Abstract
Objective: To describe the etiology and clinical course of acute colitis occurring after flexible endoscopy.
Design: Chart review.
Setting: A university teaching hospital.
Patients: Eight patients who sought assessment of potential colonic disease.
Intervention: Colonoscopy in 5 patients and flexible sigmoidoscopy in 3 patients. The indication for endoscopy was screening in 5 patients, cancer surveillance in 2 patients and preoperative evaluation of colon carcinoma in 1 patient.
Outcome measures: The relation of presenting symptoms to glutaraldehyde exposure, the response to therapy and the need for further therapy.
Results: All patients had abdominal pain, mucus diarrhea and rectal bleeding within 48 hours after endoscopy. Most patients reported that the symptoms started within 12 hours of the procedure. All patients were confirmed by sigmoidoscopy to have colitis within 72 hours of the first endoscopic procedure. One patient required hospitalization. In the first 7 patients several stool cultures were negative for Clostridium difficile using the cytotoxin assay by the cell culture method. Four patients had negative cultures for Yersinia, Salmonella and Shigella spp. Three patients were treated with metronidazole initially. Two patients underwent endoscopic biopsy and examination of the biopsy specimen showed fibrinoleukocytic exudate and ischemic type injury. One patient underwent the scheduled sigmoid resection within 48 hours of endoscopy for a Dukes’ stage B adenocarcinoma. Concomitant acute ischemic colitis limited to the mucosa and submucosa was noted in the resected specimen. Symptoms resolved in all patients and follow-up endoscopy revealed normal mucosa.
Conclusion: The entity of glutaraldehyde-induced colitis should be recognized and special attention should be given during instrument cleansing to minimize the risk of its development.
Concern about the transmission of pathogens during flexible endoscopy has led to the increasing use of glutaradehyde as a disinfecting solution. As a 2% solution glutaraldehyde is widely used because of its broad spectrum of action against acid- and alcohol-resistant bacilli, hydrophilic viruses and spores. This agent and its vapours are known to be irritants, both allergenic and toxic.1–3 Direct contact with the colonic mucosa can produce an acute self-limited colitis.4
An outbreak of unexplained acute-onset colitis at the Sir Mortimer B. Davis-Jewish General Hospital in Montreal prompted us to conduct this study. Our purpose was to highlight the complication by describing the clinical course of a series of patients who had acute colitis after undergoing flexible endoscopy in a busy endoscopy centre where 2% glutaraldehyde had been used as the disinfectant.
Patients and methods
We reviewed our experience with 8 patients (5 women, 3 men, ranging in age from 38 to 72 years) who were seen in our hospital between July 11, 1994, and Jan. 9, 1995. The indications for the flexible endoscopy were colorectal carcinoma screening (5 patients), postoperative surveillance (2 patients) and total colonic evaluation prior to an elective colon cancer resection to rule out a synchronous lesion (1 patient). The initial endoscopic procedure was colonoscopy in 5 patients and flexible sigmoidoscopy in 3 patients. All patients underwent follow-up endoscopy.
All patients denied recent travel, antibiotic use, intake of suspect food or contact with anyone who had diarrhea. No patient had a history of inflammatory bowel disease.
The bowel was prepared for flexible sigmoidoscopy by giving 2 phosphosoda enemas on the day of the examination. Before colonoscopy patients were given phosphosoda orally (90 mL in two 45-mL bottles). All the endoscopic procedures were considered uncomplicated by the treating physician and all index patients had macroscopically normal mucosa before the onset of symptoms.
The protocol for instrument care was reviewed. All endoscopes were washed by hand with the aid of an electrical pump to irrigate the channels. The initial step in cleaning the endoscopes included cleaning the outer surface, and irrigating the suction channel and biopsy channel with a proteolytic enzymatic detergent (Metrizyme; Metrex Research, St. Laurent, Que.). The suction and biopsy channels were then brushed. Both the colonoscopes and the flexible sigmoidoscopes underwent a 20-minute immersion and second channel irrigation with 2% glutaraldehyde (Metricide; Metrex Research, Morrisburg, Ont.). To completely rinse off the glutaraldehyde from the colonoscopes, the instruments were immersed and irrigated for 10 minutes in a tap-water bath. The flexible sigmoidoscopes were submerged in a tap-water bath and the channels were irrigated with at least 180 mL of tap water. It was the practice to change the rinsing water at fixed intervals. The rinse water would also be changed if it became greenish, which suggested contamination with glutaraldehyde, although this was seldom the case. The water bottles were disinfected in 2% glutaraldehyde daily. Hydrogen peroxide was not used at any stage during the processing of the endoscopes. There were no changes in endoscopy nurses or technical staff before or during any of the problems described in our patients.
Results
Symptoms
Because of their presenting symptoms, all the patients were unable to perform their daily activities and appeared mild to moderately ill. All 8 patients suffered from abdominal pain, mucus diarrhea and rectal bleeding within 48 hours of the initial endoscopy. Five patients experienced the symptoms within 12 hours.
Investigations
During the acute episode, all 7 patients who underwent stool testing had samples that were negative for Clostridium difficile as determined by the cytotoxin assay with the cell culture method. Four patients had negative stool cultures for Yersinia, Salmonella and Shigella spp. Cultures of the colonoscopes in the first 3 patients failed to reveal any pathogens.
All patients were confirmed, by sigmoidoscopy, to have colitis within 72 hours of their first endoscopy. The endoscopic features during the illness were moderately severe colitis with patchy necrosis of the mucosa, ulcerations, friability and fibrinous exudate. A biopsy was done in 2 patients during the acute episode.
Treatment
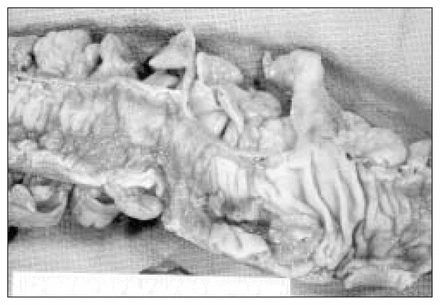
One moderately ill patient with abdominal pain and tenderness, an oral temperature of 39 °C and leukocytosis required hospitalization to receive intravenous fluids and for observation. Three patients were initially treated with metronidazole: 1 of these stopped taking her medication after 4 days because she was feeling better; another had her medication stopped by her physician 48 hours later; the last patient finished the 10-day course. One patient underwent the previously planned sigmoid resection for a Dukes’ stage B adenocarcinoma of the sigmoid colon (Fig. 1).
Acute glutaraldehyde injury adjacent to the resected adenocarcinoma in the sigmoid colon.
Outcome
The symptoms resolved in all patients within 1 week except for 1 patient whose symptoms persisted for 19 days. The patient who underwent the sigmoid resection had an uncomplicated postoperative course. Repeat endoscopy gave normal findings in all patients and none presented with recurrence of their colitis.
Pathological findings
Histologic examination was conducted in 3 cases. In 1 of the 2 endoscopic biopsy specimens of colon mucosa a patchy acute ischemic-type mucosal injury was found, characterized by the following: a marked congestion and extravasation of red blood cells in the superficial lamina propria; mucin depletion, attenuation or loss of the epithelium lining, the surface and the upper portion of the glands; a surface erosion with a fibrinohemorrhagic, purulent exudate; preservation of the regenerative base of the crypts; and a scant inflammatory cellular infiltrate. The second endoscopic biopsy specimen consisted of a nonspecific exudative fibrinohemorrhagic and purulent membrane consistent with erosion or ulceration of the mucosa.
In the third case, the morphologic documentation of the lesion was on a surgical sigmoid resection specimen. In addition to the Dukes’ stage B adenocarcinoma and diverticular disease, a patchy erythematous granular and focally eroded mucosa with submucosal edema was found (Fig. 1). Since this change was seen not only proximal to the carcinoma but also distal to it, the possibility of an obstructive colitis was ruled out. As for the possibility of an associated chronic idiopathic inflammatory disease or diverticular disease-associated colitis, these were ruled out on histologic examination, which revealed varying degrees of an acute ischemic-type colitis (Fig. 2).
A fibrinoleukocytic exudate with acute ischemic-type injury, which is limited to the mucosa and submucosa (hematoxylin–eosin stain, original magnification ×100).
Discussion
Glutaraldehyde-induced colitis, first described by Castelli and associates,5 has been well documented subsequently by others.6–10 Hanson and associates8 reported a 1.3% incidence of glutaraldehyde-associated colitis in a series of 388 flexible sigmoidoscopic examinations in a colorectal carcinoma screening program. Their review of the literature revealed a reported incidence that ranged from 0.1% to 4.7%. Likely the true incidence of gluteraldehyde-associated illness is higher than indicated in published reports because of failure to recognize the disorder or the self-limited nature of the illness, with patients attributing symptoms to some other cause.
The macroscopic findings from either endoscopy or surgical specimen have never been published. Histologically, the mucosal damage induced by the inadvertent exposure to glutaraldehyde manifests as an acute ischemic colitis.5,9,11 Other mechanisms that may mimic the condition include bacterial toxins of enterohemorrhagic Escherichia coli,12 or C. difficile,13 nonsteroidal antinflammatory agents,14,15 penicillin derivatives14 and hormonal oral contraceptives.16 Some authors stress that no morphologic feature allows a distinction among the various etiologies.10,17 However, a specific diagnosis will be reached with reliability in most cases through a clinicopathologic correlation.
The toxicity that occurred in our patients is most likely the result of direct contact with the agent. The proposed mechanism of injury was inadequate rinsing of all the endoscopic channels and surfaces with water or contamination of the rinsing water with glutaraldehyde.9 West and colleagues10 found the cause of glutaraldehyde contamination in one of their patients to be inadequate rinsing of the tubing between the water bottle and the endoscope. It is unlikely that the injury was due to the enzymatic cleaner, Metrizyme, as similar solutions have been instilled into the colon of rats with no effect.4 Direct contact of 2% glutaraldehyde with colonic mucosa has been shown in rats to produce necrosis of the crypt epithelium and infiltration with neutrophils similar to the pathological findings seen in our patients.4,6 Solutions of 0.5% revealed only mild changes and more dilute solutions had no discernible effect.
Our hospital infection control team reviewed the endoscopic cleaning, preparation and disinfection procedures and initially failed to find any part of the procedure that posed a threat to patients. After a second review, it was believed that there was gluteraldehyde contamination of the residual water in the internal channel of the colonoscope. Consequently, 2 modifications in our disinfection procedure were immediately instituted. Alterations in the rinsing process and frequent changes of the rinsing water have been incorporated in our office for the flexible sigmoidoscopes. Automated washing machines, although expensive, have been installed for the colonoscopes.
In an assessment of endoscopic cleaning in 19 family practice and internal medicine offices, Jackson and Ball18 found an average of 6.8 deficiencies per office. A short training course was able to reduce the inadequacies of technique to an average of 0.9 deficiencies per office.
Conclusions
Glutaraldehyde-induced colitis is a self-limited condition, characterized by abdominal pain, mucus diarrhea and rectal bleeding. It may occur within minutes or up to 48 hours after endoscopy. The illness may be mild or moderately severe, as in one of our patients. It is important that this entity not be confused with other causes of acute colitis such as ischemic colitis or pseudomembranous colitis. The entity should be recognized so that corrective measures can be made in the endoscopy suite and inappropriate treatment not be administered.
The break in technique that causes glutaraldehyde colitis may be very small and not evident to the endoscopy suite personnel. Therefore, special attention should be given during instrument cleaning to ensure that the glutaraldehyde is completely rinsed off the surface and irrigated through the channels of the endoscope to minimize the risk of insult.
The treatment of this disease is supportive with intravenous fluids reserved for the more severe cases. Metronidazole appears to have no beneficial effect on outcome. Most patients’ symptoms will resolve completely within a week. No long-term side effects are apparent due to direct contact of colonic mucosa with 2% glutaraldehyde.
Footnotes
Poster presentation at the annual meeting of the American Society of Colon and Rectal Surgery, Seattle, Wash., June 1996.
- Accepted March 7, 2000.