pathologic features, clinical presentation, complications, diagnostic methods and treatment of cardiac hydatid cyst are discussed in detail in the light of pertinent literature.
Cardiac echinococcoses were first documented and reported by Williams in 1936.1 Although an attempt to treat cardiac hydatid disease by operating was made in 1921 by Marter and de Crasping, the first successful surgical intervention was performed by Long in 1932. By 1964 only 42 successfully treated cases had been reported.2 In 1962 Arturcio and associates3 reported the first patient in whom a hyatid cyst was excised during open-heart surgery.
When a cardiac hydatid cyst is suspected, diagnosis can be confirmed using a 2-dimensional echocardiography, computed tomography (CT) and magnetic resonance imaging (MRI). But clinical signs and symptoms are generally nonspecific and sometimes, as in 2 of the patients reported here, may be misleading.
This article describes the diagnostic problems and surgical excision of cardiac hydatid cysts located in the subepicardium in 3 patients .
Case reports
Case 1
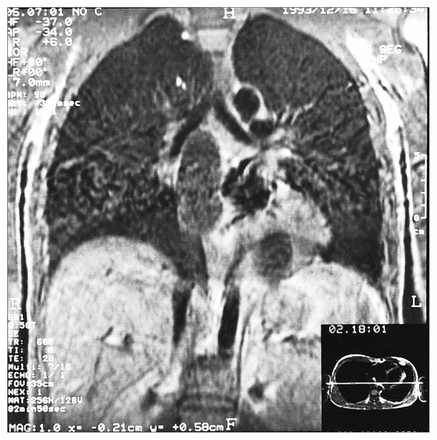
A 26-year-old man was admitted to hospital suffering from fatigue, shortness of breath, palpitations and retrosternal dull aches. He had been operated on elsewhere for a cardiac hydatid cyst 10 years previously. A left anterolateral thoracotomy incision was evident on examination. His blood pressure was 90/60 mm Hg, his pulse rate was 110 beats/min and his heart sounds were normal. There was a pericardial friction rub over the sternum. A chest x-ray film showed slightly increased cardiothoracic index. The electrocardiogram (ECG) exhibited inverted T waves on precordial leads. On echocardiographic investigation multiple cystic structures were seen growing in the pericardial sac and originating from the posterior wall of the left ventricle, the posterior interventricular sulcus and the left and right atrial walls. MRI of the heart showed the same cystic structures (Fig. 1). No disease was suggested by abdominal ultrasonography. A blood test showed an erythrocyte sedimentation rate of 15 mm/h, a leukocyte count of 11.5 × 109/L with 9% eosinophils, and a Casoni intradermal test gave a positive result.
Magnetic resonance image of the heart showing multiple cystic structures growing in the pericardial sac and originating from the posterior wall of the left ventricle, the posterior interventricular sulcus, and the left atrial and right atrial walls.
The operation was performed through a median sternotomy. The pericardium was opened. After the fibrous adhesions that were remnants of the first operation were divided, cardiopulmonary bypass was initiated. Moderate hypothermia (28 °C) was used, and myocardial protection was provided by cold potassium cardioplegia. At exploration, 4 cystic structures protruded into the pericardial sac, originating from the right atrial wall, the posterior wall of the left atrium, the posterior interventricular sulcus and the posterior wall of the left ventricle. The cysts measured 2 × 3 cm, 1 × 2 cm, 4 × 5 cm and 8 × 10 cm, respectively.
Beginning from the most superficial (right atrial) cyst, a cystectomy was performed as follows: sponges and towels moistened with a 1% povidone iodine solution were applied around the operative field to prevent adjacent structures from being contaminated by scolices. After the fluid content of the cyst was aspirated, the povidone iodine solution was instilled into the cyst. The cyst was then opened and the germinative membrane removed. The empty cavity covered with a fibrous pericystic layer was washed with the povidone iodine solution. The pericystic tissue was resected as widely as possible and the cavity was left open. The same procedure was carried out on the 3 other cysts. At the end of procedure, the pericardial sacs were thoroughly washed with the povidone iodine solution. The postoperative period was uncomplicated and the patient was discharged 2 weeks postoperatively.
Case 2
A 38-year-old woman was transferred from a provincial hospital where she had been admitted with a cough, dyspnea, thoracic pain radiating to her back, fever and headache. She had had 2 operations for a hydatid cyst of the liver and lung, 8 and 12 years before respectively. A chest film showed a spherical lesion in the left paracardiac zone. On the basis of her medical history and radiologic findings, a diagnosis of left pulmonary hydatid cyst was made. At operation through a left posterolateral thoracotomy incision, the lung was found to be free of any hydatid disease. A cystic mass nearly 10 cm in diameter was found at the apex of the heart.
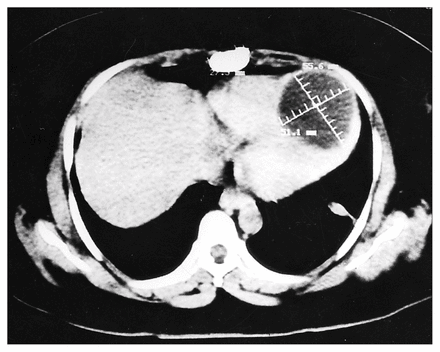
With a diagnosis of cardiac hydatid cyst the patient was transferred to our clinic. The chest film showed an increased cardiothoracic index and there was a bulge on the left border of the heart. An ECG showed inverted T waves in all leads. Echocardiography, MRI and CT revealed a cystic mass, 6 × 6 cm, growing in the pericardial cavity from the cardiac apex (Fig. 2). Findings on abdominal ultrasonography were normal. Seven days after the first intervention (a thoracotomy), the patient underwent cardiopulmonary bypass surgery with cold potassium cardioplegia and moderate hypothermia. The cyst was 6 cm in diameter and located in the cardiac apex (Fig. 3). A cystectomy was performed, as described in case 1. Her postoperative course was uncomplicated.
Computed tomography scan showing a 6 × 6-cm cystic mass growing in the pericardial cavity from the cardiac apex.
Operative view showing a cyst 6 cm in diameter located in the cardiac apex.
Case 3
An 18-year-old man was admitted to a provincial hospital after the sudden onset of abdominal pain, nausea, vomiting, dispnea, generalized rash and fever, which led to a diagnosis of an acute abdomen. An immediate laporotomy, however, did not reveal any abnormal findings.
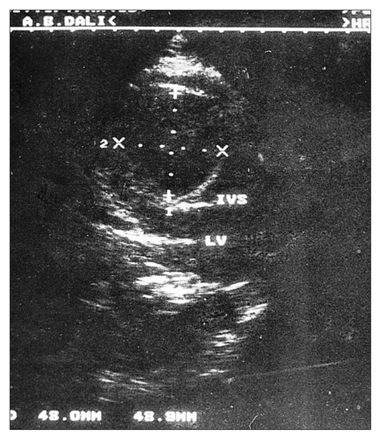
Because respiratory distress developed early in the postoperative period, the patient was transferred to the intensive care unit. On admission, he was in critical condition with acute respiratory distress. Mechanical ventilation was applied immediately. His blood pressure was 70/40 mm Hg, his pulse rate was 125 beats/min and his neck veins were engorged. The central venous pressure was 15 cm H2O and his leukocyte count was 18.0 × 109/L with 13% eosinophils. A chest film showed marked cardiomegaly suggestive of an advanced pericardial effusion and compatible with clinical signs of tamponade. Pericardiocentesis was performed and 1500 mL of seropurulent fluid was aspirated. After the pericardiocentesis, his arterial blood pressure increased to 110/60 mm Hg and his central venous pressure decreased to normal levels. Echocardiography and MRI performed 5 days after he was admitted (as soon as he was weaned from the ventilator), revealed an 8 × 10-cm cyst covered by a dense layer of pericystic tissue, located between the diaphragmatic wall of the heart and the pericardium (Fig. 4).
Echocardiogram showing an 8 × 10-cm cyst located between the diaphragmatic wall of the heart and the pericardium.
At operation, the pericardium was opened through a median sternotomy incision. Adhesions between the anterior surface of the heart and the pericardium were divided. Between the inferior ventricular wall and the pericardium a large cystic mass with a fibrous capsule was identified. On needle aspiration, approximately 175 mL of purulent fluid was removed. The fibrous capsule was then opened, the germinative membrane of a ruptured hydatid cyst removed and the operation completed as previously described. In this patient, extracorporeal circulation was not used and the excision was made while the heart was beating. The postoperative course was uncomplicated.
Discussion
Hydatid disease is endemic in many sheep-farming areas of the world, notably Mediterranean countries, the Middle East, South America and Australia. 4 The incidence in the Turkish population is 1:20 000.5
Humans serve as an intermediate host for the larval stage (hydatid cyst) of Echinococcus granulosus when the ova from contaminated dog feces is accidentally ingested. The parasites penetrate the gastrointestinal mucosa, enter the portal circulation and are carried to the liver where the majority become trapped. Pulmonary capillaries act as the second filter for parasites that escape the liver. As a result, the liver is affected in more than 65% of people who suffer from hydatid disease, and the lungs are affected in 25%. Very few embryos escape these 2 filters and most of those that do become trapped in the myocardium, although it has been proposed that the parasites may reach the heart through lymphatic channels. Therefore, hydatid cyst of the heart is rare (incidence between and 0.05% and 2%).1,6,7 In a series of 577 patients with hydatid disease in Turkey, the incidence of cardiac hydatid cyst was less than 0.1%.8 In our institution between 1978 and 1995, 418 patients were operated on for pulmonary hydatid cyst; cardiac cysts (3 patients) made up 0.7% of these.
Sixty percent of cardiac hydatid cysts are located in the left ventricle, presumably because of the richer coronary circulation. In decreasing order, the next most frequent locations are the right ventricle (15%), the interventricular septum (9%), the left atrium (8%), the right atrium (4%) and the interatrial septum (2%).6,9,10
In 2 of our patients (cases 2 and 3) the cysts were located in the left ventricle. The third patient had undergone surgery to remove a cardiac hydatid cyst 10 years previously; the 4 myocardial cysts that subsequently developed were probably the result of contamination during the first operation.
The embryo embedded in the myocardium enters the larval stage and becomes a real cyst in 1 to 5 years. The rate of growth of any particular cyst varies and the diameter can increase from a few millimetres to approximately 5 cm per year.1,2,4,7,11,12 Because the myocardium is not elastic and resists expansion, the growth of cardiac hydatid cysts is slower than that of pulmonary cysts. But, as in our patients, cysts located in the subepicardium can grow more easily toward the pericardial sac and may reach diameters of 8 to 10 cm.
During the growth period the myo cardial cyst may rupture spontaneously or degenerate and calcify. Rupture may occur within the pericardial space or into the cardiac chambers. Intracardiac ruptures have been reported to occur in 39% of patients.10 Rupture into the cardiac cavities may be followed by secondary cysts either in the lungs or the various other organs that are supplied by the systemic circulation, and of these 55% occur in the brain.2,12 Intracardiac rupture may also result in germinative membrane embolization in the pulmonary and the systemic circulation.13–18 A cardiac hydatid cyst may also lead to the formation of intracardiac thrombus.19,20 If cystic fluid enters the circulatory system, an anaphylactic reaction may occur. 2,12 About 10% of primary cardiac hydatid cysts rupture into the pericardial sac, where secondary cysts then form.21,22 Intrapericardial perforation may also result in tamponade or acute pericarditis,22,23 which may progress to chronic constrictive pericarditis.23–26 In 1 of our patients (case 3), an inflammatory reaction occurred when the cyst was perforated, resulting in the formation of an encapsulated abscess, walled off by inflammatory adhesions between the heart and pericardium around the perforated cyst.
Probably only 10% of patients with cardiac hydatid cysts have clinical manifestations. 7 Signs and symptoms are extremely variable and directly related to the location and size of the cyst.7,27 As a result, diagnosing the disease may be difficult. However, in areas where the disease is endemic, the patient’s medical history could be very helpful.26,28,29
A chest x-ray film can be used to diagnose the disease in many patients who are asymptomatic. In countries where hydatid disease is common, the signs of pulmonary hydatid cysts that can be detected radiologically are generally well known. Occasionally a cardiac cyst may be misdiagnosed as a pulmonary hydatid cyst. In fact a pulmonary hydatid cyst was diagnosed in 1 of our patients (case 2) who was operated on before being transferred to our clinic. A radiographic mass adjacent to the left ventricle should signal the possibility that a cyst is a cardiac hydatid cyst. In such cases, echocardiography, CT and MRI will be helpful in reaching a definitive diagnosis.
A common symptom is precordial pain, which is usually due to the compression of the coronary arteries and myocardium by the cystic masses and ischemia. Anginal complaints may masquerade as coronary heart disease. 7,30–32 Precordial or retrosternal pain may also be due to acute pericarditis that can occur after an intrapericardial rupture.1,12,22,23 In patients with acute pericarditis, the pain may radiate to the abdomen and symptoms may closely mimic an acute abdomen. A diagnosis of an acute abdomen was made in case 3 and a laporotomy performed before he was transferred to our hospital with respiratory distress. In this patient, acute pericarditis and cardiac tamponade occurred after an intrapericardial rupture of the cardiac hydatid cyst. An intrapericardial rupture may also lead to hypersensitivity reaction manifested by a generalized rash, urticaria, intense pruritis and fever.2,23,25
Occasionally a valvular syndrome clinically simulating mitral, pulmonary or tricuspid stenosis or regurgitation may occur.15,20,33–35 Involvement of the ventricular septum may result in right1,10,14 or left36,37 outflow tract obstruction or a variety of conduction defects.14,38–40
Germinative membrane embolization secondary to intracardiac rupture of the cyst causes symptoms of pulmonary13,15,29 and systemic embolization. 16–18 Intracranial embolization can be fatal.18 Sudden rupture of the cardiac wall, acute tamponade, arrhythmias and hypersensitive reaction with severe anaphylactic shock secondary to intracardiac rupture are the other complications causing death. Twenty percent of patients die of the disease after having no signs or symptoms of a cardiac hyatid cyst.1,3,10,14
An ECG often displays T-wave changes, loss of QRS voltage and a pathologic Q wave. Arrhythmias are occasionally seen. Atrioventricular conduction defect or bundle branch blocks may be seen when the interventricular septum is affected. P-wave abnormalities occur when the atrium is affected. 27,33 In our 3 patients, T-wave inversions were the prominent findings.
Eosinophilia, a positive Casoni intradermal test and serologic tests including complement fixation (Weinberg) and hemagglutination are not specific for hydatid disease. Because of both false-positive and false-negative results, these tests are not very helpful in diagnosing cardiac hydatid cysts.4 In a large series of patients with pulmonary hydatid cysts in Turkey,8 the Casoni intradermal test was positive in 50%, the Weinberg test elicited a positive result in 30% and eosinophilia was evident in 28%. In 2 of our patients, eosinophilia was present (9% and 13% eosinophils).
On chest radiography, the typical findings are localized deformities of the cardiac silhouette with a bulge of the cardiac border. A calcification may also be helpful in diagnosis.1,2,27
Two-dimensional echocardiography, CT and MRI are noninvasive diagnostic methods and are very helpful for a differential diagnosis; they can display the location and size of cysts. The diagnostic capacity of 2-dimensional echocardiography is excellent and it is the method of choice to detect cardiac hydatid cysts. It does not involve radiation and is relatively inexpensive.1,7,41–44 Nevertheless, CT and MRI are important diagnostic tools, especially in establishing a differential diagnosis.28,43–46
Catheterization and angiography are also helpful when diagnosing cardiac hyatid cysts. Coronary angiography is a useful diagnostic tool when examining patients with angina secondary to compression of the coronary artery by the cyst.7,30,36,47
Although drug therapy with mebendazole, and more recently with albendazole, has occasionally been reported, 44,48,49 the treatment of choice for cardiac hydatid cysts is surgical excision. The surgical results have been good with very few complications; many patients have made a complete recovery and are given an excellent prognosis.7,8,30 Since 1962 surgical interventions have been generally performed under extracorporeal circulation, 1,3,7,30 although superficially located cysts can be successfully removed while the heart is beating. Cardiopulmonary bypass was used in 2 of our patients. In the third patient, in whom the cyst was perforated and infected, the cyst was excised while the heart was beating.
Late sudden deaths after surgery are very rare and are thought to be the result of either rupture of cysts that were not discovered during surgery or secondary cysts developing as a result of leakage from the primary cyst.50
Because of the risk of contaminating adjacent structures, which might then develop secondary cyst scolices, the cystic fluid should be deactivated and leakage of cystic fluid prevented during the operation. To prevent contamination we applied sponges and towels moistened with 10% povidone iodine solution around the operative field. The same solution has been injected into the cyst to deactivate the scolices before their removal. For deactivation, different larvacidal agents such as 30% hypertonic saline, 2% formalin, 5% silver nitrate or 1% iodine solutions have also been used.7 To reduce the incidence of recurrence, drug therapy with mebendazole or albendazole as an adjunct to surgery has been used.4,20,30,36
In conclusion, cardiac hydatid cysts are life threatening. A routine follow-up with echocardiography of patients with hepatic or pulmonary hydatid disease will help detect cardiac involvement early. Even asymptomatic cardiac hydatid cysts should be surgically excised.
- Accepted September 25, 1996.