Abstract
Background: To review morbidity and mortality of patients undergoing elective, open repair of infrarenal abdominal aortic aneurysms and were admitted postoperatively to a surgical stepdown unit rather than routinely to the intensive care unit (ICU), we carried out a retrospective review.
Methods: All patients undergoing this type of repair in our centre, a division of vascular surgery in a tertiary-care teaching hospital in Ontario, over a 27-month period were reviewed. A consecutive 230 patients who underwent aneurysm repair from September 1999 through November 2001 were routinely admitted to a surgical stepdown unit postoperatively, with only a minority of patients requiring admission to ICU. We reviewed the rate of initial ICU admission and that of subsequent ICU admission after stepdown-unit admission. We also assessed morbidity, mortality and length of hospital stay for patients admitted to ICU as well as those admitted to the stepdown unit.
Results: ICU admission was avoided in 204 (89%) of these patients. The remaining 26 patients (11%) required ICU admission at some point during their hospital stay. Only 3 patients (1%) originally admitted to the stepdown unit subsequently required postoperative admission to ICU.
Conclusions: Our experience demonstrates that proper preoperative assessment and selection allows the majority of elective infrarenal aneurysm repairs to be safely cared for postoperatively in a stepdown unit, and that subsequent ICU admissions are rare.
Traditional elective, open repair of infrarenal abdominal aortic aneurysms (AAA) has a reported mortality rate ranging from 4.8% to 8.4%.1,2 Because of this high perioperative risk, most patients undergoing elective aortic surgery have been traditionally admitted to the intensive care unit (ICU) for the first postoperative day3,4 (POD) for monitoring. As ICU resources become more limited, increasing constraints have been placed on elective ICU admissions, and alternatives are being explored for routine care after AAA repair. Selective utilization of the ICU based on individual patient factors has been reported; direct admission to the surgical ward was found to be safe and cost-effective in up to 48% in one series of patients5 and 88% in another.6 To determine practice patterns here in Canada, we surveyed the current membership of the Canadian Society for Vascular Surgery (CSVS) and found that 33 of 43 respondents (77%) routinely admit their patients to the ICU postoperatively.
In our centre, postoperative infrarenal AAA patients are routinely admitted to a stepdown unit (SDU) on a surgical ward where patients undergo noninvasive hemodynamic monitoring by certified nurses. Only a minority of patients are admitted to the ICU, because of comorbid medical conditions and individual patient factors determined either pre- or perioperatively. In this report we retrospectively reviewed the results of routine admission to the SDU for mortality, major morbidity, length of stay (LoS) and need for subsequent ICU admission.
Methods
Patients
Using our Vascular Registry data we retrospectively reviewed the consecutive, elective, open AAA repairs performed at a tertiary care referral centre. From September 1999 through November 2001, 230 such repairs were performed at London Health Sciences Centre, Victoria Campus by 3 vascular surgeons. This report includes only patients who underwent standard open, infrarenal AAA repair where the repair was constructed at or below the renal arteries, regardless of the positioning of the proximal aortic clamp. Patients were excluded if they had ruptured aneurysms, a suprarenal component, aortobifemoral grafting for occlusive disease or endovascular repairs. Charts were reviewed for all patients requiring ICU admission at any point during their hospital stay. Mean age, comorbid medical problems, risk factors and outcomes were compared with those of patients judged suitable for direct postoperative admission to the SDU.
Protocol
Before the operation, patients were reviewed by the surgeon responsible and by internal medicine and anesthesiology consultants in a preadmission clinic. A preoperative decision was made for postoperative admission either to the SDU or ICU, depending on their comorbid medical conditions (Box 1). Patients with severe coronary artery disease (CAD), chronic obstructive pulmonary disease (COPD) or chronic renal failure and on dialysis were slated for direct ICU admission. Patients in the postoperative period who in the opinion of the anesthesiologist needed continued ventilatory support and those who had sustained perioperative hypotension, transfusion requirements > 3 L, hypothermia or (based on electrocardiogram [ECG] monitoring) clinically significant perioperative cardiac ischemia or arrhythmias were admitted to the ICU, either directly from the operating room or from the postanesthesia care unit (PACU).
Indications for admission to the ICU
Planned preoperative
Coronary artery disease
— Congestive heart failure
— Ejection fraction < 40%
— NYHA class III or IV angina
Chronic obstructive pulmonary disease
— Home oxygen
— FEV1 < 1 L
Chronic renal failure
— Dialysis-dependent
Perioperative
Sustained hemodynamic instability (< 90 systolic)
Cardiac ischemia, arrhythmia
Need for ongoing ventilation
Hypothermia (< 35°C)
Transfusion > 3 L
FEV1 = forced expiratory volume in 1 second; ICU = intensive care unit; NYHA = New York Heart Association
Operative technique
Patients were admitted to hospital on the day of surgery, without bowel preparation. Epidural analgesia was routine unless contraindicated, followed by general anesthesia and placement of radial arterial and central venous catheters. Pulmonaryartery catheters were used at the discretion of the anesthesiologist, according to the patient’s underlying cardiopulmonary status. All aortic procedures were performed through a standard midline, transperitoneal approach.
Postoperative care
Patients deemed eligible for admission to the SDU were extubated in the operating room at the completion of the procedure or in the PACU shortly thereafter, according to the anesthesiologist’s assessment of their oxygenation, ventilation, analgesia and overall clinical status. Those entering the PACU were monitored for approximately 3 hours, after which, if their condition was stable, they were transferred to the SDU. This unit is on the vascular surgery ward, which is remote from the ICU. Arterial lines were removed before the transfer to the SDU. The remaining patients were admitted to the ICU.
The staff in the SDU were ECG-certified vascular nurses with a 3:1 patient–nurse ratio. Medical coverage was provided by in-house general surgery residents and the internal medicine on-call service.
Patients underwent noninvasive cardiac monitoring overnight and, if their condition was judged to be stable, were transferred to a standard surgical-ward bed the next day. They were encouraged to ambulate on POD 1; diet was resumed on POD 2 and advanced according to patient tolerance. Oral analgesics were prescribed when the epidural and Foley catheters were removed, on about POD 3. Patients were discharged home when tolerating a normal diet and comfortable resuming a normal activity level.
Outcome measures
Main outcome measures were mortality, major medical morbidity, hospital and ICU LoS, and rate of subsequent ICU admission from SDU. Types of complications were defined as follows: cardiac, as myocardial infarction, arrhythmia or congestive heart failure; pulmonary, as pneumonia or atelectasis requiring ventilatory support or prolonging the patient’s stay; and renal, as acute renal failure requiring dialysis.
Results
Patient demographics
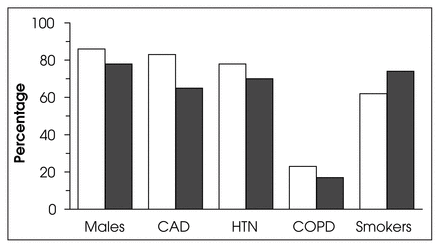
Patients initially admitted to the ICU (n = 23, 10%) were compared with those admitted to the SDU (n = 207, 90%; Fig. 1). Although the ICU group had more severe medical problems, culminating in their selection for direct ICU admission, the groups were similar in age (mean 72 yr in either group), gender ratio and overall prevalence of risk factors such as severe CAD, COPD, hypertension and smoking.
Risk factors and comorbid conditions of patients taken to the stepdown unit (white columns) or intensive care unit (dark columns) for postoperative care. CAD = coronary artery disease; HTN = hypertension; COPD = chronic obstructive pulmonary disease.
Mortality and morbidity
Overall, 6 study patients (2.6%) died. Group mortality and morbidity rates are compared in Table 1.
Mortality and morbidity in study patients admitted to the stepdown and intensive care units
ICU admissions
With use of the SDU, we were able to avoid 204 ICU admissions (89% of patients). The remaining 26 patients (11%) required ICU admission at some point during their hospital stay. Among these, 23 (10% of all study subjects) were admitted directly to ICU from the operating or recovery room; 12 of those admissions were planned preoperatively. All those taken to ICU were ventilated, to be weaned there later.
Only 3 patients originally admitted to the SDU (1% of study participants) required subsequent admission to ICU in the postoperative period (Table 2). One had a large pulmonary embolus on POD 7 requiring ICU admission. The second patient had a wound dehiscence on POD 12 and needed ventilatory support in the ICU after closure. The third underwent urgent laparotomy on POD 1, which revealed an omental infarct, and similarly required ventilation in the ICU afterward. All 3 patients survived their complications. Upon discharge from the ICU, they spent additional time in the SDU before transfer to a ward.
Subsequent ICU admissions from stepdown unit
Length of stay
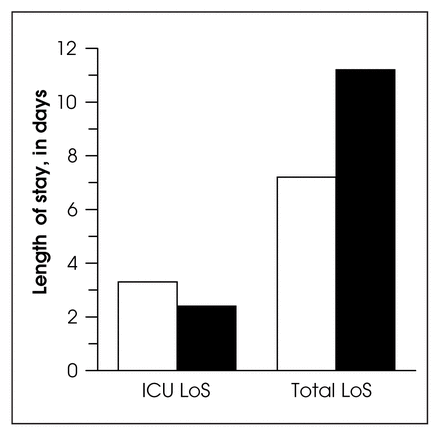
The overall average LoS was 7.7 days; the mean LoS for each group is illustrated in Fig. 2. Mean total hospital stay was 7.2 days for the 207 patients in the SDU group and 11.2 days for the 23 people in the ICU group. Three patients in the SDU group required subsequent ICU admission for periods of 2, 3 and 5 days respectively, for an average ICU stay of 3.3 days. Patients admitted directly to the ICU had an average LoS there of 2.4 days.
Mean length of stay (LoS) in the intensive care unit (ICU) for the groups admitted postoperatively to the stepdown unit (white column) and ICU (dark column), along with the mean total stay in hospital for each group. (Note that only 3 patients in the SDU group spent time in the ICU.)
Discussion
Because of serious perioperative risk and associated medical comorbidities, postoperative admission to the ICU has been traditional after elective, open, infrarenal AAA repair. Selective use of the ICU based on individual patient factors has been found to be safe and cost-effective: Bertges and colleagues5 showed that ICU admissions could be avoided in up to 48.6% of their patients. Podore and Throop6 found that up to 88% of their postoperative patients could be admitted directly to an SDU as part of a clinical pathway to shorten hospital stays. When CSVS members were surveyed, 77% reported still routinely admitting their patients to the ICU postoperatively. In our centre, postoperative infrarenal AAA patients are routinely cared for in a vascular surgical SDU with only a minority of patients being admitted to the ICU because of comorbid medical conditions. Complete care for these patients is the responsibility of the vascular surgery team. This has been very effective in our institution; we attribute this to our preoperative assessment and use of the selection criteria for ICU admission as outlined.
Our overall mortality rate in this series of 230 patients was 2.6%, which is lower than many published rates from the literature. The Canadian aneurysm study1 reported an overall mortality rate of 4.8% for a group of similar patients. The difference may be partly explained by the fact that during this same 26-month period we carried out 99 infrarenal, endovascular aneurysm repairs, a procedure currently limited to high-risk medical patients. Had our surgeries been done as traditional open procedures, the overall mortality and ICU admission rates would likely have been much higher. Thus, we have demonstrated that SDU admission is safe for most patients undergoing open AAA repair, with no deaths in our cohort of patients treated postoperatively in the SDU rather than the ICU.
Although ICU admission for the purposes of more intensive monitoring has been traditional following AAA repair, the literature does not support any added benefit from more invasive monitoring with pulmonary-artery catheters.7,8
Not unexpectedly, the mortality rate was higher in the group initially admitted to the ICU (17%): these patients preselected for ICU admission were known to be at higher risk. Major medical morbidity also differed between the groups; the higher incidences of complications can also likely be attributed to preselection. The cardiac complication rate of 15.5% in patients admitted to the SDU, which is considerable, is nonetheless consistent with published rates, ranging from 11.5%5 to 23% (the Canadian Aneurysm Trial).9 Again, the literature does not support more aggressive routine cardiac investigation; selective investigation based on clinical factors appears to be adequate.10–12
The average hospital stay for all our study patients, 7.7 days (standard deviation 4.5 d), approximates reports from the literature (5.9–9.9 d).3,4 Differences in average LoS for patients admitted to the SDU (7.2 d) and ICU (11.2 d) also likely reflect the medical comorbidities in the ICU group, as do incidences of postoperative morbidity.
ICU admission was avoided for 89% of patients undergoing elective, open infrarenal AAA repair. Only 3 patients (1%) originally admitted to the SDU subsequently required admission to the ICU during the postoperative period. The complications precipitating their ICU admissions occurred after the first postoperative day, which is when most centres that routinely admit patients to the ICU postoperatively transfer patients to the surgical ward. Considering the nature of the complications, it seems unlikely that they would have been avoided had we admitted these 3 patients to the ICU immediately after surgery.
Our experience demonstrates that the majority of elective infrarenal AAA patients undergoing open repair can be safely admitted to an SDU for postoperative management, and that routine postoperative ICU admission is unnecessary. Being able to provide safe, effective postoperative care while sparing valuable and costly ICU resources is an important benefit of this approach.
Footnotes
This work was presented at the 24th Annual Meeting of Canadian Society for Vascular Surgery, October 2002, Halifax, NS.
- Accepted October 24, 2003.