Abstract
Objective: Recently generated randomized screening trial data have provided good evidence in favour of routine screening for abdominal aortic aneurysm (AAA) to reduce AAA-related deaths in men aged 65 years and older. We developed an economic model that assessed the incremental cost–utility of AAA screening to help decision makers judge the relevance of a national screening program in Canada.
Methods: We constructed a 14 health state Markov model comparing 2 cohorts of 65-year-old men, where the first cohort was invited to attend screening for AAA using ultrasonography (US) and the second cohort followed the current practice of opportunistic detection. Lifetime outcomes included the life-years gained, AAA rupture avoided, AAA-related mortality, quality-adjusted life years (QALYs) and costs. Transition probabilities were derived from a systematic review of the literature, and a probabilistic sensitivity analysis was carried out to examine the effect of joint uncertainty in the variables of our analysis. The perspective adopted was that of the health care provider.
Results: Invitations to attend screening produced an undiscounted gain in life expectancy of 0.049 years and a gain in discounted QALY of 0.019 for an estimated incremental lifetime cost of CAN$118. The estimated incremental cost–utility ratio was CAN$6194 per QALY gained (95% confidence interval [CI] 1892–10 837). The numbers needed to invite to attend screening, and the numbers needed to screen to prevent 1 AAA-related death were 187 (95% CI 130–292) and 137 (95% CI 85–213), respectively. The acceptability curve showed a greater than 95% probability of the program’s being cost-effective, and the model was robust to changes in the values of key parameters within plausible ranges.
Conclusion: Our results support the economic viability of a national screening program for men reaching 65 years of age in Canada. More clinical studies are needed to define the role of screening in subgroups at high risk, especially in the female population.
Abdominal aortic aneurysm (AAA) is the most common type of true aneurysm, affecting 4%–8% of men and 1.5% of women over age 60 years.1 Because of its high propensity for rupture, AAA remains a serious health problem, especially in Western nations, where it accounts for about 2%–4% of all deaths in the male population.2 Despite advances in general surgical care, the overall mortality rate from a ruptured AAA can be as high as 80%–90%.3 Conversely, mortality rates are now less than 5% for elective surgical repair and appear to effect complete cure.4
Currently, only opportunistically detected cases are offered elective surgical treatment despite the availability of ultrasonography (US), a highly accurate, inexpensive and noninvasive screening tool.5,6 Nevertheless, screening remains controversial because of the general uncertainty about the cost-effectiveness of population-based programs and the lack of agreement about which patients should be screened. In 1991, the Canadian Task Force on Periodic Health Examination reviewed available evidence and concluded:
“there is poor evidence to support the inclusion or exclusion of screening by physical examination or US for abdominal aneurysm in the periodic health examination of asymptomatic individuals” (C recommendation based on grade II-2 and III-3 evidence).7
In the last decade, however, 2 prospective nonrandomized trials8–10 and 4 randomized screening trials11–15 have provided strong evidence in favour of routine AAA screening in men aged 65 years and older to reduce AAA rupture and AAA-related death.
Before any formal policy recommendations can be made, however, it is critical that results of international trials be interpreted in a Canadian context in terms of both cost and effectiveness. Randomized screening trials have provided an estimate of the efficacy of screening for AAAs (with efficacy meaning the performance of a program under highly controlled conditions). Whether similar results would occur in routine clinical practice depends on the context in which the program is implemented, the population involved, the level of compliance and the characteristics and performance of the health care system. In addition, published results of AAA screening trials are short-term with limited follow-up. For cost-effectiveness analysis to be valid, however, a much longer time horizon is required to include all the relevant lifetime costs and benefits resulting from the program under study.16 Moreover, estimates of resource quantities and costs from trials conducted outside Canada are unlikely to be directly generalizable to the Canadian system because of major differences in the way health care is delivered internationally.17
In that perspective, mathematical models have been developed that combine the best available evidence from several sources in a model that mimics real-life situations as closely as possible.18 Obviously, a long-term pragmatic trial would be ideal, but this is impractical because of time and fiscal constraints. Recommendations often have to be made despite this lack of perfect data. The following modelling exercise is therefore intended to aid decision makers in assessing the relevance of a screening program for AAAs in men reaching age 65 years in Canada.
Methods
Model
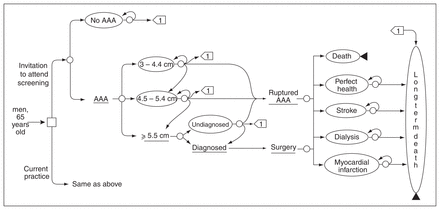
Decision analysis software (TreeAge Pro; TreeAge, Williamstown, Mass.) was used to construct a 14 health state Markov model comparing invitation to AAA screening with current practice for a hypothetical cohort of 65-year-old male patients. All subjects offered screening were assumed medically and anatomically suitable for AAA repair. The model portrayed screening at a point where the screening service had reached equilibrium, that is, when the annual intake of new subjects was constant after an initial build-up. Health states were mutually exclusive and collectively exhaustive; at a given time, each person in the hypothetical population was in one of the 14 possible health states but could not be in more than 1 state simultaneously. The health state of an individual could change between years according to predefined transition probabilities. The cycle length was 1 year, and each subject was followed until death. Outcome variables were the life-years gained, AAA rupture avoided, AAA-related death avoided, quality-adjusted life years (QALY) gained and costs. AAA-related death included death from rupture and death from elective or emergency surgical repair. A simplified version of the model is depicted in Figure 1.
Simplified Markov model. AAA = abdominal aortic aneurysm.
The clinical relevance of AAAs that develop after age 65 years is known to be negligible because they are very unlikely to reach critical diameter. 19–23 We assumed that freedom from AAAs at age 65 persists lifelong, and no rescreening policy was therefore added. In the model, US was considered 100% accurate, a simplifying assumption that parallels current data.5,6
The only variable assigned a distinct value for each of the 2 model cohorts was the relative proportion of diagnosed and undiagnosed AAAs. In the screening group, this proportion reflected subjects’ responsiveness to a request for screening. In the screened and nonscreened cohorts, undiagnosed AAAs could be detected opportunistically in each cycle. Diagnosed and undiagnosed AAAs were classified according to their largest diameter and assigned an annual risk of rupture and transition rate from one category to a higher category.
We assumed that screen-detected and nonscreen-detected aneurysms behaved in the same manner, although the latter were diagnosed later and followed a different size distribution at the time of diagnosis.
Subjects that did not require surgical treatment (aneurysm diameter < 5.5 cm) underwent annual US surveillance (if the aneurysm diameter was 3.0–4.4 cm) or semiannual surveillance (if the aneurysm diameter was 4.5–5.4 cm) and were assumed to be compliant. Because many of our variable estimates were data derived from intention-to-treat analyses of randomized screening trials, a certain level of noncompliance was implicit in the model; however, dropouts from US surveillance were tested in the sensitivity analysis.
Subjects with an aneurysm ≥ 5.5 cm in diameter were offered surgical repair. We assumed no patient older than age 85 years would be offered surgery. The threshold diameter for surgery was selected on the basis of level I evidence.24–28 Open surgical repair of AAAs was selected as the standard treatment for nonruptured and ruptured AAAs, despite the increased worldwide use of endovascular aneurysm repair (EVAR) in the last decade. Results of randomized clinical trials of EVAR are only just beginning to emerge and have shown promising short- and mid-term results.29–32 However, there is as yet no evidence of the long-term effectiveness of EVAR, and considerable uncertainty remains regarding long-term costs of surveillance and secondary interventions.33,34 Moreover, current data show a relatively conservative dissemination of EVAR in Canada, compared with other countries.35 We therefore chose to exclude EVAR from our base case analysis but tested the impact of the estimated incremental lifetime treatment cost of EVAR31,34,36,37 (including costs associated with follow-up and readmission for procedure-related complications) in 1-way sensitivity analysis.
Major surgical complications incorporated into the model include stroke, dialysis-dependent renal failure, myocardial infarction and death.
Transition probabilities
Transition probabilities were derived from a systematic review of the literature with a top-down approach to data identification. Whenever possible, we selected only studies of the highest quality, such as randomized screening trials in men and prospective naturalistic trials. We computed weighted average probabilities and distributions, using sample size as the weighting factor (Table 1). Except for time-dependent probabilities such as age-specific annual mortality, extrapolation of short-term study results to long-term outcomes assumed an exponential function.
Annual transition probabilities
A base case attendance rate of 73.3% was derived from 19 population screening studies in men.9,11–15,38–55 The prevalence of AAAs > 3.0 cm in diameter was estimated at 4.2%, derived from age-specific combined data from the 4 randomized screening trials in men.56
There are no reliable data in the literature that can be used to estimate the potential for incidental detection of undiagnosed AAAs. From the ratio of elective surgeries in the nonscreened and screened cohorts in the 4 randomized screening trials, we assumed that 7% of undiagnosed AAAs would be discovered annually, regardless of their diameter.
Data related to the risk of rupture are scarce and difficult to interpret because patients with large aneurysms are usually offered surgery. Prospective follow-up data on patients with large AAAs are available only for those who either refused surgery or were considered unfit for surgery.57–65 The risk of rupture for screen-detected AAAs ≥ 5.5 cm is therefore unknown; however, valuable information can be extracted from the 3 largest randomized screening trials13–15 by assessing the number of AAA ruptures in the unscreened cohort relative to the assumed prevalence of undiagnosed large AAAs. This led us to assume a 16% rupture rate per year for the base case but with a wide range of plausible values.
Outcomes of elective and emergency surgical repair were derived from Canadian multicentre prospective data and are detailed in Table 1. The age-specific annual mortality is based on the 2000 Canadian life table for males (www.bdlc.umontreal.ca). The overall survival of patients with AAAs is known to be reduced when compared with an age- and sex-matched population because of the greater associated comorbidities in patients with an aneurysm.66 Their survival curve was therefore adjusted to account for an excess mortality of 2% yearly.67,68 The reduction in life expectancy due to dialysis-dependant renal failure was adjusted with age-and sex-specific data from the 2004 US Renal Data System (www.usrds.org). An annual excess mortality of 2.25% was assigned for patients who survived myocardial infarction after AAA repair.69,70 The annual mortality rate was assumed to be 2.3 times greater for patients who survived a stroke after AAA repair.71,72
Utilities
The quality adjustment factors of each state depicted in Figure 1 are derived from health state preference (utility) data. Current evidence suggests that screening has no significant adverse effect on quality of life.13,73,74 Utility estimates were therefore calculated from published figures derived from the Health Utility Index values for men aged 65 and older from the 1994–1995 Canadian National Population Health Survey.75 QALY weights are detailed in Table 2. Subjects undergoing elective or emergency surgical repair were only assigned a “disuse” because of current evidence suggesting the lack of significant long-term effect on quality of life.78,79 Adjustments were also made for long-term complications.
Health state preference (utility) estimates
Costs
Direct costs of health care resources were estimated from data on several Canadian published sources (Table 3). These included the cost of abdominal US, prehospitalization workup, inpatient care for surgical treatment of ruptured and nonruptured AAAs, professional fees and lifetime costs of complications. A cost for inviting people to screening was not included because we assumed that they would be identified during a regular health care visit. Given the age of the cohort, costs from lost productivity were not included, and a health care provider perspective was therefore adopted.
Estimated direct costs of health care*
The cost of abdominal US was derived from our hospital costs accounting system and was estimated at CAN$69.75 (physician fees included). A cost for a vascular surgery consultation was added for those who tested positive, and a cost was added for a physician visit after each US surveillance examination. Inpatient hospital care costs exclude overhead costs because these are unlikely to vary appreciably in response to small changes in the number of surgical procedures. The future health care costs of unrelated diseases were not included in the model.
Costs are displayed in 2005 Canadian dollars after adjustment by the health care component of the consumer price index. A discount rate of 5% per year was applied to costs and QALY in accordance with Canadian Coordinating Office for Health Technology Assessment guidelines.81
Sensitivity analysis
Probability distributions were used to characterize the uncertainty in the mean value of each parameter. We selected values at random from each distribution, using a second-order Monte Carlo simulation with 1000 iterations for each cohort. Probabilistic sensitivity analysis was selected as a means of examining the effect of joint uncertainty in the variables of our analysis.82 Incremental cost-effectiveness ratios (ICERs) were calculated and the results presented as a scatter plot of ICER and the cost-effectiveness acceptability curve. Key variables and variables with the greatest level of uncertainty were also subjected to a 1-way sensitivity analysis and a threshold analysis.
Results
An invitation to attend screening produced an undiscounted gain in life expectancy of 0.0499 years (18 d). The survival advantage resulted from a reduced number of ruptured AAAs (risk ratio 0.43) and AAA-related deaths (risk ratio 0.50). The estimated risk differences were 0.83% (95% CI 0.52%–1.18%) for AAA rupture and 0.54% (95% CI 0.34%–0.77%) for AAA-related deaths. The numbers needed to invite to attend screening and the number needed to screen to prevent 1 AAA rupture were 121 (95% CI 85–193) and 89 (95% CI 63–142), respectively. Similarly, the numbers needed to invite to attend screening and the number needed to screen to prevent 1 AAA-related death were 187 (95% CI 130–292) and 137 (95% CI 85–213), respectively.
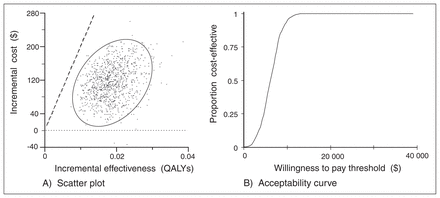
In the base case analysis, invitation to screening produced a gain in discounted QALYs of 0.019 for an estimated incremental lifetime cost of CAN$118, giving an ICER of CAN$6194 per QALY (95% CI $1892–$10 837). The result of the probabilistic sensitivity analysis is shown on the cost-effectiveness plane and acceptability curve (Fig. 2). Assuming a willingness to pay threshold of CAN$20 000 per QALY, the probability of a screening program being cost-effective is greater than 95%.
(A) Scatter plot of the incremental cost-effectiveness ratio with 95% confidence interval ellipse derived from probabilistic sensitivity analysis. Dashed line represents a willingness-to-pay threshold of CAN$20 000 per QALY. (B) Cost-effectiveness acceptability curve. QALY = quality-adjusted life year.
The results of 1-way sensitivity analysis are shown in Table 4. Our model is robust to variation in the value of key parameters within plausible ranges, although the prevalence of AAA, the risk of rupture for a large AAA and the age at initial screening were those that had the greatest impact on the cost–utility ratio.
Results of 1-way sensitivity analyses
Discussion
It is generally accepted that, for a disease to be considered a candidate for screening, it should pose an important health problem, the natural history should be well understood and there should be a recognizable early stage at which treatment is more beneficial than at a later time.83 There should also be a suitable diagnostic test that is acceptable to the population and adequate existing facilities for the diagnosis and treatment of the condition. The costs of a screening program should also be balanced against potential benefits.
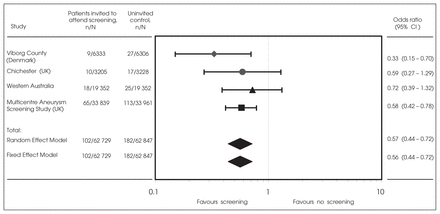
There is now good evidence that screening reduces the incidence of ruptured AAAs and AAA-related mortality. Studies that provided level I evidence were the Chichester trial11,12 and the Multicentre Aneurysm Screening Study (MASS)13 trial in the United Kingdom, the Viborg County study in Denmark14 and the Western Australia study.15 All trials identified potential participants who were 65 years old or older via population registries or regional health directories; collectively, the studies included more than 125 000 participants. Pooled results show an odds ratio of 0.56 (95%CI 0.44–0.72) for AAA-related deaths (Fig. 3). All-cause mortality was not significantly reduced, however, because AAA-related mortality accounted for only about 3% of all deaths in the control cohorts.
Meta-analysis results of randomized screening trials for abdominal aortic aneurysm in men. CI = confidence interval.
Concerns about the economic viability of a screening program promoted construction of several economic models.84–94 Although these studies differed considerably in design and evaluated parameters, all but 1 study86 concluded that screening of specific cohorts was economically appealing. Only 2 models, however, were developed from recently generated randomized screening trial data.92,94
Three economic analyses alongside clinical trials have also been conducted.10,95,96 The largest of these, the MASS trial,96 found screening to be only marginally cost-effective after 4 years of follow-up. On the basis of conservative assumptions, however, the authors projected a 10-year incremental cost-effectiveness ratio of about £8000 per life-year gained. This improvement was due largely to the accumulation of life-years gained by preventing the death of individuals during the first 4 years of the trial. In a recent systematic review of published cost-effectiveness analyses,97 the authors concluded that existing evidence pointed to a cost-effectiveness ratio between US$14 000 and US$20 000 per QALY for population-based AAA screening in men, compared with non-screening.
Our own results also suggest that screening for AAA in 65-year-old men would be efficacious and cost-effective. The gain in undiscounted life expectancy appears modest (18 d), but in practice, this survival advantage would apply to only 4.2% of the cohort. The expected gain in patients with AAA is therefore 24 times 18 days, that is, 432 days. In the first 2 cycles (2 y), the reduction in AAA-related deaths is offset by increased deaths from elective surgical repair in the screened cohort, a finding consistent with the observations from the early years of randomized screening trials. Thereafter, mortality in the screened cohort separates from that of the control and reaches a plateau after about 15 years of follow-up. Although no clear consensus exists about our threshold value of willingness to pay for an additional QALY,98 our estimated ICER (CAN$6194 per QALY) is below the value that is generally considered to be cost-effective. Model results are robust to changes in the value of key variables within the range of plausible values and the acceptability curve is steep, indicating a low level of uncertainty (variance) regarding our main estimate.
Key variables
The prevalence of screen-detected AAAs among Canadian men is unknown, and reported prevalence worldwide is highly variable owing to differences between studies in terms of how AAA is defined, the age and sex distribution of study populations and the prevalence of risk factors and preexisting morbidities. To avoid biasing our results in favour of screening, we used a very conservative estimate for the prevalence of screen-detected AAAs (4.2%),56 the lowest in all models published to date. The extent to which this applies to the Canadian male population is unknown, but 1-way sensitivity analysis yielded a threshold prevalence of 0.67%, which is very unlikely.
The use of existing epidemiologic data to determine the risk of rupture for large aneurysms is highly problematic. For models that involve screening of the general population, published data are often unusable because of ill-defined or otherwise inappropriate denominator populations. The high level of uncertainty regarding the base case value is reflected in our choice of a wide triangular distribution. Not surprisingly, this variable accounts for 30% of the variance of the estimated ICER. In 1-way sensitivity analysis, the threshold value was about 6.7%, which is, again, very unlikely.
Age at initial screening has a significant impact on the cost–utility ratio, and its relevance is a result of the inverse relation between life expectancy after elective surgery and the cost per QALY gained. For example, studies including a high proportion of men older than 75 years at the time of randomization failed to show statistically significant reduction in AAA-related deaths.15 These results are corroborated by our own findings, which show that screening after the age of 80 is not cost-effective and that the upper boundary of the 95% CI of the cost–utility ratio is above threshold after the age of 76. There is now a growing consensus that screening at age 65 years is probably the most appropriate strategy99– 101 because a single ultrasound at that age can rule out significant disease for life in 95% of men.19–23 Reducing the screening age would pick up the few cases that would have ruptured before age 65 years; however, the need to repeat scanning at a later time would double the cost of screening overall.
Our model was robust in the face of changes to the cost of US; however, because that cost item applies to 100% of individuals attending screening, it accounted for 51.4% of the estimated incremental lifetime cost. Any strategy aimed at reducing the cost of US examination would therefore have a significant effect on the projected budgetary impact of a screening program. Options such as the use of portable US machines should be examined because the cost of a portable unit is 75%–80% less than that of a conventional duplex unit.102 Moreover, portable units were used in several screening studies, including the MASS trial, which validates their use. A quick screen (single organ) examination can be performed in less than 5 minutes89 and avoids unexpected findings in other intra-abdominal organs for which the value of screening is unknown.
Endovascular aneurysm repair (EVAR) is now considered a valid alternative to open repair. This new technology provides a clear reduction in 30-day mortality but no survival advantage over 1 year of follow-up.31,32 More important, this short-term benefit comes at considerable additional cost, especially if follow-up costs are considered. The current status of use of EVAR in Canada (about 10% for nonruptured AAA)35 is unlikely to affect the cost-effectiveness of a screening program. A trend toward increased use is to be expected and was tested in 1-way sensitivity analysis (Table 4). Those results should be interpreted cautiously, however, especially because of the lack of long-term cost data and the unproven long-term longevity of EVAR.
The costs of long-term complications accounted for only 4.5% of the incremental lifetime cost, demonstrating very little impact on the cost–utility ratio. Conversely, inpatient care cost (for elective and emergency surgical repair) accounted for 40.2% of the incremental lifetime cost. Because screening changes the need for elective and emergency surgical repair in the opposite direction, varying the estimated cost of inpatient care had very little impact on the estimated ICER. The response rate to an invitation to attend screening also had very little impact on the estimated cost–utility ratio owing to the positive correlation between response rate and the numerator (incremental cost) and denominator (incremental QALY) of the ICER. The importance of response rate should not be neglected, however, because the overall cost and benefit will obviously depend on whether individuals attend screening or not.
The performance of a screening program in routine clinical practice depends largely on the level of compliance. In the randomized screening trials, 81%–88% of patients complied with surveillance US. Despite a certain level of noncompliance implicit in our model, we adjusted for a possible protocol-driven effect by testing additional dropouts from US surveillance in a 1-way sensitivity analysis. Unlikely values, such as 50% annually, did not result in an above-threshold estimated ICER.
Controversial issues
Selective screening
Cost-effective screening relies on the selection of a subpopulation at high risk for the disease in question. Documented risk factors for AAAs, in addition to age and sex, are smoking, cardiovascular disease, hypertension and familial clustering.103 Today, most screening programs are targeted at men older than age 65 years and are nonselective to other risk factors.
Smoking, which is associated with a 2- to 4-fold increase in AAA prevalence, 104 has been proposed as a possible criterion for selective screening. In a 2005 report, the US Preventive Services Task Force recommended 1-time screening only for men aged 65–75 years who have ever smoked (grade B recommendation)100,101 but made no recommendation for or against screening the same age class of men who have never smoked (grade C recommendation). From a theoretical model, it was estimated that 89% of AAA-related deaths could be prevented by screening 69% of men; however, targeting high-risk patients for AAAs restricts screening to a cohort with an increased risk of long-term mortality. AAA prevalence and life expectancy are variables affecting the cost-effectiveness in the opposite direction. Not surprisingly, models that took into account the increased future mortality of high-risk groups found selective screening to be nonbeneficial.90,105
Screening in women
The Chichester study was the only randomized screening trial that included both men and women. A total of 9342 women aged 65–80 years were randomly assigned to an invitation to screening group or a control group.106 The screened cohort had an AAA prevalence of 1.3%, and no benefit was observed after 10 years of follow-up. AAAs occurred on average a decade later in women than in men, and most ruptured after 80 years of age. The low prevalence of AAAs in the female population has been repeatedly documented and has resulted in the exclusion of women from subsequent large screening trials.
Because women share the same risk factors as men, targeted screening based on cardiovascular risk factors has been recommended,99 although no clinical trial supports that recommendation. More studies using different screening strategies are therefore needed before any conclusions can be reached.
Limitations
A model can only be as good as the data it is based on. We therefore tried to structure our model to match the highest-quality data available. As discussed previously, the main limitations were the lack of good-quality data on the natural history of undetected large AAAs and the fact that we had to extrapolate long-term outcomes from studies with limited follow-ups. Another limitation arose from the lack of Canadian data on a screening program’s generation and use of resources and their related costs. Although precise cost estimates are available from other countries, the trade-off between precision and relevance prompted us to adapt our model to the Canadian context by using what we considered to be the most locally relevant cost data. Head-to-head comparison of our results with that of published economic evaluations needs to be interpreted in light of known differences in approaches that have been adopted for the funding and delivery of health care services. In the United States for example, treatment of a given condition is known to be more resource-intensive when compared with Canada or the United Kingdom, and prices of medical supplies, labour and administrative infrastructure are also much higher.107,108 Using currency conversion to compare results from one setting to another is therefore inappropriate.
Conclusion
Despite the limitations of the present model, it appears that screening for AAAs in men reaching 65 years of age is efficacious and cost-effective. Our model revealed that the incremental cost per QALY is lower than the value generally considered to be cost-effective and compares favourably with the estimates cited in other screening programs. AAA screening has many advantages, namely, its simplicity and reliance on a safe and highly accurate screening tool. For such a program to meet its goals, however, sufficient resources need to be made available for both the diagnosis and treatment of screen-detected AAAs. More clinical studies are also needed to define the role of screening in subgroups at high risk, especially in the female population.
Footnotes
Competing interests: None declared.
Contributors: Drs. Montreuil and Brophy designed the study. Dr. Montreuil acquired the data, which both authors analyzed. Dr. Montreuil wrote the article, and both authors revised it. Both authors gave final approval for the article to be published.
- Accepted May 17, 2006.