Abstract
Objective: In lower-extremity surgery there are significant risks associated with the use of tourniquets. This prospective study was done to assess to what extent these risks may be offset by the potential advantages of tourniquets, namely reductions in blood loss, length of hospital stay and complication rates.
Design: A prospective case study.
Setting: A major urban hospital.
Patients: Sixty-three consecutive patients scheduled for primary cemented total knee arthroplasty (TKA) were blindly randomized into tourniqet (n = 33) and non-tourniquet (n = 30) groups.
Intervention: TKA during which a pneumatic tourniquet was applied or not applied to control blood loss.
Main outcome measures: Perioperative blood loss, operating time, complication rates, hospital stay and transfusion needs.
Results: Differences in the total measured blood loss, intraoperative blood loss and the Hemovac drainage blood loss between the 2 groups were not significantly different (p > 0.25). The calculated total blood loss was actually lower in the non-tourniquet group (p = 0.02). Between the groups there were no statistical differences in surgical time, length of hospital stay, transfusion requirements or rate of complications (although there was a trend to more complications in the tourniquet group (p = 0.06)).
Conclusion: The effectiveness of a pneumatic tourniquet to control blood loss in TKA is questionable.
The use of the pneumatic tourniquet has been advocated in total knee arthroplasty (TKA) to minimize blood loss and to improve visualization in the surgical field.1 But even with a tourniquet, patients experience substantial blood loss in the course of the procedure.
A disadvantage of tourniquets is the morbidity that arises from their use, especially neuromuscular injury secondary to the ischemia of neural and muscular tissues, and direct compressive injury to nerves.2,3 Also, hemodynamic changes that accompany tourniquet inflation and deflation may depress cardiorespiratory function in the perioperative period.4–6 Although serious complications are rare, fatal pulmonary embolism and arterial occlusion have been reported7–10 with peripheral vascular disease.11 Allied to this, the use of the tourniquet has been associated with the appearance of showers of embolic debris coincident with deflation.12–16 These were detected by transesophageal echocardiography in the right atrium and ventricle of 29 of 30 patients who underwent TKA12 and have been observed in other lower-extremity procedures, including arthroscopy.13 The larger emboli (5 to 10 mm in diameter) appear to be venous thrombi rather than marrow elements and may cause increased pulmonary vascular resistance, and lower partial pressure of oxygen.15 Berman and associates12 reported that the emboli are not seen in the absence of tourniquet use.
Increasing awareness of the risks attending the use of tourniquets requires a re-evaluation of their potential benefits, especially the reduction of perioperative blood loss. The purpose of this study was to test whether tourniquets are effective in this regard, also noting the effects on operating time, complication rates, hospital stay and transfusion needs.
Patients
Sixty-three consecutive patients (37 women, 26 men) scheduled for primary TKA were entered into the study. Fifty-nine had a primary diagnosis of osteoarthritis, and the remainder had rheumatoid arthritis. The average age of the patients was 70.5 years (range from 47 to 83 years). Exclusion criteria were as follows: bilateral TKA required either simultaneously or staged at less than 3-month intervals; a history of bleeding diathesis; revision TKA; a history of musculoskeletal infection of the affected limb; or a history of peripheral vascular disease.
Patients were prospectively and blindly randomized to 1 of 2 groups (tourniquet or non-tourniquet) by the method of sealed envelopes. The senior surgeon and the anesthetist were unaware of the allocation until the envelopes were opened, after induction of anesthesia The composition and properties of the 2 groups are described in Table 1.
Data for Patients Who Underwent Total Knee Arthroplasty With and Without the Use of a Pneumatic Tourniquet
The preoperative data included the patients’ height, weight, age and medical history, including the preoperative use of nonsteroidal anti-inflammatory drugs. The patients’ hemoglobin levels were recorded within 24 hours of operation. Platelet count, prothrombin time and partial thromboplastin time were also measured to help ensure that the exclusion criteria were not violated.
Methods
Operative procedure
The type of anesthesia used depended on the patient’s needs. The ratio of general to spinal anesthesia (about 7 to 1) was similar in the 2 groups (Table 1). The groups were also similar with respect to operative procedure. Lateral releases were performed in 22 patients in the tourniquet group and 20 in the non-tourniquet group (Table 1). One synovectomy was performed in each group.
All patients received a primary cemented total knee replacement with a cemented polyethylene patellar replacement. A press-fit condylar design (Johnson & Johnson, New Brunswick, NJ) was used in 41 patients, 17 received the Omnifit 7000 prosthesis (Osteonics, Allendale, NJ) and 5 received AMK components (Depuy Orthopedics, Warsaw, Ind.). All operations were performed by a single surgeon (J.F.R.).
In the tourniquet group the leg was elevated and exsanguinated (without the use of an esmarch bandage) before tourniquet inflation. The tourniquet was set at 125 to 150 mm Hg above the systolic blood pressure, up to a maximum value of 300 mm Hg. The tourniquet was deflated after the bone cement had set, and only then was electrocautery used for hemostasis. In the non-tourniquet group electrocautery was used as necessary throughout the procedure.
In all patients bleeding from the femur was controlled by plugging the medullary canal with Gelfoam before wound closure. Uniformly, a single-lead suction drain was inserted and the wound closed in layers. A soft Jones dressing was then applied for 48 hours.
Intraoperative blood loss was recorded by the anesthetist using method estimation (i.e., suction drainage less irrigation volume [soaked sponges were considered equivalent to 80 mL of blood]). Also recorded were the operative and tourniquet times. Cell savers and autologous blood were not used.
Postoperative care and bloodwork
Postoperative care was standardized. Hemoglobin values were recorded on postoperative days 1, 2, 3, 5 and 7 (2.5-mL blood samples). Coumadin was used as prophylaxis for deep venous thrombosis, beginning in the evening on the day of surgery, and was followed by daily measurement of prothrombin time (4.5-mL blood sample) to reach an international normalized ratio of 2.0 to 2.5.
Suction drainage volume was recorded for 36 hours after the operation. Patients were up with assistance 24 hours after the operation. Continuous passive motion was not used.
Transfusion requirements (with donor homologous blood) were determined by clinical criteria rather than by hemoglobin levels, typically tachycardia (more than 100 beats/min), hypotension (systolic blood pressure less than 100 mm Hg), syncope and fatigue, and the patient’s general medical condition. Dates and times of transfusions were documented.
All complications were noted, including deep venous thrombosis, infections and delayed wound healing. Patients were not uniformly screened for deep venous thrombosis, rather the approach was to look for symptoms and then confirm a the condition by venography or ultrasonography.
Blood loss measurements and calculations
Total measured blood loss (TMBL) was the sum of the intraoperative blood loss (IBL) estimated by the anesthesiologist and the Hemovac drainage blood loss (HBL). The calculated total blood loss (CBL) was obtained by the method of Gross.12,17 With this method, the estimated blood volume (EBV) was calculated from the height and weight of the patient, using a correction factor for gender:
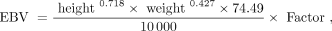
where Factor for males = 2.74 and for females = 2.37.
CBL was then calculated from the maximum reduction in hemoglobin (Hgb), standardized with respect to EBV (assuming isometric hemodilution):
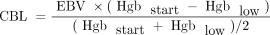
Blood was analyzed on a Coulter STK6 machine (Beckman Coulter, Fullerton, CA) having a mechanical error of about ±0.8%. Accumulated blood loss due to serial sampling was recorded and taken into account when computing measured blood loss.
Statistical procedures
Tests were analyzed by Student’s t-test or the χ2 test (where indicated). The level of significance was established at 95% confidence limits (p < 0.05). The data were processed using SYSTAT software (Systat, Evanston, IL).
Results
Blood loss measurements and calculations
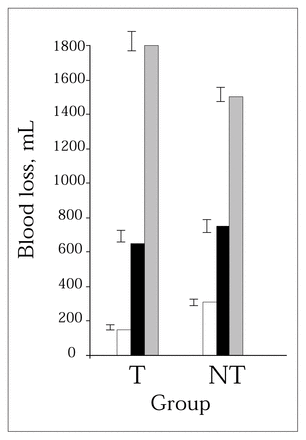
Blood loss data for the tourniquet and control groups are detailed in Table 2. Whereas the IBL was significantly greater in the non-tourniquet group (p < 0.0001), HBL did not differ significantly (p > 0.4). Therefore, in terms of the total measured loss of blood (TMBL = IBL + HBL) the non-tourniquet group had the greater loss, but high standard deviations rendered the intergroup difference insignificant (p > 0.25). These trends are summarized in Fig. 1. Combining both groups yielded a mean (and standard deviation) TMBL of 696 (308) mL.
Mean (and standard deviation) blood loss (mL) as affected by the use of a pneumatic tourniquet in total knee arthroplasty. Grey bars = calculated blood loss, black bars = total measured blood loss, white bars = intraoperative blood loss. T = tourniquet group (n = 33), NT = non-tourniquet group (n = 30).
Blood Loss and Other Parameters for Patients Who Underwent Total Knee Arthroplasty With (33 Patients) and Without (30 Patients) the Use of a Pneumatic Tourniquet
When blood loss was calculated by the method of Gross to yield the total CBL, the tourniquet group, surprisingly, had significantly greater loss than the non-tourniquet group (p = 0.02).
To obtain an overall comparison of TMBL and CBL, the patients were combined into a single group and the means calculated. The TMBL (696 [308] mL) amounted to only 42% of the CBL (1652 [505]) (p < 0.0001).
The mean preoperative and postoperative hemoglobin levels did not differ significantly between tourniquet and non-tourniquet groups. Although the perioperative drop in hemoglobin (not standardized to the patients’ blood volume) was greater in the tourniquet group, the difference was not significant (p > 0.1) (Table 2).
Blood loss due to phlebotomy for the hemoglobin assays was not significantly different between the groups (p > 0.4) and amounted to a mean total of less than 40 mL in each patient.
Effects of gender on blood loss
Although the female to male ratio was greater in the non-tourniquet group (Table 1), it was not significantly so (p > 0.05, χ2 test). As a trend, males lost more blood than females. This was of borderline significance in the case of CBL (male 1785 [642] mL, female 1547 [336] mL) (p = 0.06). For TMBL the difference was not significant (male 726 [342] mL, female 672 [280] mL) (p > 0.4).
The mean (and standard deviation) hemoglobin levels were higher in men than women (preoperative: men 140 [15] g/L, women 128 [14] g/L; postoperative: men 102 [16] g/L, women 88 [12] g/L). These gender differences were significant (p < 0.001).
The transfusion requirement was numerically greater in the women (11 of 37) than in the men (5 of 26), although the difference in the distribution was not significant (p > 0.15, χ2 analysis).
Transfusion rates and other factors in convalescence
Transfusions were required in about one-quarter of patients. The odds ratio for transfusion requirement was 1.7:1 in favour of the tourniquet group but was not significant (p > 0.1). Within each group the mean amounts of blood administered, per case, were similar: 1.7 units (tourniquet), 1.5 units (no tourniquet). When the 16 patients needing transfusions were considered as a separate group, and compared with all the others, there was no difference in IBL, TMBL or CBL (p > 0.2 in each case). However, the requirement for patient transfusion (determined on clinical grounds) did correlate with low preoperative mean hemoglobin values: 119 (10.9) g/L in those transfused versus 138 (13.8) g/L in all others (p < 0.0001). There was a similar correlation with the lowest postoperative hemoglobin values: 79.5 (5.4) g/L (transfused) versus 99.0 (14) g/L in the others (p < 0.0001). However, the actual perioperative drop in hemoglobin was almost the same in the 2 groups (39.5 versus 39.0 g/L) (p > 0.5).
Operative time was not significantly affected by the use of a tourniquet: 83 (13) minutes (tourniquet) versus 81 (14) minutes (non-tourniquet) (p > 0.5).
The mean hospital stay of the tourniquet group was longer (p = 0.075, Table 2), and the number of documented complications was higher in this group (odds ratio 3.3:1, p = 0.06, χ2 test). In the tourniquet group there were 4 superficial wound infections (requiring antibiotic treatment), 1 case of skin blistering and 6 cases of significant wound hematoma. In the non-tourniquet group there was 1 superficial wound infection, 3 hematomas and 1 gastrointestinal hemorrhage (no transfusion needed).
Discussion
The 2 groups used in the study were similar in all respects except for the use of a tourniquet (Table 1). This provided a useful means to assess the efficacy of the tourniquet with respect to blood loss during TKA and postoperatively.
Regarding blood-loss measurement, it is now known that the TMBL represents only a fraction of the actual blood loss incurred by the patient. Our finding of 42% falls within the range reported previously.18–20 One source of the error in TMBL is that generally less than half of the lost blood exits through the suction drain.19–21 Most of the loss, which can be monitored experimentally by the tracking of technetium-labelled red cells, appears due to perioperative hemorrhage into the tissues at the site of surgery.22 CBL also has the limitation (based on diminishing hemoglobin levels) that it is valid only to the point at which a transfusion is required,17,19,23 after which the true loss is underestimated. Nevertheless, various studies have indicated that CBL likely provides a much more accurate indication of actual loss, than TMBL.18,19 For this reason we attach particular importance to the CBL data arising from this and similar studies. Although CBL is not widely used in the clinic, its value is now well documented in the context of clinical research studies.
IBL was clearly reduced by the use of the tourniquet (p < 0.001), but the opposite trend for HBL meant that the TMBL was not diminished (p > 0.25). This is consistent with the report of Abdel-Salam and Eyres.4 The association we have found between tourniquet usage and greater CBL (p < 0.02) was supported by a trend for greater change in the levels of hemoglobin, a finding that agrees with the findings of Jarolem and associates.24 One reason for this may be that the ischemic conditions brought about by the use of a tourniquet could induce sustained local reactive hyperemia lasting several hours after the tourniquet deflation. This would in turn promote more hemorrhage into the traumatized tissue in the perioperative period.6,25–27 Another possiblility is that the increased fibrinolytic activity associated with tourniquet-induced ischemia promotes bleeding into the local tissues following the procedure.26,28,29 The step-by-step hemostasis of blood vessels that is necessary when operating without a tourniquet may be more effective than the measures for hemostasis that are applied after tourniquet deflation. The conclusion remains that the use of a tourniquet in TKA was associated with increased total blood loss.
In view of the risks associated with blood products, the rate of transfusion requirement is an important parameter in relation to surgical technique. Although there was a higher odds ratio for transfusion in the tourniquet group, the trend was not statistically validated, and the total number (both groups) requiring transfusion was low (16 cases). However, given a larger sample size one might expect a significant difference in keeping with the statistically greater calculated blood loss seen in our tourniquet group. Nevertheless, in both our groups the prime determinant of the need for transfusion was a low initial hemoglobin level, resulting in a clinically depressed postoperative level. These data are in agreement with those of other reports.12,30
Our finding that the odds ratio for complications was greater in the tourniquet group (3.3:1) albeit at borderline significance (p = 0.06), is consistent with the finding of Abdel-Salam and Eyres,4 who reported a higher incidence of TKA wound complications when a tourniquet was used, with increased postoperative pain and slower recovery postoperatively. Similarly Maffulli and colleagues27 noted with ankle fractures that more frequent wound complications, more tissue swelling and a slower recovery all accompanied the use of tourniquets. These testify to the mounting evidence that tourniquet use may compromise wound healing and incite local tissue trauma, affecting recovery from operation.
The operative time is an interesting parameter since it may be the only objective measure of the degree of difficulty or visualization associated with a procedure. Since there was no significant change in operating time with the use of the tourniquet, it appears it had no affect on either difficulty or visualization in TKA. Furthermore, in the opinion of the senior surgeon (J.F.R.) it was no more difficult without a tourniquet to obtain dry-cut bone surfaces for cementing. Similar opinions have been expressed elsewhere.4,24
Conclusions
The use of a tourniquet can no longer be considered a benign procedure in the light of the well-defined and worrisome morbidities that have been reported. We found no data to support its continued use on the grounds of blood loss reduction. Although there was some advantage in terms of reduced intraoperative blood loss, this was offset by significantly greater overall blood loss. This extended to a greater transfusion demand in the tourniquet group which, although not statistically validated, is worthy of pursuit in further studies. Also to be evaluated in future work is whether the tourniquet might be recommended on other grounds, such as longer implant survival or benefits to cementing techniques.
Acknowledgement
We thank Dr. A. Scudamore for advice and assistance in the assembly of the manuscript.
- Accepted October 26, 1999.