Abstract
Introduction: Barrett’s esophagus, a syndrome in which the squamous mucosa that normally lines the distal esophagus is replaced with columnar epithelium, is found in a small percentage of patients presenting with gastroesophageal reflux disease (GERD). The columnar epithelium may be protective, guarding people afflicted with Barrett’s esophagus from experiencing symptoms related to acid reflux. The purpose of this study was to investigate whether people with Barrett’s esophagus subjectively experience fewer symptoms or symptoms of decreased severity, despite sustaining greater acid exposure, than those with GERD but without Barrett’s syndrome.
Methods: We conducted a chart review of patients with GERD. Criteria for inclusion in the study were esophagogastroscopy, motility testing and a 24-hour pH study. Fifty-eight patients (29 men, 29 women) fulfilled these criteria. The diagnosis of GERD was based on an abnormal 24-hour pH study (DeMeester score). Of these 58 patients, 21 (14 men, 7 women) were found to have histologically confirmed Barrett’s esophagus. A questionnaire to assess the key symptoms of GERD was administered, with a severity score ranging from 0 to 3 (3 being the most severe) for each symptom.
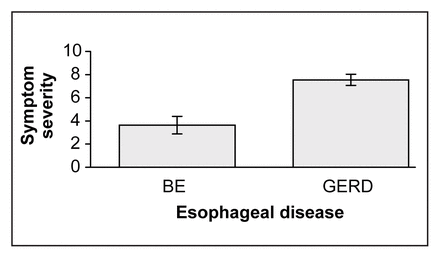
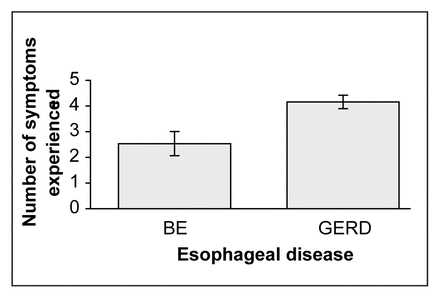
Results: Patients with Barrett’s esophagus experienced symptoms significantly less severe (p < 0.01) than those with GERD. Patients with Barrett’s esophagus also had a greater degree of acid exposure as identified by higher DeMeester scores (p = 0.056), longer episodes of acid exposure, a greater number of long episodes (> 5 min) of acid exposure (p = 0.033) and an increased percentage of time when their pH was less than 4. Patients with Barrett’s esophagus had decreased resting lower esophageal sphincter tone, and number and amplitude of peristaltic contractions.
Conclusions: For patients with Barrett’s esophagus, the columnar epithelium may serve a protective function in guarding against symptoms of acid reflux. This has implications for the diagnosis and management of this condition.
Barrett’s esophagus is a distinct complication of gastroesophageal reflux disease (GERD), occurring in approximately 12% of patients presenting with GERD symptoms.1 It has been reported that patients with Barrett’s esophagus have a greater than 50-fold risk of progression to esophageal adenocarcinoma.2 This risk is so great that Fitzgerald and associates3 considered Barrett’s esophagus to be the single most important risk factor for the development of esophageal adenocarcinoma. Since esophageal cancer has a high death-to-incidence ratio, it is important to identify patients with Barrett’s esophagus before invasive esophageal cancer develops. In this study we attempt to determine the role Barrett’s esophagus plays in the expression of reflux symptoms, as patients will be less likely to seek care for symptoms of lower severity despite the significance of their underlying condition.
Histologic confirmation is paramount in documenting the presence of Barrett’s esophagus. The pathogenic feature of this complication of GERD is the finding of specialized intestinal-like columnar epithelium lining the distal esophagus. In addition to this endoscopically observable feature, increased acid exposure (as measured through 24-hour pH monitoring) and decreased lower esophageal sphincter (LES) tone have been consistently reported as having a positive relationship in the development of Barrett’s esophagus.2–6
We hypothesized that patients with Barrett’s esophagus may experience fewer symptoms despite significantly greater acid exposure than other patients with GERD because the columnar lining may serve a protective function, which limits the experience of symptoms. Unfortunately, patients experiencing fewer and less severe symptoms may not seek medical attention or may be reluctant to comply with the recommended surveillance of their condition. This may increase their chances of death if cancer develops. We sought to determine if this is the case.
Methods
We carried out a chart review of patients assessed from 1993 to 2000 in the esophageal function laboratory at the Toronto General Hospital. Inclusion criteria were esophagogastroscopy, motility testing and an abnormal 24-hour pH study. Only 58 patients (29 men, 29 women) had undergone all 3 tests and met these criteria. In these 58 patients with documented GERD based on an abnormal DeMeester score, 21 patients (14 men, 7 women) had documented Barrett’s esophagus, confirmed histologically by biopsy through the presence of specialized intestinal metaplasia. Six of the 37 patients with GERD alone who had columnar lining on biopsy but without intestinal metaplasia were excluded from the analysis because of uncertainty as to whether the biopsy was taken from the tubular esophagus or from the gastric cardia.
The assessment protocol used for this study was designed to address the key symptoms identified by patients with GERD and Barrett’s esophagus. To this end, we drew up a questionnaire adapting the gastrointestinal symptom rating scale outlined by Svedlund and colleagues in 1988.7 This symptom scale was originally developed to assess gastrointestinal symptoms in irritable bowel syndrome and peptic ulcer disease. Physician and patient input was used to generate items and the scale was validated with an interrater reliability of 0.86–1.00. In addition, the scale can be used either in its entirety or as a pool of items from which appropriate items can be selected to give subscales for different syndromes, with interrater reliability of 0.92–0.94 for the syndromes.7 Symptoms of heartburn, acid reflux, nocturnal reflux, positional reflux, acid regurgitation, nausea and vomiting, abdominal distension, eructation, increased flatus, dysphagia, cough, asthma and hoarseness were included. Within each of these symptoms, a severity score of 0 to 3 (with 3 being the most severe) was identified using the criteria outlined by Svedlund and colleagues.7 For example, under the symptom of heartburn, the severity index was identified as: 0, no or transient heartburn; 1, occasional discomfort of short duration; 2, frequent episodes of prolonged discomfort with requests for relief; or 3, continuous discomfort with only transient relief with antacids.
To obtain a portrait of a patient’s experiences, 2 summary scores were created. The first was a simple sum of the patient’s individual symptom severity scores, creating a general patient severity index. This index ranged from 0 to 39, with 39 being the most intense experience of all listed symptoms (severity of 3 within each of the 13 symptoms). The other summary score was an index of the number of symptoms experienced by the patient (to a maximum of 13 for the 13 listed symptoms). These 2 summary scores were then compared between patients having GERD with and without Barrett’s esophagus.
Categories were also included to identify demographic factors such as date of birth and gender. The participants’ motility test results, 24-hour pH results and pathological (in the case of the Barrett’s patients) findings were also recorded.
Statistical analysis
We used t tests to compare the 2 samples. Prior to the t test, a Levene F test was used to test the equality of the variability of the 2 samples being tested. If the samples had equal variability (identified by a Levene’s F test value of p < 0.01), the value of the t test for samples with equal variability was recorded. Conversely, the value of the t test for samples with unequal variability was recorded for F tests with p values greater than 0.01. A p value of less than 0.05 was considered significant.
Procedure
Patients were identified as having GERD or Barrett’s esophagus from their hospital records. They were then screened for having undergone motility testing, 24-hour pH study and endoscopy. Those who had not undergone these tests were excluded. Each patient’s hospital chart, including a symptom questionnaire that was completed at the time of esophageal function testing, was reviewed. The symptom questionnaire used in the esophageal function laboratory was not the gastrointestinal symptom rating scale of Svedlund and colleagues, 7 but included the symptoms we wished to study and was completed temporaneously by the patient and the nurse working in the mobility laboratory. Data transcribed from these events and recorded in the assessment protocol were used to identify the patient’s symptoms and the severity of these symptoms. Histologic confirmation of Barrett’s esophagus was obtained through the pathology report.
Results
Patient characteristics
Table 1 displays the demographics of the patients assessed in this study. Although significantly more females than males had GERD irrespective of the presence or absence of columnar epithelium (p < 0.01), a prominent difference between the 2 patient groups was that there were significantly fewer women in the group with Barrett’s esophagus than in the group with GERD alone (p < 0.01).
Demographic Characteristics of 58 Patients With Gastroesophageal Reflux Disease (GERD) Alone or GERD With Barrett’s Esophagus
Differences in acid exposure
Patients with Barrett’s esophagus experienced significantly greater acid exposure as demonstrated by higher DeMeester scores, longest reflux episode, more reflux episodes lasting longer than 5 minutes, and a greater percentage of time during a 24-hour period within which the pH value was less than 4 (Table 2).
Findings* in Patients With Gastroesophageal Reflux Disease (GERD) Who Have Barrett’s Esophagus, Compared With Findings in Patients Having GERD Alone
Differences in motility and lengths
Although patients with Barrett’s esophagus and with GERD alone had similar lower esophageal and intra-abdominal sphincter lengths, LES tone was significantly lower in patients with Barrett’s esophagus than patients with GERD alone (p = 0.03). The former group was also found to have significantly diminished peristalsis in the body of the esophagus (p = 0.04), as well as diminished lowest (p = 0.006) and highest amplitude (p = 0.003) of peristalsis. The prevalence of hiatus hernia differed in that a hiatus hernia was identified in 91% of patients with Barrett’s esophagus and only 55% of those with GERD alone (p = 0.007).
Symptoms
Table 2 shows the difference between those with Barrett’s esophagus and those with GERD alone with respect to the severity of the symptoms they experienced and the number of different types of symptoms experienced (Fig. 1 and Fig. 2). Significantly lower symptom severity (p < 0.01) was experienced by patients with Barrett’s esophagus.
The severity of symptoms experienced by patients with Barrett’s esophagus (BE) and with gastroesophageal reflux disease (GERD) alone.
The number of symptoms experienced by patients with Barrett’s esophagus (BE) and with gastroesophageal reflux disease (GERD) alone.
Discussion and conclusions
The results of our study demonstrate that patients with histologically confirmed Barrett’s esophagus experience decreased severity of symptoms despite proven increased acid exposure when compared with patients suffering from GERD without Barrett’s epithelium. Johnson and associates8 first reported that patients with Barrett’s esophagus had decreased acid sensitivity, based on the result of an acid perfusion test. In a study of quality of life comparing patients with GERD and those with Barrett’s, Eloubeidi and Provenzale9 found no significant difference in quality-of-life scores, but those with Barrett’s syndrome were more likely to describe their symptoms as mild versus moderate or severe.
This has implications in the surveillance of patients with Barrett’s esophagus as these patients may discontinue treatment and routine surveillance if their symptoms are minimal. Several reports in the literature have documented that when medical therapy is used in patients with Barrett’s syndrome, symptom control does not equate with control of acid reflux.10–12 This has significant implications if continued uncontrolled acid reflux promotes carcinogenesis in patients with Barrett’s esophagus. The lack of symptoms they experience may contribute to the delay in diagnosis of cancer and the high ratio of incidence to mortality. In addition, this may explain the low diagnosis or prevalence rate of Barrett’s esophagus in the general population. Thus, the primary contribution of our study is the need for education on the paradoxical nature of the symptom–disease relationship.
The contribution of acid exposure on the development of Barrett’s esophagus has been reported consistently in the literature.2–6,13 Our study further reinforces the reported significance on the rate of occurrence of Barrett’s esophagus of the number of reflux episodes lasting longer than 5 minutes.3,4 In addition to the contribution of acid exposure, the influence of LES in Barrett’s syndrome has also been reported.2,4,6,13 The results obtained in our study are consistent with those in the literature and further reinforce the role that LES plays in the rate of occurrence of Barrett’s esophagus.
Of interest was the finding that a higher proportion of men had Barrett’s syndrome even though GERD is more common in females. This is consistent with the predominance of men in adenocarcinoma of the esophagus. Gender may be an underlying factor predisposing to the development of segments of columnar epithelium within the esophagus.
Limitations of this study extend from its design. This was a retrospective study, so only those factors identified by patients and recorded by their physicians were included. This may have resulted in only those factors most important to a patient being identified; some of the more minor inconveniences may have been ignored. In addition, most patients with columnar epithelium were male. This gender distinction may have introduced symptom-reporting differences that occur as a result of gender roles. Another limitation of this study is the small sample size.
This study has documented decreased severity of symptoms in patients with histologically confirmed Barrett’s esophagus despite objective evidence based on 24-hour pH study of increased acid exposure. Also, impaired esophageal motility, including diminished amplitude and decreased total percentage of peristaltic contractions, was identified. Along with reduced resting LES tone this impairment in motility contributes to prolonged acid exposure in the esophagus and promotes ongoing mucosal damage. Symptom control is an unreliable end point for esophageal antacid therapy (both medical and surgical). This is important in the diagnosis and surveillance of patients with Barrett’s esophagus.
Footnotes
Presented at the annual meeting of the Canadian Association of Thoracic Surgeons, London, Ont., Sept. 19–22, 2002.
Competing interests: None declared.
- Accepted November 3, 2003.