Abstract
Objective: To evaluate the role of chemotherapy with a combination of doxorubicin (adriamycin) and cisplatin in high-grade, nonosteogenic, non-Ewing’s sarcoma (non-OSA) of bone.
Design: A case series comparison with a literature-derived control group.
Setting: A university-affiliated tertiary care centre.
Patients: Thirty patients with a diagnosis of non-OSA. Of these, 8 had low-grade disease (grade 1 or 2) and 22 had high-grade disease (grade 3). Eleven of the 22 with high-grade disease had malignant fibrous histiocytoma. Seventeen patients with nonmetastatic high-grade non-OSA were compared with a literature cohort of 37 patients who met the eligibility criteria of nonmetastatic, high-grade non-OSA treated with surgery, with or without radiotherapy. The mean follow-up was 25.2 months.
Interventions: Eight patients with low-grade tumour underwent surgery alone; 22 patients with high-grade tumour underwent surgery and 6 courses of adriamycin (75 mg/m2 every 3 weeks) and cisplatin (100 mg/m2 every 3 weeks).
Main outcome measures: Disease-free survival and overall survival in those with high-grade tumours treated with or without chemotherapy.
Results: Of 8 patients who had low-grade tumours and underwent surgery alone, 3 had systemic relapse. Of the 22 having high-grade tumours, 4 did not receive chemotherapy because of age and comorbid conditions. Of the other 18, 13 received 3 courses of chemotherapy preoperatively and 3 courses postoperatively, 4 received all 6 courses postoperatively and 1 received all chemotherapy preoperatively to treat metastatic disease. In the 17-patient cohort used for comparison with the literature control group, disease-free survival was 57% at a mean follow-up of 25.6 months and overall survival was 57% at a mean follow-up of 30.1 months. In the control group, disease-free survival was 16% at a mean follow-up of 20.9 months and overall survival was 26% at a mean follow-up of 29.9 months. These differences are significant: p = 0.0000, χ2 = 41.61 for disease-free survival and p = 0.0000, χ2 = 46.49 for overall survival.
Conclusions: The findings of this study support the use of adjuvant chemotherapy in patients with high-grade non-OSA, in whom malignant fibrous histiocytoma was the predominant histologic subtype.
Nonosteogenic, non-Ewing’s sarcoma (non-OSA) of bone is an uncommon diagnosis, the tumour accounting for less than 15% of primary malignant mesenchymal bone tumours.1–4 Non-OSA includes the histologic diagnoses of malignant fibrous histiocytoma, fibrosarcoma, leiomyosarcoma, dedifferentiated chondrosarcoma, neurofibrosarcoma, spindle cell sarcoma of bone not otherwise specified and angiosarcoma. All these lesions are characterized by cytologically high-grade regions of spindle cell sarcoma without evidence of osteoid formation. The natural history of these sarcomas is characterized by aggressive local tumour growth associated with bone destruction and a high risk of metastasis5,6 despite resection or amputation of the primary tumour.
The extremely low incidence and heterogeneous nature of these tumours has precluded the development of randomized clinical trials or standard therapy protocols. Treatment options reported in the literature have included various combinations of surgery, radiotherapy and chemotherapy for each subtype of non-OSA.7–10
Although the literature demonstrates no definitive evidence of the efficacy of chemotherapy on the overall survival and disease-free survival of patients with non-OSA of bone, a number of small retrospective studies have been published offering support for the use of neoadjuvant and adjuvant chemotherapy in these diseases. Beginning in 1989, patients presenting to our centre with high-grade non-OSA of bone were treated with a standard protocol comprising complete surgical resection and doxorubicin (adriamycin)-cisplatin chemotherapy. In evaluating the results of this treatment protocol, we compared the outcome with that in a control group derived from the literature.
Patients and methods
From the prospective database maintained at the University of Toronto Musculoskeletal Oncology Unit we identified 30 patients (18 men, 12 women) treated for non-OSA of bone between March 1989 and August 1995. The mean age of the group was 45.5 years (range from 17 to 83 years). Two patients (cases 12 and 21 in Table I) had lung metastases at the time of presentation.
Patients Having Nonosteogenic Sarcoma of Bone, Which Was Diagnosed Between March 1989 and August 1995
Initial assessment included a history-taking, physical examination and tumour staging for all patients. Staging studies included plain radiography, MRI or CT, or both, of the local tumour site, total body bone scanning, chest CT and diagnostic biopsy of the tumour. All biopsies were characterized by an expert musculoskeletal pathologist as grade 1, 2 or 3 out of 3. Grade 3 tumours were considered high grade and grades 1 and 2 were considered low grade. A standard treatment protocol based on the histologic tumour grade was then followed for all patients.
Patients having a diagnosis of low-grade non-OSA were treated by excision of the local tumour; those with high-grade non-OSA were treated by 6 courses of chemotherapy, which consisted of adriamycin (75 mg/m2 every 3 weeks) and cisplatin (100 mg/m2 every 3 weeks) if their age and general health permitted.
When the lesion was diagnosed as high grade on the initial biopsy, 3 courses of chemotherapy were administered over the 6 weeks before surgical excision of the primary lesion (neoadjuvant treatment) and 3 further courses were given postoperatively. If the sarcoma was diagnosed as low grade on the initial biopsy and subsequently upgraded after examination of the resected specimen, 6 courses of chemotherapy were given postoperatively. Patients having limited numbers of pulmonary metastases after chemotherapy and resection of the primary lesion underwent thoracotomy and resection of the metastatic lesions if their condition permitted.
Routine follow-up was conducted every 3 months for the first 3 years, then every 4 months for 2 more years, and at 6-month intervals thereafter. The follow-up consisted of physical examination and chest radiography at each visit plus chest CT every 6 months for the first 2 years.
A subset of patients with high-grade non-OSA treated with chemotherapy and surgery served as the “treatment” group for a comparison of outcome against a cohort of high-grade non-OSA patients treated without chemother apy (control group) drawn from the literature. The reason for drawing a control group from the literature relates to the rarity of this disease. We could not develop a control group from our patient cohort because we had selected the most high-grade tumours for chemotherapy based mainly on the histologic grade of the tumour. This would bias a study strongly in favour of the control group because the lower grade tumours would be less likely to metastasize. The best control group therefore would be patients drawn from the literature and matched to group entry criteria.
A control group of patients with non-metastatic, high-grade non-OSA treated by surgery, with or without radiotherapy, was identified from the literature by a search of the MEDLINE and CANCERLIT databases from January 1976 to May 1996. The computer search was limited to English language and human studies. The MeSH headings “fibrosarcoma,” “angiosarcoma,” “hemangioendothelioma,” “leiomyosarcoma,” “dedifferentiated chondrosarcoma” and “histiocytoma, fibrous” were combined with “bone neoplasms” in 6 separate searches. Studies that addressed the treatment of non-OSA with surgery alone or in combination with adjuvant chemotherapy were retrieved for complete review. Cited abstracts were reviewed and all that clearly did not meet the eligibility criteria were discarded, but all citations without abstracts were kept for further review. The references of articles were hand searched to identify further relevant articles. Attention was paid to the centre where the study was done, the histologic type of the disease and individual patient details to ensure that the same patient was not included twice.
From the group of eligible articles, those containing case descriptions or tables, or both, summarizing the clinical findings of each patient in the study series were examined. These articles were used to assemble a literature cohort group that met the following criteria: nonmetastatic, high-grade non-OSA, treated with surgery, with or without radiotherapy but without chemotherapy. Individual case reports were excluded. For the case to be included in the literature cohort, the time to metastases, death and last follow-up as well as the patient’s status had to be given from a discernible time zero of surgery or first treatment. Furthermore, surgery had to be at least marginal, with no gross tumour remaining after resection, in keeping with the procedures performed at our centre.
Descriptive statistics including means, standard deviations and frequencies were used as appropriate. Disease-free and overall survival rates were calculated by the Kaplan–Meier method.11 In comparing the subset of patients having nonmetastatic, high-grade non-OSA who were treated by chemotherapy with the literature control cohort, independent t-tests and χ2 analyses or Fisher’s exact tests were used to evaluate the similarity of the groups with respect to age, gender, frequency of histologic type and type of surgery. A p value < 0.05 was considered significant. Disease-free survival and overall survival for the “treatment” and “control” groups were compared according to the log-rank test.12
Findings
Complete data were available on all 30 patients identified by the University Musculoskeletal Oncology Unit database for a mean follow-up of 25.2 months (range from 12 to 82 months). There were pre-existing bone abnormalities in 5 of these patients: previous giant cell tumour (cases 16 and 18 in Table I), multi ple osteochondromas (case 2), non-ossifying fibroma (case 30) and Rothmund–Thompson syndrome (case 8). Two patients had a sarcoma arising in an area of a previous irradiation field for treatment of lymphoma (case 6) and melanoma (case 7).
Twenty-two patients had high-grade lesions; the remaining 8 had low-grade lesions. Of the 22 with high-grade lesions, 11 had malignant fibrous histiocytoma, 3 each had dedifferentiated chondrosarcoma, leiomyosarcoma and fibrosarcoma and 2 had high-grade spindle cell sarcoma not otherwise specified. Of the 8 with low-grade lesions, 3 had leiomyosarcoma, 2 had angiosarcoma and 1 each had neurofibrosarcoma, fibrosarcoma and malignant giant cell tumour.
Nine of the 30 patients had amputations, 2 after attempted limb salvage. Of these 2 amputations, 1 was required because of an involved margin after resection and reconstruction (case 26) and 1 was secondary to a chronic postoperative infection (case 25).
Of the 8 patients with low-grade tumours treated by surgical excision alone, 3 suffered systemic relapse, 2 of them after experiencing local recurrence, and 5 were disease free at last follow-up.
Eighteen of the 22 patients having high-grade non-OSA tumours were treated by chemotherapy and surgery; the other 4 did not receive chemotherapy because of age or comorbid conditions. Of the 18 patients, 13 received 3 preoperative and 3 postoperative courses of chemotherapy, 4 received 6 postoperative courses when the final pathological examination revealed high-grade disease and 1 received 6 preoperative courses in an attempt to achieve regression of metastatic pulmonary disease. Consequently, the “treatment” group for comparison with the control literature-derived group consisted of 17 patients with nonmetastatic, high-grade non-OSA treated by surgery and chemotherapy.
Comparison of the treatment and control groups
The mean age of the 17 patients (13 men, 4 women) in the treatment group was 40.3 years. Nine patients had malignant fibrous histiocytoma and 2 each had leiomyosarcoma, dedifferentiated chondrosarcoma, fibrosarcoma and high-grade spindle cell sarcoma not otherwise specified. Three patients required amputation.
For the control group, the literature search identified 984 citations; 85 were kept for further review. From these, a group of 37 patients who met the eligibility criteria was assembled. Although the search was conducted between January 1976 and May 1996, all eligible cases were identified from studies published between 1982 and 1996.2,3,9,13–18 The inclusion criteria were such that not all patients from each study were included. Table II identifies the specific cases included in the control group from each published study.
Literature Cohort of Patients With High-Grade Nonosteogenic Sarcoma of Bone, Grouped According to Tumour Type
The mean age of the control group (24 men, 13 women) was 51.4 years. Twenty-two patients had malignant fibrous histiocytoma, 8 had leiomyosarcoma and 7 had dedifferentiated chondrosarcoma. Thirty-five were treated by surgery alone, 2 had surgery and radiotherapy. Twenty-eight had an amputation.
Table III compares the treatment and control groups. Patients in the control group were slightly older than those in the treatment group (t = 2.00, p = 0.03), but there were no gender differences between the 2 groups (p = 0.59). The histologic types were similar (malignant fibrous histiocytoma versus other, χ2 = 1.163, p = 0.202), but a significantly greater proportion of patients in the control group had an amputation (p < 0.0002).
Characteristics of Treatment and Control Groups of Patients With High-Grade Nonosteogenic Sarcoma of Bone
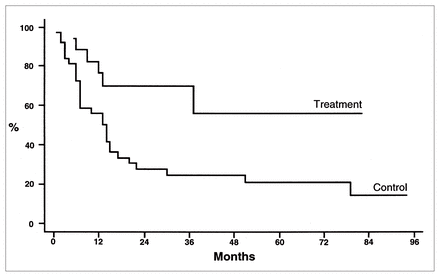
Disease-free survival in the treatment group was 57% with a mean follow-up of 25.6 months (range from 5.5 to 82 months) and in the control group was 16% with a mean follow-up of 20.9 months (range from 1 to 94 months) (Fig. 1).
Disease-free survival between chemotherapy (treatment) and no chemotherapy (control) groups of patients with high-grade nonosteogenic sarcoma of bone, calculated by the Kaplan–Meier method.
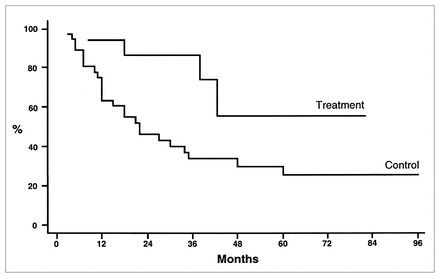
Overall survival in the treatment group was 57% with a mean follow-up of 30.1 months (range from 8.5 to 82 months) and in the control group was 26% with a follow-up of 29.9 months (range from 3 to 94 months) (Fig. 2). One patient in the treatment group (case 9) died of causes unrelated to the tumour at 19 months after surgery and was excluded from the analysis. These findings were significant: p = 0.0000 for disease-free and overall survival; χ2 = 41.64 and 46.49, respectively (log-rank test). It should be noted that time zero for the treatment group was the date of surgery, whereas time zero for the control group varied between the date of first treatment and the date of surgery. The net effect is a bias in favour of longer disease-free and overall survival in the control group, such that the differences in this study likely underestimate the effect of chemotherapy on both survival rates.
Overall survival in treatment and control groups, calculated by the Kaplan–Meier method.
Discussion
The efficacy of chemotherapy for high-grade bone sarcoma was initially demonstrated by the Pediatric Oncology Group randomized trial. This study, first published in 1986,19 demonstrated a marked therapeutic benefit for children and young adults receiving adjuvant chemotherapy for nonmetastatic disease. Other groups20–23 have confirmed these initial results, demonstrating that the historical systemic relapse risk of 70% to 90% was reduced to 10% to 35% in patients receiving multiagent chemotherapy.
As a result of these studies, all patients having osteosarcoma are currently treated with chemotherapy. However, the use of multiagent adjuvant therapy has not yet been accepted as standard therapy in non-OSA for 2 reasons. First, this disease occurs much less frequently than osteosarcoma, precluding the evaluation of the protocols even in multicentred randomized trials. Second, patients with non-OSA are generally older than patients with osteosarcoma and are less able to tolerate the intensive, toxic drug regimens that have been successful in osteosarcoma.
The drug regimen used for patients in the treatment arm of our study is identical to that used in an osteosarcoma treatment trial by the European Osteosarcoma Intergroup.23 This regimen is ideal for older patients who are less likely to tolerate the year-long protocols frequently used in the management of osteosarcoma.
The method used in this study, comparing a treatment group with a historical cohort obtained from the literature, is recognized as a less robust measure of clinical evidence than a randomized clinical trial, a case-control study or even a sequential cohort study obtained from the same treatment centre. However, given the rarity of non-OSA, comparison studies are extremely difficult to complete, even in multicentre groups. Indeed, given the results of this study and other small studies comparing outcome in treated and control groups, it is questionable whether it would be ethical to propose a trial with a control group of patients having non-OSA treated without chemotherapy.
Investigators from the Rizzoli Institute24 found that only 28% of patients treated for malignant fibrous histiocytoma with surgery alone were alive at 5 years. Other small series comparing the outcome of patients receiving adjuvant chemotherapy for malignant fibrous histiocytoma with historical controls at their same institutions found an advantage for patients treated with cytotoxic therapy.
In our series, several aspects should be critically evaluated. The treatment and control groups were gathered from the same clinical “era” of sarcoma therapy and were treated in clinical centres with experience in the management of primary bone cancer. The stage of disease at the onset of treatment was similar in the 2 groups since one of the criteria for inclusion in the control group was that patients had no evidence of disease on staging chest CT. The adequacy of surgery in the literature cohort was controlled by the inclusion criteria that at least marginal surgery had been undertaken. Indeed, the patients in the control group had undergone more radical surgery than those in the treatment group, a higher proportion (about 80%) having been treated by amputation.
The fact that more patients in the control group underwent amputation might be considered as a bias against that group since it is possible that these patients had larger, more advanced local disease than could be treated with limb-salvage surgery. It is impossible to refute this potential bias because there was inadequate documentation of tumour size in the literature to include this variable as an inclusion criterion for the control group. However, it is important to remember that the performance of limb-salvage surgery for high-grade bone sarcoma has developed in conjunction with the use of neoadjuvant chemotherapy to control the risk of local recurrence in the salvaged limb.
It is probable that limb salvage was considered to be an unsafe oncologic procedure in patients treated without chemotherapy in the control group. It is important to recognize that follow-up remains short in this study (mean follow-up under 3 years) and these results may degrade with time. However, follow-up in the control group was even shorter. Finally, most sarcoma metastases develop within 2 years of diagnosis of the primary tumour.
Both groups had equal proportions of patients with a diagnosis of malignant fibrous histiocytoma (9 of 17 in the treatment group and 22 of 37 in the control cohort). This proportion may be important because this tumour may be particularly responsive to chemotherapy. Most of the comparative1,7,14 and single-arm studies3,13,24– 26 reporting on the efficacy of chemotherapy in non-OSA have treated patients with malignant fibrous histiocytoma of bone.
In summary, patients presenting with possible bone sarcoma should undergo local and systemic staging followed by biopsy performed by the surgeon who will do the resection. If the diagnosis is high-grade non-OSA, we suggest that the patient should receive neoadjuvant chemotherapy followed by complete surgical resection of the lesion, using limb-salvage surgery if the examination and staging of the lesion suggest that this is a safe oncologic procedure. Although there is no definitive clinical evidence demonstrating the efficacy of chemotherapy in this disease, the sum of available evidence suggests that patients with these tumours have a very high risk of metastasis if treated by surgery alone. This study adds weight to the evidence in the literature that this prognosis can be improved by treatment with sarcoma chemotherapy.
- Accepted November 26, 1997.