Abstract
Objective: To determine if epidural anesthesia after the subcutaneous injection of low-dose unfractionated heparin (LDUH) in patients who undergo elective bowel surgery is safe with respect to hemorrhagic complications.
Design: A prospective cohort study.
Setting: Two hospitals affiliated with the Université de Montréal.
Patients: Fifty patients scheduled for elective bowel surgery.
Intervention: Subcutaneous injection of 5000 units of LDUH and elective surgery for colonic carcinoma, chronic diverticulosis or inflammatory bowel disease.
Main outcome measures: Activated partial thromboplastin time (APTT), anti-IIa and anti-Xa heparin levels measured before and 2 and 4 hours after injection of LDUH.
Results: In no case was the heparin anti-IIa or anti-Xa level higher than 0.20 U/mL, which is considered a significant detectable level of heparin.
Conclusion: LDUH given subcutaneously is not associated with significant detectable heparin levels, so epidural anesthesia should be safe when performed 2 hours after LDUH injection in patients who undergo general abdominal surgery in the absence of any other impairment of hemostasis.
Antithrombotic prophylaxis in high-risk patient groups is the most efficacious way to reduce thromboembolic complications and death rates secondary to pulmonary embolism.1,2 The incidence of venous thrombosis after general abdominal surgery varies from 3% to 50%, depending on the objective diagnostic test used.2 Relative risk reduction varies from 45% to 82%, depending on the method of thromboprophylaxis.2 The most impressive trials, including over 4000 patients given 5000 units of low-dose unfractionated heparin (LDUH) subcutaneously for prophylaxis 2 hours before surgery3,4 have demonstrated a statistically significant reduction of the relative risk of pulmonary embolism after general abdominal surgery. These studies define the “gold standard” method to be used for prophylaxis during abdominal surgery.3–5 A meta-analysis of 46 studies has demonstrated a slightly increased rate of hemorrhagic complications of 5.9% with standard un-fractionated heparin prophylaxis compared with 3.8% in a control group.6 This difference is not statistically significant and is outweighed by the important reduction in the death rate.
In recent years, epidural analgesia has become increasingly popular,7,8 because of its advantages over conventional methods of analgesia. These advantages include better pain control,9 benefits to the cardiovascular,10 pulmonary10–12 and gastrointestinal systems, 12 improved stress response13 and immune function, and a decreased risk of thromboembolism.11,14 A critical review demonstrates that the relative risk reduction of thromboembolism with regional anesthesia compared with general anesthesia is 50% lower in patients who do not receive prophylaxis. 14 However, this observation could not be extrapolated to patients receiving prophylaxis.14
To comply with accepted guidelines for subcutaneous heparin administration, the preoperative placement of the epidural catheter frequently occurs within 2 hours after the administration of heparin, and many anesthesiologists are concerned about possible hemorrhagic complications,15,16 particularly spinal hematoma.17,18 Although the rate of this devasting complication of epidural anesthesia is probably around 1:100 000,19 the majority of reported cases appear to be related to an underlying coagulation abnormality. More recently, the increasing use of low-molecular-weight heparin (LMWH) as thromboprophylaxis in high-risk orthopedic surgery has been associated with higher rate of spinal hematoma.20 Some anesthesiologists recommend delaying insertion of the catheter until 4 to 6 hours after the subcutaneous LMWH injection on the basis of evidence that therapeutic changes in coagulation are restricted to the first 4 hours after heparin injection.21,22 However, this practice is inconsistent with that suggested in large series in which the timing of standard unfractionated heparin administration for optimal venous thromboembolism prophylaxis is 2 hours before general abdominal surgery.
Unfractionated heparin (UH) is a large, complex polysaccharide that forms a complex with antithrombin III, itself a slow-acting α2-globulin protease inhibitor in plasma. The heparin-antithrombin III complex binds to the activated clotting factors, Factor IIa (thrombin), Factor Xa and to a lesser extent Factors IXa, XIa and XIIa, thereby blocking their actions. Heparin-mediated inhibition of activated Factor Xa is particularly important because of the key central position that Factor X holds in the coagulation cascade, which enables it to generate thrombin through both the intrinsic and extrinsic pathways.
Since several studies22,23 have demonstrated a good correlation among the intensity of anticoagulation therapy, heparin levels as defined by antithrombin (anti-IIa) activity and hemorrhagic complications, we hypothesize that epidural anesthesia after subcutaneous LDUH administration may be safe if these coagulation parameters are not significantly modified.23 To support this hypothesis, we assessed 50 patients who underwent elective bowel surgery for which 5000 U of LDUH was injected subcutaneously. Activated partial thromboplastin time (APTT), anti-IIa and anti-Xa were assayed for heparin activity before, and 2 and 4 hours after injection.
Patients and methods
Patients
Any of a consecutive group of patients who underwent elective bowel surgery for carcinoma of the colon, chronic diverticulosis or inflammatory bowel disease were eligible for inclusion in the study until 50 patients were enrolled. Patients were excluded for any of the following: refusal to give informed consent, presence of a known coagulopathy, required heparin therapy, previous use of oral anticoagulants or concomitant use of antiplatelet agents. These patients did not have an epidural catheter placed. Informed consent was obtained for all patients.
Samples
Citrated blood samples were obtained before the injection of 5000 units of standard UH (Leo Laboratories Canada, Ajax, Ont.), given 2 hours before surgery, and at 2 hours and 4 hours after the injection. Platelet-poor plasma was obtained after centrifugation at 2500 × g for 20 minutes. Aliquots were kept at −80 °C until assayed.
APTT, anti-IIa and anti-Xa
The APTT assays were performed using commercial reagents (DADE FSL cephalin or DADE FS, Baxter Diagnostic, Deerfield, Ill.) on an Automate ACL 300 (Instrumentation Laboratory, Coulter Electronics, Miami, Fla.). Anti-IIa and anti-Xa activity was determined by amidolytic assays using the same instruments. The anti-Xa assay utilized CBS.3139 specific synthetic chromogenic substrate and bovine factor Xa (Diagnostic Stago, Asnières, France).24 Chromogenic substrate and purified human thrombin (Instrumentation Laboratory) were used in the anti-IIa activity assay system.25 The rate of amidolysis of chromogenic substrate was recorded at 405 nmol/L, and results were expressed as anti-IIa or anti-Xa U/mL. Calibration curves were constructed for each assay using a pool of platelet-poor plasma collected from healthy volunteers. The lowest sensitivities of both assays were 0.10 U/mL. All samples were analysed in duplicate.
Statistical analysis
The results of heparin measurement assays (APTT, anti-IIa, anti-Xa) are expressed as means (and standard deviations); 99% confidence intervals were also calculated. Analysis of variance was used to test the difference between pre-heparin and post-heparin injection.
Results
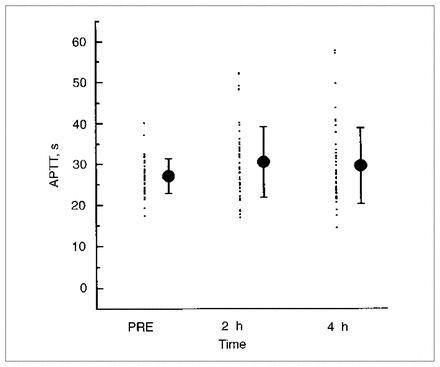
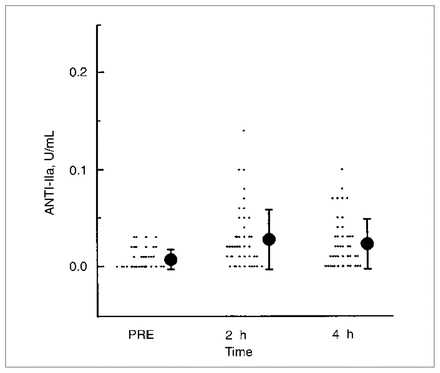
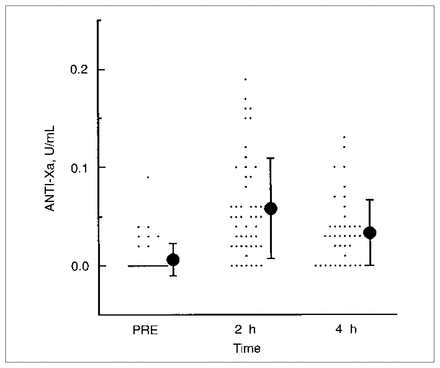
Among the 50 patients enrolled, the plasma of 2 patients demonstrated a prolonged APTT (more than 34 s) before they received the LDUH. Table I shows the initial APTT, anti-IIa and anti-Xa measurements, as well as those 2 hours and 4 hours after heparin injection. These results suggest that LDUH does not to prolong the mean APTT values significantly. However, 20% of the plasma samples demonstrated an APTT above the normal upper range (34 s) either at 2 hours or at 4 hours after injection (Fig. 1). Even though anti-IIa and anti- Xa levels were slightly elevated 2 and 4 hours after injection compared with the preinjection measurement, with a statistically significant difference (p < 0.05) (Table I), these levels were below the true sensitivity range of the assay (Fig. 2). The plasma sample of 1 patient showed an anti-IIa level higher than 0.10 U/mL at 2 hours after injection whereas in 8 patients the plasma demonstrated anti-Xa levels higher than 0.10 U/mL. Four patients had an anti-Xa level between 0.15 and 0.20 U/mL at 2 hours after injection. The plasma of 2 patients had an anti-Xa level higher than 0.10 U/mL, and none had an anti-IIa level higher than 0.10 U/mL at 4 hours (Figs. 2 and 3). None of the plasma samples assayed had a heparin level higher than the 0.20 U/mL (99% CI) cutoff considered as a significant detectable level. There was no correlation among APTT levels above the upper normal range and the anti-IIa or anti-Xa levels.
The patient distribution (mean and standard deviation) of APTT before injection of heparin (PRE), and at 2 and 4 hours after heparin injection.
The patient distribution (mean and standard deviation) of anti-IIa activity before heparin injection (PRE), and at 2 and 4 hours after heparin injection.
The patient distribution (mean and standard deviation) of anti-Xa activity before injection of heparin (PRE), and at 2 and 4 hours after heparin injection.
Mean (and Standard Deviation) Values and 99% Confidence Intervals (CI) for Activated Partial Thromboplastin Time (APTT), Antigen-IIa (Anti-IIa) and Antigen-Xa (Anti-Xa) Activities Before and After Heparin Injection
Discussion
Subcutaneous LDUH has been shown to provide efficacious thromboprophylaxis in general elective surgery but to be inadequate in high-risk groups such as patients undergoing orthopedic surgery.2–5 The hemorrhagic risk associated with heparin prophylaxis remains problematic,2,6,22,23 particularly for epidural anesthesia. Previous data have shown a good correlation between significant detectable plasma heparin anti-Xa levels higher than 0.20 U/mL and hemorrhagic complications.23 In our study, 16% of subjects had detectable heparin levels above the sensitivity range of the amydolytic functional anti-Xa assay 2 hours after the injection as did 4% of patients in the 4-hour sample, but none had heparin levels higher than 0.20 U/mL. The different sensitivities between IIa and Xa inhibition by heparin-antithrombin III complex in the diluted in vitro amydolytic assay may account for the different data shown.26,27 Similarly, with a more sensitive plasma thrombin neutralization assay, LDUH has shown low detectable levels compared with LMWH in normal volunteers.26
Samples with a prolonged APTT above the upper normal range did not correlate with detectable heparin levels above the sensitivity range of the amydolytic assay. Therefore, the APTT could not be used as a predictor of detectable heparin levels. In addition, we could not find any plasma factor deficiency to account for this prolonged APTT. All plasma exhibiting a prolonged APTT at 2 hours and 4 hours was normal when analysed with a cephalin APTT high phospholipid content (DADE FS) (data not shown), suggesting a plasma inhibitor effect similar to a lupus anticoagulant. Whether anesthetic agents or other intraoperative factors may exert a nonspecific inhibitor effect on phospholipid-dependent coagulation systems, thus contributing to the significant percentage of prolonged plasma APTTs, needs further evaluation. Our observations based on amydolytic end-point anti-IIa or anti-Xa assay, both highly specific and sensitive for heparin activity measurement, are not in accord with those of studies reporting therapeutic changes in coagulation after subcutaneous LDUH utilizing an anti-factor Xa clotting end-point assay.28 In a similar fashion to the APTT, a clotting end-point assay may well be affected by several factors such as a nonspecific inhibitor or variability of a reagent sensitivity to heparin.29 Thus, recommendation to delay epidural catheter insertion for at least 4 to 6 hours after LDUH injection appears to be inacurrate.20–22,30,31
The data show that none of the 50 patients had heparin levels above 0.20 U/ml (99% CI), so the expected risk of hemorrhage during epidural anesthesia after the administration of LDUH would be nearly negligible. However, it remains that subcutaneous heparin administration may be a contributory factor when associated with other coagulopathies. In the only 2 cases of spinal hematoma reported afterk the subcutaneous injection of LDUH,17,18 we could clearly identify other factors contributing to the hemorrhagic event (i.e., in the first case, several puncture attempts with a large 16-gauge needle in an area treated previously with radiotherapy and, in the second case, abnormal coagulation parameters preoperatively due to bileduct carcinoma). Still, given the low incidence of spinal hematoma complicating epidural anesthesia, a sufficiently large clinical study to evaluate the safety of spinal anesthesia after prophylactic subcutanous injection of 5000 units of LDUH in general abdominal surgery would appear infeasible. Our study is an indirect way of demonstrating the safety of this clinical practice that has become so popular.
Conclusions
Our findings indicate that subcutaneous injection of 5000 units of LDUH injection in general abdominal surgery is not associated with coagulation hemorrhage. Unable to postulate a safety recommendation, we are confident that epidural anesthesia could be performed after 2 hours of LDUH administration without a high risk of hemorrhage in the absence of other impairment of hemostasis.
Acknowledgments
We thank Drs. Andrew Mitchell (pathologist) and Pierre Drolet (anesthesiologist) for their valuable help in reviewing the manuscript.
This work was supported by a grant from the Centre de Recherches Guy-Bernier, Hôpital Maisonneuve-Rosemont, Montreal
- Accepted August 30, 1999.