Abstract
Introduction: Colorectal carcinoma (CRC) is an ideal tumour for population-based screening policies. Screening guidelines are based on differing levels of evidence and opinion. The objective of this study was to determine current CRC screening practices in Alberta.
Methods: A questionnaire was mailed to all family physicians, gastroenterologists, general and colorectal surgeons, and general internal medicine specialists in a large Canadian urban centre. Results tabulated included response rate, specialty, presence/absence of a screening policy, and policy type.
Results: The 922 surveys mailed resulted in a 61% response rate. Among the responses, 58% (95% confidence interval [CI] 53.6%–62.6%) of physicians recommended screening to healthy individuals without a family history of CRC, and 96% (CI 94.7%–98%; p < 0.0001) to those with a family history. For patients without a family history of CRC, the majority of physicians chose the age of 50 to initiate screening (63%) by fecal occult blood testing (79%) or, less frequently, colonoscopy (26%). Screening frequency varied by the modality used. In individuals with a family history of CRC, most physicians initiated screening prior to the index case (45%) or by age 40 (31%), mostly with colonoscopy (84%); screening frequency varied mainly by modality.
Conclusion: Despite the evidence supporting CRC screening in all persons at average risk, only 58% of physicians currently recommend screening to patients with no family history of CRC. Fecal occult blood testing was recommended most frequently in this subgroup, whereas most physicians screen people with a family history of CRC via colonoscopy, suggesting that they believe it to be a superior screening modality. As this looks to be the trend in practice, a randomized controlled trial comparing fecal occult testing with screening colonoscopy is needed.
Colorectal carcinoma (CRC) is a leading cause of cancer-related death in Canada.1 It is an ideal tumour for screening, with its high incidence, long lag time between adenomatous polyp and carcinoma, and increased potential for curative treatment when detected at an earlier stage.2 Level I evidence3–6 confirms that mortality rates decrease with fecal occult blood testing (FOBT); however, evidence for the ideal or best screening program is unclear. Guidelines are therefore based on differing levels of evidence and expert opinion.
The aim of this study was to determine current screening practices in an urban centre for asymptomatic individuals with and without a family history of CRC. The acquired data will be used to develop a randomized controlled trial to determine the ideal screening method against CRC.
Methods
After review of current CRC screening guidelines, the literature and discussion with colleagues, a content expert developed a screening questionnaire. One page long and containing 9 closed-ended questions and an area for additional comments, it was designed to be “respondent-friendly.” On average, it took less than 5 minutes to complete.
The questionnaire was mailed to all family physicians, gastroenterologists, general and colorectal surgeons, and general internal medicine specialists (including medical oncologists) in Calgary, Alberta. A total of 922 were mailed; results were tabulated after 1 mailing, including response rate, type of practice/specialty, use or absence of a screening policy, and type of policy (patient age at initiation, tests employed and frequency). Descriptive statistics were used to present the results; 95% confidence intervals (CIs) were used to estimate the true proportion of physicians with a CRC screening policy in Alberta.
Results
Of the 922 letters mailed, 563 were returned, for an overall response rate of 61%. Ninety-five surveys were not completed for various reasons, e.g., the physician had moved, retired or was deceased, or had changed specialties and was no longer involved in primary care of patients. The remaining 468 were entered for analysis. Response rates varied depending on specialty (Table 1).
Questionnaire return and use of a colorectal (CR) screening policy (in %) for patients with no family history of colon cancer
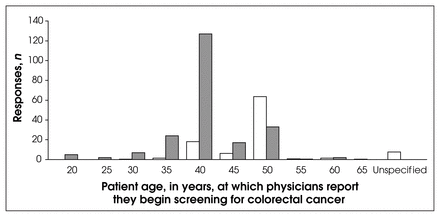
Of the 468 responses considered, 272 physicians (58.1%, CI 53.65%–62.59%) had a screening policy for healthy individuals without a family history of colon cancer. Again, this varied according to specialty (Table 1). Overall, 41.9% of the physicians surveyed had no screening policy for this population subgroup. For the 272 physicians using a screening policy, 50 years was by far the most common age (173 responses, 63.6%) to initiate screening, followed by 40 years (18.0%) as the next most common. Policy ages for initial screening ranged from 30 to 65 years (Fig. 1).
Initial screening ages chosen by physicians for healthy adults with no family history of colon cancer (white columns) and those who do have a family history (dark columns).
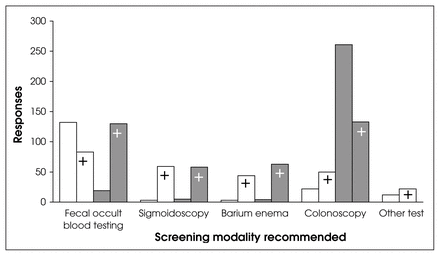
Screening modalities recommended by respondents (Fig. 2) included FOBT, sigmoidoscopy, air-contrast barium enema, colonoscopy and “others”: physical examination, digital rectal exam, complete blood count, total iron binding content, serum alkaline phosphatase and risk-factor assessment. FOBT was employed most commonly, either alone (132 of 272, 48.5%) or in combination with other modalities (83, 30.5%). Screening with colonoscopy, either alone (22, 8.1%) or in combination with other tests (50, 18.4%) was the policy of 72 respondents. (Physicians could choose more than 1 screening modality, so values do not add up to 100%.) Frequency of screening varied mainly by the modality used: FOBT, annually; colonoscopy, every 5 years (see Table 2).
Screening modality recommended for healthy adults with no family history of colon cancer (white columns) and those who do have a family history (dark columns): plain columns, that modality alone; columns with crosses, in combination with other screening tests.
Screening frequency in years, by family history of colorectal cancer
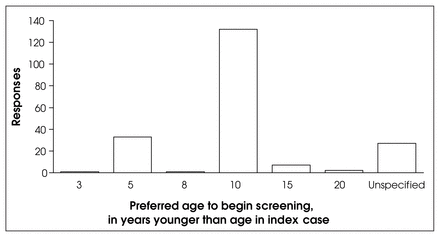
For asymptomatic individuals with a family history of colon cancer, the majority of physicians (451 of 468, 96.4%; CI 94.67%–98.05%) reported that they employ a screening policy, including all gastroenterologists and surgeons surveyed. However, 2.5% of family physicians and 18.91% of general medicine specialists had no screening policy for this population subgroup. A notable number of physicians (204/451, 45.2%) initiated screening sometime before the index case in the family, ranging from 3 to 20 years prior (Fig. 3). A second group (247, 54.8% gave absolute recommendations for initiation of screening ranging from age 20 to 60 with a mean of 40 (Fig. 1).
Initial screening ages chosen by physicians for healthy adults with a family history of colon cancer, in years younger than the age of the first affected family member.
Colonoscopy was the most common screening modality chosen for those with a family history of CRC (Fig. 2). It was used either alone (261/468, 55.8%) or along with other modalities (133, 28.4%) in an overall 84.2% of responses. FOBT was employed by 31.8% of our respondents, alone (19, 4.1%) or in combination (130, 27.8%; Fig. 2). Again, the frequency of screening varied by modality (FOBT, every year; colonoscopy, every 5 years).
Discussion
The overall response rate of 61% is comparable to most survey or questionnaire studies directed at physicians. Cummings and associates7 noted an average 61% response rate to surveys mailed to physicians overall, decreasing to 52% for large surveys involving over 1000 observations. The mean response rate to surveys of physicians published in 1991 was 54%.8 Further contacts (i.e., second or third mailings or telephoned encouragement) may have increased our response rate.9,10
Level I evidence3–6 shows screening for CRC in the general population with FOBT can reduce mortality from CRC. The Canadian Task Force on Preventive Health Care (CTFPHC) recommends this screening modality in all asymptomatic individuals over the age of 50.11 Despite this, many physicians (about 42% of those who responded to our questionnaire) have no screening policy whatsoever. Despite a report12 that in Texas, 82% of primary care physicians self-reported recommending FOBT and 87% flexible sigmoidoscopy, a 1999 American survey13 of the general population found that only 20.6% of respondents over 50 had been screened by FOBT within the past year, and 33.6% by sigmoidoscopy or colonoscopy within the last 5. In Michigan, Ruffin and colleagues14 found that over half had not been screened during the preceding testing period (1 yr, FOBT; 5 yr, other tests). Finally, a 1985 Canadian survey15 of family physicians in New Brunswick noted that only 20% recommended FOBT as a screening policy. The number of physicians in Canada with a screening policy for CRC has increased considerably since 1985, as our study confirms; but still some 40% of physicians do not employ a screening strategy for CRC. As well, 60% may be an erroneously high estimate of actual screening rates; surveys in the USA13,14 have disclosed that compared with responses from the population, physicians may overestimate the screening taking place.
Our study was not designed to determine reasons for the lack of a screening policy. However, potential reasons include:
Current information has not been disseminated broadly.
There is a lack of belief in the results of recent trials.
The best screening strategy is still undetermined.
Small reductions in mortality from CRC (from current studies) may not result in a large population benefit.16
Information on compliance and cost is still relatively unavailable.
The modality of choice for screening healthy adults with no family history of colon cancer varied considerably. In our study, the majority of physicians with a screening strategy 215/272 (79.0%) did recommend FOBT, either alone or in combination with other methods. However, the sensitivity of this modality ranges from 46% to 92%, prompting concern that it may not the best test.3–6,16
Sigmoidoscopy used alone was infrequently recommended (3/272), but more frequently in combination with FOBT (17 responses) or multiple tests (42). Well-designed prospective trials17–19 have shown a decrease in mortality rates with sigmoidoscopy; concerns remain, since at least one-third of lesions may be out of reach of the flexible sigmoidoscope. 20–24 Barium enema, which is often more accessible than sigmoidoscopy or colonoscopy, was recommended by 30 respondents (11.0%) either alone or in combination with other methods. However, recent data confirms the superiority of colonoscopy over air-contrast barium enema for detection of adenomatous polyps.25
Colonoscopy has the highest sensitivity and specificity to detect polyps or CRC.25 About one-quarter of physicians with a screening policy in this survey already recommend colonoscopy, either alone (22, 8.1%) or in combination with other tests (50, 18.4%). However, data supporting its use as a screening modality is controversial. Specific concerns include possible complications, cost and resource issues.
Of the physicians in this survey, 84.19% use colonoscopy as part of their policy in individuals with any family history of colon cancer. Although opinions and guidelines for CRC screening are stronger for those with a positive family history, CTF-PHC does not recommend any additional screening in this subgroup.11 However, many groups support the use of screening colonoscopy in this population subgroup,22,26–29 including the US Preventive Services Task Force, American Society of Colon and Rectal Surgeons, American Cancer Society, American Gastroenterological Association and others. Now that the forces of recommendations, guidelines and public education have combined, it may not be possible to design studies comparing different screening modalities to find the ideal in this population. Colonoscopy in this risk subgroup has become the recommended screening tool before level I evidence has been established supporting its use.
The major finding of this survey is that FOBT and colonoscopy are the 2 main modalities considered for CRC screening. The different implications of the 2 types of screening are obvious: FOBT, which is supported by level I evidence, has a sensitivity of 46%–92%,3–6,16 prompting concerns that it may not be the best test. Towler and colleagues16 estimated that 10 000 people would have to be offered biennial screening to prevent 8.5 deaths from CRC over 10 years. This relatively small clinical benefit is another major criticism.
In contrast, screening colonoscopy is the best method for diagnosing adenomatous polyps and carcinoma. 11,25 In the healthy population over 50, incidences of advanced neoplasia (≥ 1-cm adenomatous polyp, villous adenoma, high-grade dysplasia or cancer) of 5.6%23 to 10.5%24 have been reported, suggesting a potentially high clinical impact. The superior sensitivity of screening colonoscopy should translate into improved mortality data, although this has yet to be tested in a randomized trial. Furthermore, prevention of CRC with avoidance of a major operation and adjuvant therapy may be the best way to measure “success” with screening colonoscopy.
Having said that, there is no doubt that screening colonoscopy is controversial. Concerns about possible complications and cost and resource issues remain. Rates of serious complications from screening colonoscopy are recently estimated at 0.2%–0.3%.24,30,31 Perforation at colonoscopy is associated with up to a 14% chance of mortality, in one series.32 Nevertheless, in more than 3000 cases in a multi-centre trial, Lieberman and coworkers24 reported no perforations or deaths related to colonoscopy. Costs and resources may be less worrisome if the ideal frequency of screening is low (i.e., every 5–10 years if results are normal). All screening methods seem to be cost-effective, with several investigators33–36 estimating costs at about US$10 000–$25 000 per year of life saved. Overall costs should be less in the Canadian health care environment.
Further screening studies need to be undertaken to establish realistic cost–benefit ratios at individual and society levels in Canada. A prospective, randomized controlled trial to compare FOBT (with or without sigmoidoscopy) and screening colonoscopy represents the best design. It would compare recommendations supported by level I evidence to the current trend in real practice. Although some feel there is enough indirect evidence to support screening colonoscopy for the general population, a randomized controlled trial would answer such important questions as, by how much would each screening modality reduce CRC incidence and mortality? And what are the true costs, complication rates, compliance rates and resource implications of widespread screening in the general population?
Conclusion
Despite evidence-based medicine supporting CRC screening, 42% of physicians in our survey had no screening policy for this common neoplasm. Reasons for a lack of screening need to be determined.
For physicians with a screening policy in CRC, the majority (79%) employed fecal occult blood testing; about one-quarter recommended colonoscopy. Individuals with any family history of CRC were screened using colonoscopy by the majority (~85%). A randomized controlled trial to determine the ideal screening modality is the next step in the battle against colorectal carcinoma.
Footnotes
Competing interests: None declared.
- Accepted November 26, 2003.