Abstract
Background: There are no comprehensive evidence-based guidelines for deep vein thrombosis (DVT) prophylaxis in patients undergoing minimal-access surgery (MAS).
Methods: We completed a cross-sectional survey of general surgeons practising in Ontario, in order to establish current practice patterns for DVT prophylaxis for MAS procedures.
Results: The mean duration of practice of respondents was 15.4 years, with most (67.0%) practising outside an academic centre. For minor MAS, most surgeons do not give DVT prophylaxis (73.8% in laparoscopic cholecystectomy and 63.7% in laparoscopic inguinal hernia repair). For major MAS, a minority of surgeons do not give DVT prophylaxis (4.1% in laparoscopic colorectal surgery and 13.6% in laparoscopic splenectomy). However, there remains considerable variation in the mechanism (pharmacological, mechanical), approach and duration (perioperative, postoperative) of DVT prophylaxis among respondents in all case scenarios evaluated. Academic surgeons and surgeons in practice for 15 years or less are more aggressive with preoperative heparin administration.
Conclusions: There is substantial and important variability in the current practice of general surgeons with respect to DVT prophylaxis for MAS. Considerable benefit will be derived from clinical trials that provide data to establish appropriate DVT prophylaxis guidelines for MAS.
There are no evidence-based guidelines for deep vein thrombosis (DVT) prophylaxis in minimal-access surgery (MAS). To date, the safety and efficacy of DVT prophylaxis has not been established for either minor or major MAS procedures. Moreover, the risk of developing a DVT following MAS has not been well characterized.1
Anecdotally, it is suggested that the risk of DVT may be reduced following MAS procedures, paralleling the overall reduction in postoperative morbidity. However, some have theorized that the risk of DVT may increase following MAS, largely related to the physiological impact of a pneumoperitoneum (i.e., compression of the inferior vena cava impedes venous return and worsens venous stasis).2–6 In addition, positioning of patients for MAS (reverse Trendelenburg position) has also been considered a risk factor for DVT.2,7 Concern has also been raised by a study that identified a 55% incidence of DVT following laparoscopic cholecystectomy.8 Despite these concerns, other large patient series have not shown a high incidence of DVT or thromboembolic complications following laparoscopic cholecystectomy. In a retrospective review of 587 patients following laparoscopic cholecystectomy, no symptomatic DVT or pulmonary embolism were detected.9 Furthermore, a literature review of 153 832 MAS procedures documented a low incidence of DVT (0.03%) and pulmonary embolism (0.06%).10
This study examines current DVT prophylaxis strategies for patients undergoing minor and major MAS procedures. Surgeons’ responses are compared and contrasted with findings for comparable open procedures. These data will establish current practice patterns and provide insight into the approach of surgeons to DVT prophylaxis for MAS. These data will also serve to provide a basis for further clinical studies.
Methods
We identified 599 surgeons in the province of Ontario through an online database made available to us through the Centre for Minimal Access Surgery at St. Joseph’s Healthcare in Hamilton. A survey was sent to each of these 599 surgeons; 2 follow-up surveys were sent to nonresponders (at 1-month intervals). Data were collated over this 3-month period.
The surgeons were asked to select the management options they would consider for DVT prophylaxis on the basis of 8 clinical vignettes (Box 1). The vignettes represented 2 minor MAS procedures and 2 major MAS procedures as well as their conventional open counterparts. The management options for prophylaxis are presented in Box 2.
Clinical vignettes
A 30-year-old patient presents with cholelithiasis and classic biliary colic. He/she is booked for laparoscopic cholecystectomy as an outpatient procedure. There are no risk factors for thrombosis. Which of the options listed in Box 2 would you consider for DVT prophylaxis?
A 30-year old-patient presents with cholelithiasis and classic biliary colic but refuses laparoscopic surgery because of a complication that occurred with a family member. You therefore plan an open cholecystectomy. Which of the options listed in Box 2 would you consider for DVT prophylaxis?
A 40-year-old man comes in with a reducible left inguinal hernia. You recommend an open tension-free mesh repair. Which of the options listed in Box 2 would you consider for DVT prophylaxis?
A 40-year-old man presents with a recurrent inguinal hernia following open repair. You suggest a laparoscopic repair (done by yourself or a colleague). Which of the options listed in Box 2 would you consider for DVT prophylaxis?
A 55-year-old patient presents with biopsy-proven cecal carcinoma. An open right hemicolectomy is recommended. His/her past medical history is otherwise unremarkable (the findings of preoperative laboratories, coagulation status, CXR and ultrasonography of the abdomen are normal). Which of the options listed in Box 2 would you consider for DVT prophylaxis?
A 60-year-old patient presents with a 3-cm sessile polyp in the cecum. You consider a laparoscopic right hemicolectomy. His/her past medical history is otherwise unremarkable (the findings of all preoperative investigations are normal). Which of the options listed in Box 2 would you consider for DVT prophylaxis?
A 50-year-old patient with ITP is referred to you for an open splenectomy. The patient has had a previous Hartmann procedure and reconstruction for complicated diverticular disease. The platelet count is about 50, and the coagulation profile is normal. Which of the options listed in Box 2 would you consider for DVT prophylaxis?
A 40-year-old healthy patient presents with ITP. The platelet count is 40, and coagulation status is normal. The patient is thin and has had no previous abdominal surgery. You consider a laparoscopic splenectomy for this patient. Which of the options listed in Box 2 would you consider for DVT prophylaxis?
CXR = chest x-ray; DVT = deep vein thrombosis; ITP = idiopathic thrombocytopenic purpura.
Deep vein thrombosis prophylaxis options
No medical prophylaxis
Graded compression stockings
Intermittent pneumatic compression devices
Heparin started preoperatively
Heparin started postoperatively
LMWH (low-molecular-weight heparin) postoperatively
Obtain a hematological or medical consultation
Other ____________________________
Data were analyzed as follows: DVT prophylaxis strategies were broken down into 3 groups: no prophylaxis, mechanical prophylaxis or pharmacological prophylaxis. The χ2 test or Fisher’s exact test was used to compare the proportions of surgeons using prophylaxis between MAS and open surgery for each of these 3 categories (for both minor and major procedures). An analysis of the approach to DVT prophylaxis for major procedures was performed by stratifying pharmacological and mechanical prophylaxis strategies into the most common approaches. Preoperative heparin was considered a relatively aggressive strategy; thus, we specifically examined the proportions of surgeons using this technique. We further examined the influence of years in practice and practice location (academic v. nonacademic) on the use of DVT prophylaxis. Given that this study was hypothesis generating, we have chosen not to adjust our levels of significance for multiple comparisons. We acknowledge that the number of comparisons performed implies that some of our significant results may have arisen due to chance. For all data displayed, respondents were permitted more than 1 treatment option; therefore, percentages may exceed 100. Statistical significance was set at p < 0.05.
Results
Of the 599 surveys distributed, 343 were completed and returned; 76 surveys were excluded from analysis because the respondents reported that they were not primarily in general surgery practice, leaving 267 surveys for analysis. Seventy-four surgeons were involved in academic practice and 150 were community based (43 surgeons did not comment on the nature of their practice). The mean duration of practice was 15.4 years.
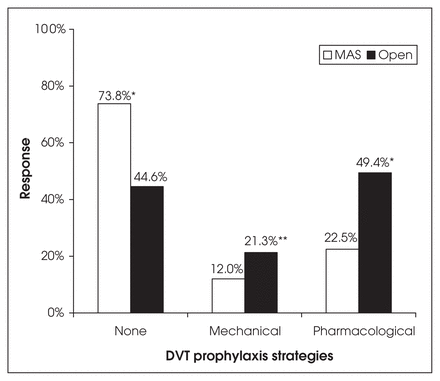
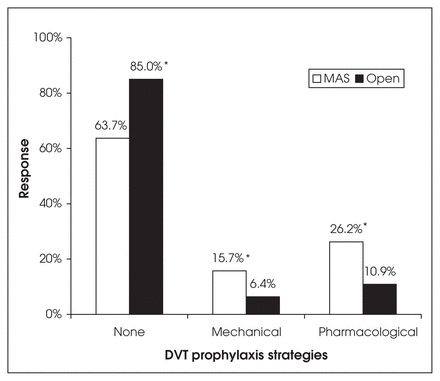
For laparoscopic cholecystectomy, although most surgeons do not provide DVT prophylaxis (73.8%, 95% confidence interval [CI] 68.2%– 78.6%), almost one-quarter provide pharmacological prophylaxis (22.5%, 95% CI 17.9%–27.8%) (Fig. 1). Others will provide either mechanical prophylaxis alone (12.0%, 95% CI 8.6%–16.4%) or a combination of mechanical and pharmacological prophylaxis (4.9%, 95% CI 2.9%–8.2%) (data not shown). There was a significant difference in the provision of DVT prophylaxis for laparoscopic cholecystectomy versus open cholecystectomy (laparoscopic cholecystectomy, no prophylaxis: 73.8%, 95% CI 68.2%–78.6%; open cholecystectomy, no prophylaxis: 44.6%, 95% CI 38.7%–50.5%; p < 0.001). The use of mechanical or pharmacological prophylaxis was significantly more likely following open cholecystectomy (p = 0.003, p < 0.001, respectively). Although most surgeons do not provide DVT prophylaxis for inguinal hernia repair, surgeons are significantly more aggressive in laparoscopic inguinal hernia repair (LIHR) versus an open approach (LIHR, no prophylaxis: 63.7%, 95% CI 57.7%– 69.2%; open repair, no prophylaxis: 85.0%, 95% CI 80.2%–88.7%, respectively; p < 0.001) (Fig. 2). The use of mechanical or pharmacological prophylaxis was significantly more likely in LIHR (p < 0.001 in each case.
Deep vein thrombosis (DVT) prophylaxis strategies for cholecystectomy (*p < 0.001, **p = 0.003). MAS = minimal-access surgery.
Deep vein thrombosis (DVT) prophylaxis strategies for inguinal hernia repair (*p < 0.001). MAS = minimal-access surgery.
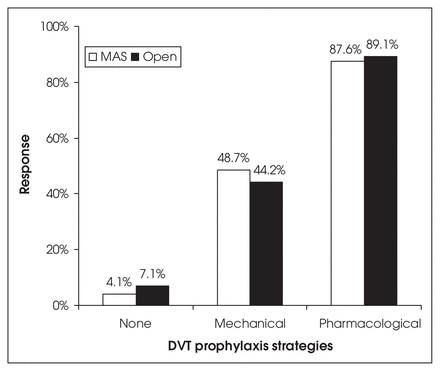
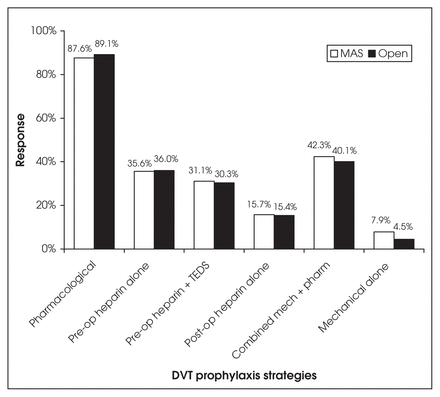
In colorectal surgery, most surgeons offer some form of DVT prophylaxis (Fig. 3). There was no significant difference between MAS or open surgery for any treatment option (MAS, no prophylaxis: 4.1%, 95% CI 2.3%–7.2%; open surgery, no prophylaxis: 7.1%, 95% CI 4.6%–10.8%, p = 0.13; MAS, mechanical prophylaxis: 48.7%, 95% CI 42.7%–54.6%; open surgery, mechanical prophylaxis: 44.2%, 95% CI 38.3%–50.1%, p = 0.30; MAS, pharmacological prophylaxis: 87.6%, 95% CI 83.1%– 91.0%; open surgery, pharmacological prophylaxis: 89.1%, 95% CI 84.8%–92.3%, p = 0.60). When the specific approach to DVT prophylaxis is analyzed further, there is variation between surgeons according to the use of pharmacological and mechanical prophylaxis and its duration (Fig. 4). Fewer than one-third of all respondents provide preoperative heparin in combination with mechanical prophylaxis for DVT in colorectal surgery (MAS: 31.1%, 95% CI 25.8%–36.8%; open: 30.3%, 95% CI 25.1%–36.1%). DVT prophylaxis for splenectomy was provided with about equal frequency for open and laparoscopic procedures (MAS, no prophylaxis: 13.6%, 95% CI 10.0%–18.2%; open surgery, no prophylaxis: 8.7%, 95% CI 5.8%–12.6%, p = 0.07; MAS, mechanical prophylaxis: 41.9%, 95% CI 36.1%–47.9%; open surgery, mechanical prophylaxis: 47.2%, 95% CI 41.6%–53.5%, p = 0.19; MAS, pharmacological prophylaxis: 37.7%, 95% CI 32.1%–43.7%; open surgery, pharmacological prophylaxis: 45.3%, 95% CI 38.7%–51.6%, p = 0.07) (Fig. 5). We found that 40.7% of surgeons would consult a hematologist specifically for DVT prophylaxis in cases of splenectomy, and in 54% of these cases they would do no more than that which the hematologist recommended.
Deep vein thrombosis (DVT) prophylaxis strategies for colon resection. MAS = minimal-access surgery.
Specific approaches to deep vein thrombosis (DVT) prophylaxis for colon resection. MAS = minimal-access surgery; TEDS = thromboembolism stockings.
Deep vein thrombosis (DVT) prophylaxis strategies for splenectomy. MAS = minimal-access surgery.
When the approach to DVT prophylaxis is stratified by practice setting (academic v. nonacademic) or years in practice, there were no differences in treatment options A (no medical prophylaxis), B (graded compression stockings), C (intermittent pneumatic compression devices) or E (heparin started postoperatively). However, surgeons working in academic centres are significantly more aggressive with preoperative low-dose unfractionated heparin (LDUH) in all scenarios (Table 1). Surgeons in practice for 15 years or less are significantly more aggressive with preoperative LDUH for laparoscopic cholecystectomy and open cholecystectomy, LIHR, laparoscopic colorectal surgery (LCS) and open colectomy. Analysis of the use of postoperative low-molecular-weight heparin (LMWH) revealed that community surgeons are significantly more likely to use LMWH for both LCS (12.0% v. 2.7%, p = 0.024) and open colorectal surgery (12.7% v. 1.3%, p = 0.005) than academic surgeons.
Use of preoperative heparin stratified by years in practice and practice location of surgeons
Discussion
The use of a minimal-access approach to gastrointestinal disease is becoming more common. Clinical trials continue to demonstrate the safety and efficacy of an MAS approach to gastrointestinal diseases, thereby expanding the application of MAS to more complex benign and neoplastic gastrointestinal pathology (e.g., colorectal cancer). Research characterizing the thromboembolic risk following MAS has not kept pace with these advances.1 Currently, there are no evidence-based guidelines for DVT prophylaxis in MAS. In an effort to manage patients appropriately, surgeons are required to adopt the recommendations for open surgery and apply them to MAS procedures.
Substantial effort has gone into establishing the feasibility of minor or “short-stay” laparoscopic surgery, which is now considered routine (e.g., cholecystectomy). Patients who undergo minor or short-stay MAS are discharged from hospital on the day of surgery and usually return to normal activities sooner than following open surgery.11 Therefore, the risk of DVT is generally presumed to be low. An early report has suggested that there is potential for high rates of symptomatic DVT following laparoscopic cholecystectomy.8 In addition, there is some evidence from physiological studies that a pneumoperitoneum may predispose a patient to DVT.2,7 However, the true incidence of DVT following minor MAS is still unknown, because most studies have not searched specifically for pre-existing DVT or postoperative subclinical DVT. In this study, we have found that a moderate percentage of surgeons provide DVT prophylaxis for minor MAS procedures (Fig. 1, Fig. 2). We made the unexpected observation that a significantly greater proportion of surgeons provide DVT prophylaxis in LIHR versus open hernia repair. Clearly, surgeons still consider an aggressive approach to DVT reasonable in LIHR. This may be explained by surgeons’ practice of completing open hernia repair under local anesthesia. Further study is required to understand the appropriate risk stratification for DVT in patients undergoing MAS and to understand the risks and benefits of DVT prophylaxis specifically related to minor MAS surgery.
A Cochrane systematic review has provided clear guidelines for the best approach to DVT prophylaxis in open colorectal surgery, characterizing the associated risk reduction and complications of treatment.12 Despite level 1 evidence for this practice, only one-third of respondents in this study provide combined perioperative pharmacological and mechanical DVT prophylaxis for open colorectal surgery (Fig. 4). Wasey and colleagues13 have identified similar findings in a recent Canadian study. This lack of compliance may be an issue of continuing medical education. We found in this study that younger surgeons and surgeons working in an academic environment were significantly more aggressive with pharmacological prophylaxis (Table 1).
In this study, we examined the current approaches to DVT prophylaxis by surgeons in practice. We have shown substantial and important variability in current practice not only for MAS but in open surgery. Surgeons are giving prophylaxis for minor, short-stay MAS procedures in the absence of reasonable evidence, and many surgeons do not give DVT prophylaxis for major open procedures where benefit has been proven. Considerable benefit will be derived from clinical trials that provide further data to establish appropriate DVT prophylaxis guidelines for MAS. Once such data and guidelines are established, specific initiatives must be developed to educate surgeons in practice so that these guidelines may be followed appropriately.
Acknowledgements
We wish to thank the Surgical Outcomes Research Committee (SOURCE) for providing assistance with data entry and the Department of Surgery, McMaster University, Hamilton, Ont., for financial support. Dr. Crowther holds a Research Scholarship from the Canadian Institutes of Health Research.
Footnotes
Competing interests: None declared.
- Accepted March 29, 2005.