Abstract
Objective: To determine the efficacy of using the harmonic scalpel and robotic assistance to facilitate thoracoscopic harvest of the internal thoracic artery (ITA).
Design: A case series.
Setting: London Health Sciences Centre, University of Western Ontario, London, Ont.
Patients and methods: Fifteen consecutive patients requiring harvest of the ITA for coronary artery bypass grafting.
Intervention: Robot-assisted, video-enhanced coronary artery bypass (RAVECAB) through limited-access incisions, using the harmonic scalpel and a voiceactivated robotic assistant.
Main outcome measures: Ease and duration of the harvesting technique, complications of the procedure, graft flow and patency, and duration of postoperative hospitalization.
Results: RAVECAB facilitated thoracoscopic dissection of the ITA with the harmonic scalpel in all cases. There were no conversions to a standard approach and no reoperations for bleeding. The mean (and standard deviation) ITA harvest time was 64.1 (22.9) minutes (range from 40 to 118 minutes). Robotic voice command capture rate was greater than 95%. Mean (and SD) intraoperative graft flows were 33.1 (26.8) mL/min (range from 14 to 126 mL/min). There was 100% graft patency on postoperative angiography. There were no deaths, perioperative myocardial infarction or arrhythmias. Mean (and SD) postoperative hospitalization was 3.3 (0.8) days.
Conclusions: RAVECAB is a demanding procedure that addresses many of the disadvantages of the “conventional” minimally invasive coronary artery bypass. It allows complete pedicle dissection with minimal ITA manipulation and assures sufficient conduit length and a tension-free coronary artery anastomosis. All anastomoses were performed under direct vision through a 5- to 8-cm inferior mammary incision.
Minimally invasive coronary artery bypass (MIDCAB) grafting of anterior coronary arteries with in-situ internal thoracic artery through a limited anterior thoracotomy is a procedure that is rapidly gaining acceptance. The procedure is most commonly performed through a 5- to 10-cm left anterior thoracotomy with the left internal thoracic artery (ITA) harvested under direct vision. Through this approach, direct ITA takedown can be difficult and incomplete and often necessitates the removal or division of costal cartilage.1,2 To facilitate direct ITA harvest, a minithoracotomy is often performed more medially, compromising optimal surgical orientation for ITA–left anterior descending (LAD) coronary artery anastomosis, which is more lateral and centred over the LAD coronary artery. The directly harvested ITA may also be too short since dissection of the conduit inferior to the thoracotomy site is difficult. ITAs that are not long enough are at risk of either kinking or avulsion.3 Video thoracoscopic harvesting of the full length of the ITA avoids these technical problems and further decreases the trauma of surgery.4 Proficiency at thoracoscopic ITA harvesting, however, involves a significant learning curve and for surgeons inexperienced in thoracoscopic techniques may present a formidable challenge.4 Valid concerns have been raised about the safety of the technique.5,6 The use of surgical robots capable of positioning an endoscope in response to a surgeon’s verbal command is a concept that recently has been successfully applied to thoracic surgery after demonstrating effectiveness in endoscopic abdominal procedures.7 The automated endoscope system for optimal positioning (AESOP 3000; Computer Motion, Santa Barbara, Calif.) facilitates video thoracoscopic ITA dissection by providing the surgeon with steady images and voice-activated camera control.
The harmonic scalpel (Ethicon Endo-Surgery, Cincinnati, Ohio) is an ultrasonic surgical instrument for cutting and coagulating tissues, operating at a frequency of 55.5 kHz. Bleeding vessels are coapted by tamponading and then sealed with a denatured protein coagulum, eliminating the need for hemostatic clips. Initial thermal studies have indicated that heat generated from the high-frequency vibration of the tissue is limited.8 Use of the harmonic scalpel for ITA dissection enables a less traumatic, spasm-free conduit dissection while minimizing the need for instrument transfers when compared with conventional electrocautery.9 This prospective study reviews our clinical experience with off pump RAVECAB (robot-assisted, video-enhanced off-pump minimal access coronary artery bypass) using the harmonic scalpel to facilitate thoracoscopic harvest of the ITA.
Patients and methods
Between Sept. 1 and Nov. 19, 1998, 15 consecutive patients underwent RAVECAB through limited-access incisions at the London Health Sciences Centre, London, Ont. The mean (and standard deviation) age of the patients was 55.8 (13.5) years. The preoperative patient demographics are shown in Table 1. All ITAs were harvested totally endoscopically using the harmonic scalpel and the AESOP 3000, a voice-activated robotic assistant. No patients were rejected on the basis of small size of the target vessel, and one-third of patients underwent successful grafting of vessels less than 1.5 mm in diameter (Table 2). No patients were refused surgery because of obesity or body habitus. All patients underwent pulmonary function testing before operation to confirm sufficient pulmonary reserve to tolerate isolated right lung ventilation during left ITA dissection.
Preoperative Demographic Data for 15 Patients Who Underwent Internal Thoracic Artery Harvesting
Graft Analysis for 15 Patients Who Underwent Robotic-Assisted, Video-Enhanced Coronary Artery Bypass
Anesthetic considerations and surgical technique
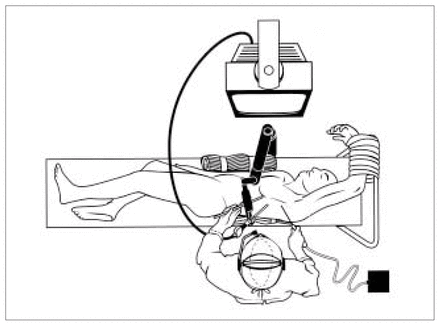
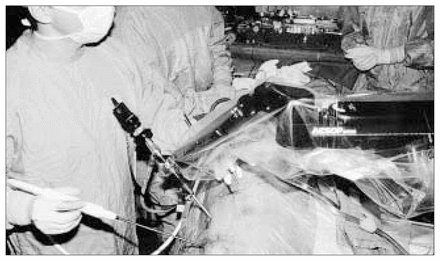
After induction of general anesthesia with a double-lumen endotracheal tube, and allowing complete collapse of the left lung, each patient was placed in the 30° right lateral decubitus position. The left arm was abducted for sufficient exposure of the axilla to allow placement of the access ports and was draped as for a conventional bypass procedure. External defibrillation pads were placed, and warming blankets were used to prevent hypothermia. The video monitor was placed directly across the operating table from the surgeon who was seated on the patient’s left side. The AESOP 3000 was positioned on the operating table directly opposite to the proposed camera port-access site (Fig. 1). Thoracoscopic ITA harvest was performed using 3 incisions, each 5 mm long. A 5-mm 30° video thoracoscope was first placed through an insufflation port in the fifth intercostal space in the anterior axillary line and adapted to the robotic arm. The harmonic scalpel was then inserted through a 5-mm stab incision in the third intercostal space in the anterior axillary line (Fig. 2). A third 5-mm incision was made in the sixth intercostal space in the lateral clavicular line for a Kittner dissector (Ethicon Endo-Surgery, Cincinnati). During the procedure, warm carbon dioxide was insufflated into the pleural cavity to a pressure of 5 to 10 mm Hg. A Veress needle was inserted near the camera port and vented under suction when higher carbon dioxide flows were used so as to clear intrathoracic vapour created by the harmonic scalpel. A nitroglycerin infusion was routinely started before ITA harvest and titrated to maintain a systolic blood pressure greater than 100 mm Hg. All patients were monitored with catheters placed in the radial and pulmonary arteries. The cardiopulmonary bypass pump was on standby in the operating room. The left ITA was then identified, and with voiceactivated robotic and video assistance the harmonic scalpel was used to dissect the ITA as a pedicle from the first to the sixth rib. The parietal pleura was first scored 1 to 2 cm medial and lateral to the ITA from the first rib to the sixth intercostal space. Often arterial pulsation observed through the soft tissue guided distal dissection, but occasionally no obvious pulsation was seen. In these cases, gentle distal compression of the soft tissues with the Kittner dissector augmented arterial pulsations, identifying the ITA. Dissection was usually started over the second rib, and progression of the dissection was always initiated over subsequent ribs which acted as “safe zones” without intercostal vascular tethering. Once the ITA was safely separated from the superior and inferior ribs, the intervening intercostal spaces were carefully dissected. The Kittner dissector was used to provide gentle countertraction during the dissection and was often positioned by rolling the Kittner dissector on the ITA. When dividing the intercostal arteries and veins with the harmonic scalpel, gentle pressure was applied with the blunt side of the blade at a power setting of 3. During cutting and coagulation, harmonic scalpel loiter times averaged 5 seconds but varied with the size of the intercostal branches being divided. When the dissection reached the fifth or sixth intercostal space, it was sometimes necessary to change the harmonic scalpel to a lower interspace through the same skin incision because of excessive torque and limited manoeuvrability caused by the soft tissues and ribs. Occasionally it was necessary to interchange the harmonic scalpel and the camera sites to complete the dissection.
Surgical positioning for robot-assisted, video-enhanced coronary artery bypass. Ports are made in the third and fifth intercostal spaces along the anterior axillary line and at the sixth intercostal space on the lateral clavicular line.
Intraoperative surgical instrument orientation and robotic arm position.
Once ITA dissection was complete, the pericardium was opened vertically and the target vessel identified thoracoscopically. The LAD coronary artery was approached through a small (5 to 8 cm) left anterior thoracotomy along the fourth intercostal space. There was no need to excise or divide ribs or costal cartilages in any patient. After 7500 to 10 000 units of heparin were administered, and the activated clotting time was verified to be longer than 300 seconds, the ITA was divided distally and the pedicle injected with papaverine. Silk sutures were immediately placed on either side of the pedicle and held with hemostats to maintain pedicle orientation and prevent twisting.
The pericardium was then tethered to the skin edges to elevate the heart. A 100-mg intravenous bolus of lidocaine was given before the LAD artery proximal to the proposed arteriotomy was encircled with either 4-0 Prolene (Ethicon, Somerville, NJ) or a silicone elastomer vascular loop (Quest Medical, Allen, Tex.). Ischemic preconditioning was carried out for 5 minutes if the target vessel was not occluded. Stabilization was accomplished with the CardioThoracic Systems retractor (CardioThoracic Systems, Cupertino, Calif.). Anastomoses were completed with a continuous 8-0 Prolene suture with either a single- or double-suture technique. Before tying the distal sutures, the size and patency of the LAD coronary artery were verified by coronary probes.
After completion of the anastomosis, graft flows were checked with a transit-time ultrasonic flow probe (Transonic Systems, Ithaca, NY). A small chest tube was placed in the left pleural space. If a thoracic epidural catheter was not inserted preoperatively, an intercostal catheter was positioned in the fourth intercostal space before the wound was closed. Transthoracic Doppler studies were carried out to verify graft patency on the first postoperative day. Angiography was performed postoperatively on 14 patients before discharge. All patients were seen in the outpatient clinic 6 weeks postoperatively.
Results
RAVECAB with use of the harmonic scalpel facilitated total thoracoscopic dissection of the ITA in all cases. There were no cases of injury to the graft necessitating conversion to a standard approach and no bleeding necessitating reoperation. Vascular branches were transected with excellent hemostasis, and no hemoclips were required for side-branch bleeding. There were no deaths, perioperative myocardial infarctions or arrhythmias, and no blood transfusions were required perioperatively. One patient had a superficial wound incision, which was treated successfully with orally administered antibiotics. One woman who had a 1.0-mm diameter coronary artery and an anastomotic irregularity at postoperative angiography suffered recurrent angina. She underwent coronary angioplasty 3 weeks postoperatively and is presently symptom free. The mean (and SD) intraoperative graft flow was 33.1 (26.8) mL/min. The mean (and SD) ITA harvest time was 64.1 (22.9) minutes (range from 40 to 118 minutes) and LAD anastomosis time was 14.2 (3.9) minutes. No patient was declined RAVECAB because of body habitus. Patients 1 and 3 from the series (Table 2) weighed 150 kg and 136 kg respectively. Robotic assistance significantly reduced the number of endoscopic cleanings required for ITA dissection from 1 cleaning every 8 minutes without robotic assistance to 1 cleaning every 21 minutes with robotic assistance. It also facilitated the anticipated difficult dissection required in these obese patients. Robotic voice command capture rate was greater than 95%. There was no evidence of conduit spasm or trauma after ITA harvesting. Postoperatively, the majority of patients (73%) were extubated in the operating room. There was 100% graft patency in the 14 patients who underwent postoperative angiography.
All anastomoses were reviewed and graded according to the criteria of FitzGibbon and associates:10 grade A, excellent graft with unimpaired run off; grade B, stenosis reducing the calibre of proximal or distal anastomosis or trunk to less than 50% of the grafted coronary artery (overall, B grading was determined by the lowest of the 3 specific site grades); grade O, occlusion. On qualitative analysis of the anastomoses, 13 of 14 (93%) were grade A, and 1 (7%) was grade B. Postoperative angiography for 1 patient was cancelled because of an emergency in the catheter laboratory. The patient refused to wait in hospital for the study and was discharged. The mean (and SD) postoperative stay in the intensive care unit was 16.3 (10.6) hours. Four of the 15 patients were temporarily monitored in the recovery room and transferred to the ward several hours later. Mean (and SD) postoperative hospital stay was 3.3 (0.8) days (median 3.0 days) (Table 3).
Perioperative Data for 15 Patients Who Had Robotic-Assisted, Video-Enhanced Coronary Artery Bypass*
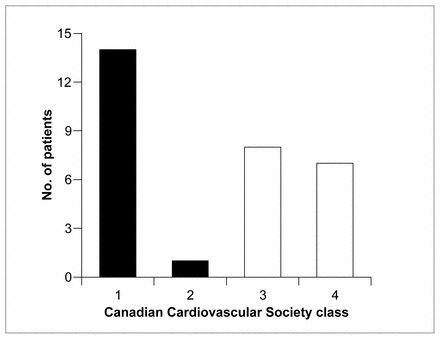
All patients were seen 6 weeks postoperatively, and their postoperative angina class was compared to their preoperative angina class according to the Canadian Cardiovascular Society classification (Fig. 3).
Comparison of Canadian Cardiovascular Society angina class preoperatively (white bars) and postoperatively (black bars).
Discussion
Minimally invasive direct coronary artery bypass grafting of vessels on the beating heart is a procedure in evolution. Over the last few years, the procedure has undergone several modifications as new technologic developments become available. Two such advances include the introduction of the voice-activated robotic assistant and the harmonic scalpel.
The results of this study confirm that RAVECAB has significant advantages over previously described ITA harvesting techniques for MIDCAB.1,2 These advantages include complete dissection of the ITA from the first rib to the sixth intercostal space with transection of all collateral branches arising from the ITA without instrument transfer; greatly improved exposure of the ITA beyond the level of the second rib, and elimination of the need to retract the thoracotomy for ITA exposure.
The use of robotics to enhance minimally invasive operations has been described in other disciplines.11 The surgical robot used in this series to aid in harvesting the ITA, the AESOP is capable of positioning a thoracoscope in response to a surgeon’s verbal commands. AESOP approximates the form and function of a human arm and provides the surgeon with direct control of the thoracoscope. In our initial experience, the use of the AESOP 3000 robotic assistant allowed the surgeon to safely perform solo thoracoscopic ITA harvesting comfortably without prolonging operative time and without additional equipment. The benefits of robotic assistance include greater image stability, less inadvertent smearing of the thoracoscope12 and a consistent quality of assistance. By providing instantaneous “surgeonbrain-camera” positioning, accurate instrument tracking and optimal magnification, the robot markedly increased surgeon video-dexterity and lessened surgeon fatigue.
Successful application of robotic assistance to thoracoscopic ITA harvesting involves attention to several technical details. To avoid mechanical “elbow limits” that prevent satisfactory inferior robotic camera placement, the robot should be positioned on the bedrail opposite the inferior limit of proposed surgical dissection. This position is usually one interspace below the camera port site at the sixth intercostal space. Working port positions must also be slightly modified to accommodate the extrathoracic space requirements of the robotic arm. We have found that port positions that are oriented more linearly along the anterior axillary line are less likely to cause instrument interference and limit working angles than ports oriented at more acute working angles as advocated by experienced thoracoscopic surgeons when harvesting the ITA without robotic assistance.9,13
Because of the concern regarding potential thermal injury with the use of diathermy as the method of hemostatic control of collateral branches during dissection of the ITA, we adopted the harmonic scalpel for thoracoscopic conduit harvesting. This scalpel has proven to be a safe and efficient means of dissection as it operates at a lower temperature than diathermy and does not require instrument transfers to control side branches.9
Thoracoscopic ITA harvesting allows a complete dissection of either the left or right ITA from the first rib to the sixth intercostal space under clear magnified views that are even superior to those of the open approach. A sufficient length of ITA is readily obtained without costal resection or vigorous retraction of the thoracotomy. Dissection of the ITA inferior to the proposed thoracotomy site is also considerably easier with thoracoscopic techniques than with presently available thoracotomy retractors designed to directly expose the ITA. Coronary anastomosis may also be accomplished through a smaller incision centred directly over the target vessel by video guidance.
In our early experience with RAVECAB we have demonstrated anastomotic and early graft quality that is comparable to anastomosis constructed and ITAs harvested through a sternotomy under conventional cardioplegic arrest. A longer angiographic follow-up period is still needed, however, to confirm the durability of our findings.
Conclusions
Modification of the current MIDCAB technique by performing thoracoscopic and robot-assisted ITA harvesting has significantly decreased the morbidity associated with this procedure. The essential conditions required are good visualization of the thoracic cavity and the course of the ITA and definitive control and division of collateral branches. The use of robotic assistance and the harmonic scalpel greatly facilitate the conduct of the procedure, decreasing morbidity and potentially improving surgical outcome.
Footnotes
Presented at the Evolving Techniques and Technologies in Minimally Invasive Cardiac Surgery meeting, San Antonio, Tex., Jan. 21 to 24, 1999.
- Accepted March 21, 2000.