Abstract
Background: Denervation substantially impairs healing of the medial collateral ligament (MCL). Because normal ligaments are sparsely innervated, we hypothesized that neuropeptide-containing neurons would sprout or proliferate after ligament transection, followed by later regression with healing, in a manner analogous to blood vessels.
Methods: We transected the right MCL in 9 mature female New Zealand white rabbits and killed 3 rabbits at 2, 6 or 14 weeks. Alternate sets of 12-mm serial sections of healing MCL scars were examined by fluorescent immunohistochemistry for substance P (SP), calcitonin gene-related peptide (CGRP), neuropeptide Y (NPY) and pan-neuronal marker PGP9.5.
Results: Normal MCLs had few peptidergic fibres located in the epiligament in a perivascular pattern. At 2 weeks, PGP9.5-, SP- and CGRP-positive fibres had increased in the epiligament adjacent to the injury. By 6 weeks, there were increases in CGRP- and PGP9.5-positive fibres in epiligament and scar, with similar but less marked increases in SP-positive fibres. At 14 weeks, there was notable regression of immunostained peptidergic nerve fibres in the scar.
Conclusion: This experiment shows evidence for a remarkable plasticity of ligament innervation after injury, supporting the idea that neuronal factors play a fundamental role in wound healing.
Wound healing is a continuous process that can be said to occur in 3 somewhat arbitrary and overlapping stages: initial inflammation followed by proliferation of cells and matrix and then a prolonged remodelling phase.1 Within hours of an initial injury, hemorrhage and clot formation occur, accompanied by an inflammatory response that subsequently leads to angiogenesis, cellular proliferation and scar formation. Then, in the following weeks to months, tissue remodelling takes place. It is reasonable to suggest that any factors promoting inflammation or cellular proliferation are likely to have a significant effect on the outcome of wound healing.
Numerous studies have indicated a potential role for neuropeptides (small peptide-signalling molecules derived from sensory and sympathetic neurons) in wound healing.2 Substance P (SP) and calcitonin gene-related peptide (CGRP) are potent vasodilators, making these substances particularly important during the initial inflammatory stage of healing.2 Neuropeptide Y (NPY) is produced by sympathetic neurons, has vasoactive properties and, in concert with CGRP, modulates endothelial and fibroblast cell proliferation associated with angiogenesis. 3,4 Mice lacking the NPY Y2 receptor exhibit impaired skin wound healing.4
Other investigations have demonstrated the benefits of an intact peripheral nervous system on wound healing. Most of these studies have focused on well-innervated structures such as skin. In a rat study, denervation via chemical sympathectomy resulted in decreased healing of skin incisions, compared with control animals.5 Another study in rats revealed impaired fracture healing after peripheral nerve transection. 6 Intact innervation also plays a role in fetal skin wound repair. Stelnicki and colleagues7 found that innervation was necessary for scarless wound healing in fetal lambs, and in some cases, healing did not occur at all in denervated tissues.
We have previously reported that denervation significantly impairs healing of the rabbit medial collateral ligament (MCL).8 Six weeks after injury, denervated ligament scars had one-third the blood flow, exhibited two-thirds the angiogenesis response and were 33% weaker when compared with innervated ligament scars. Normal ligament is rather sparsely innervated, with most fibres found in a perivascular distribution in the enveloping epiligament and only rare unmyelinated fibres found within the deep substance of the ligament proper.9 The unexpectedly profound effect of denervation on the healing of such a poorly innervated tissue suggested that significant changes in the pattern and density of innervation could be occurring during normal ligament healing.
In the brain and spinal cord, neuronal plasticity is a phenomenon well-known as a substrate for such diverse phenomena as motor learning, memory consolidation and the recovery of function after brain or spinal cord injury10,11; however, less attention has been paid to the structural plasticity of sensory and sympathetic terminal arborizations in the peripheral nervous system. Injury induces dramatic changes in ligament vascularity and blood flow,12,13 but no previous study has investigated the response of the neural elements in ligament at multiple time points after injury.
We hypothesized that after ligament injury the response of neuropeptide-producing (peptidergic) neurons would parallel or presage the vascular response, with initial sprouting and proliferation followed by remodelling and/or regression at later time points. To investigate this intriguing possibility, we elected to examine injured ligaments with immunohistochemistry to identify changes in the pattern and number of peptidergic nerve endings at different times after injury.
Methods
Surgical procedures
We used 9 skeletally mature female New Zealand white rabbits in this experiment. All animals were given an intravenous injection of 25 mg/mL, 0.16 mL acepromazine maleate (Atravet; Ayerst Veterinary Laboratories) followed by the induction of general inhalational anesthesia (2%–5% halothane and nitrous oxide; 1 L/min O2). Using a sterile technique, we completely transected the right MCL of each rabbit. Postoperatively, the animals were given a subcutaneous injection (3 mL) of the antibiotic Duplocillin LA (Intervet), and the injury site was treated with gentamicin antibiotic spray (Gentocin; Schering-Plough Animal Health). Three rabbits were killed at each of 2, 6 and 14 weeks. Each animal was handled in accordance with the guidelines of the Canadian Council on Animal Care. The University of Calgary Animal Care Committee approved all research protocols. All possible efforts were taken to minimize animal suffering and the number of animals used.
MCL transection
The MCL was approached through a medial incision followed by blunt dissection of overlying connective tissues. A 5-mm spatula was gently inserted under the entire ligament, and the ligament was sharply transected with a scalpel. The cut ends of the ligament were noted to retract spontaneously, leaving a 3-mm gap. The skin was closed with 4–0 nylon sutures.
Sample collection
Animals were killed by an intravenous overdose of sodium pentobarbital (Euthanyl; Bimeda-MTC Animal Health Inc.). The whole MCL was taken from the right leg and immersed in phosphate-buffered 4% paraformaldehyde overnight. The tissue was then cryoprotected in 2 changes of 30% sucrose phosphate buffer overnight, embedded in Tissue-Tek OCT compound (Sakura Finetek) and snap frozen in 2-methyl butane cooled in liquid nitrogen.
Histology
Blocks were stored at −80°C until they were cut into 12 μm sections with a Leica CM3050 cryostat (Leica Microsystems). For each ligament, serial sagittally oriented longitudinal sections were cut through the entire ligament and scar. The sections were alternately collected into 4 sets so that each set comprised every fourth section of the entire series. This allowed us to use 4 different antibodies for each ligament. Sections were thaw-mounted onto gelatin-coated slides and allowed to dry overnight.
Immunohistochemistry
Staining was done in batches that included specimens from the 3 different postinjury time points, all stained at the same time so that inherent variability in the preparation and staining methods would not affect interpretation of the results. Alternate sets of sections were processed for fluorescent immunohistochemistry with polyclonal antibodies specific for SP (Bachem), CGRP (Bachem), NPY (Millipore) or protein gene product 9.5 (PGP9.5) (Serotec). SP and CGRP are peptides found in unmyelinated sensory neurons. Neuropeptide Y is a peptide widely distributed in the brain but restricted to sympathetic nerve fibres in the peripheral nervous system. PGP9.5 is a peptide marker found in virtually all neurons of the peripheral nervous system. Specific antibody staining was visualized by the appropriate Cy3-conjugated fluorescent secondary antibody (Jackson Immunoresearch Laboratories).
Sections were washed (3 × 5 min) in 0.1 M phosphate buffer saline solution/ 0.1% Triton X-100 (Sigma-Aldrich) and then blocked with the appropriate normal blocking serum at room temperature for 30 minutes. Primary incubation was performed overnight in a humidity chamber at 4°C, with the recommended titres for each antibody being used: PGP9.5, 1:200 in 2% normal rabbit serum; SP, 1:200 in 2% normal goat serum; CGRP, 1:200 in 2% normal goat serum; and NPY, 1:1000 in 2% normal rabbit serum. One or 2 sections from each series were stained identically, with the exception that the primary antibody was omitted to test for nonspecific staining.
The following day the sections were rinsed (3 × 5 min) in PBS and then incubated with the specific Cy3-conjugated secondary antibodies for 1 hour at room temperature in a humidity chamber. Finally, the sections were washed (3 × 5 min) in PBS, cover slipped with Fluorosave reagent (Calbiochem) and stored in the dark at 4°C.
Semiquantitative analysis
We examined the complete set of serial alternate sections for each ligament (40–50 sections for each neuropeptide or marker), which allowed us to assess the presence throughout the tissue of nerve fibres (profiles) containing SP, CGRP, NPY and PGP9.5. We observed slides on a Leitz DMRB fluorescent scope (Leica), using filters appropriate for Cy3 (excitation 515–560 nm). It was evident that changes in the number of nerve fibre profiles were occurring in the scar region, so we limited the histologic examination to that region of the injured ligaments. The scar region was easily distinguished from native ligament tissue by the abrupt transitions from the regular bundles of collagen fibres seen in normal ligament to the amorphous-appearing matrix and increased cellularity of the scar filling the gap left after the injury (Fig. 1). We graded innervation density semiquantitatively as follows: We counted the number of axonal profiles in the scar region by systematically examining every section of the series for each marker. We defined an axonal profile as a homogeneously stained linear or branching sinuous structure with a minimal length of 5 μm. We defined a growth cone (the growing tip of a sprouting neuron) as a similar structure with an amoeboid-appearing tip. Axonal profiles and growth cones could be readily distinguished from background staining, which occurred in an evenly distributed, amorphous or finely granular pattern. We determined the total number of profiles per ligament scar region for each marker. The data are presented in Table 1.
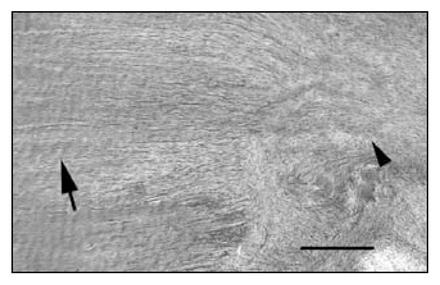
Low-power view of a longitudinal section of rabbit medial collateral ligament at 6 weeks postinjury, showing the transition zone between normal ligament matrix and scar. Note the organized parallel bundles of crimped collagen fibres in the normal matrix at left (arrow) and the disorganized non-crimped collagen fibres in the scar at right (arrowhead) (hematoxylin–eosin stain, scale bar = 100 μm).
Total numbers of specific peptidergic neuronal profiles in the scar formed at different times after MCL transection*
Results
Immunohistochemistry
Normal MCL was found to have few peptidergic fibres located almost exclusively in the epiligament on the surface of the ligament in a predominantly perivascular pattern. Rarely were fibres seen within the ligament substance proper. After ligament injury, different temporal patterns of staining distribution were seen for each peptidergic marker (summarized in Table 1). We found that the increases in number of nerve fibres were almost exclusively occurring in or near the scar tissue that formed in the gap between the cut ends of the transected ligament. By definition, scar tissue is formed de novo in the gap created by the injury. Therefore, all nerve fibres found in the scar were newly derived by sprouting and in-growth from preexisting fibres in the surrounding tissue. Bright-field examination of the ligament sections under low power permitted a visual identification of the scar region that was easily distinguished by its increased cellularity and disorganized collagen matrix (Fig. 1).
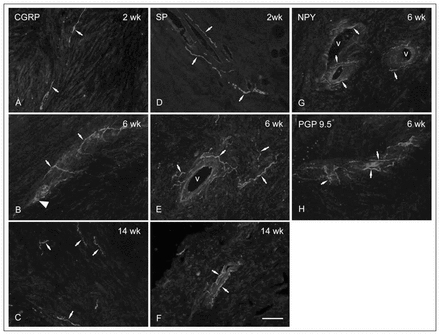
At 2 weeks, there were moderate increases in the number of profiles staining for the sensory neuropeptides CGRP and SP (Fig. 2A and 2D respectively), occurring primarily in epiligament adjacent to the developing scar. By 6 weeks postinjury, there were marked increases in the counts of CGRP- (Fig. 2B) and PGP9.5-immunoreactive neurons (Fig. 2H) in both epiligament and scar, with similar but less marked increases in SP labelling (Fig. 2E). All the neuronal markers identified nerve fibres closely associated with blood vessels in the scar and epiligament. CGRP- and PGP9.5-immunoreactive fibres were also seen as free nerve endings in the scar matrix. At 14 weeks, there was a regression of immunostained nerve fibres (Fig. 2C and 2F) to numbers slightly greater than those seen in normal controls (not shown). Of note, there were no noticeable changes in the number of NPY-positive fibres at any time point.
Photomontage of typical high-power images of immunohistochemically stained nerve fibres (arrows) at different time points after injury. (A) At 2 weeks, CGRP fibres appear very fine in the early scar. (B) At 6 weeks, CGRP-immunoreactive growth cone–like structures (large arrow head) and sprouting appear, as well as a predominantly perivascular distribution of fibres. (C) At 14 weeks postinjury, there are fewer CGRP-immunoreactive fibres in the scar, with most found again in the epiligament. (D) All SP-positive profiles were found to be perivascular at 2 weeks after injury. (E, F) Similar to the appearance at 2 weeks, SP-positive profiles were only found associated with proliferating vessels within the scar at later time points. (G) Typical image of NPY-containing fibres associated with small arterial vessels in a 6-week postinjury scar. (H) At 6 weeks postinjury, staining for the marker PGP9.5, which stains all nerve fibres, revealed a pattern that combined aspects of the other markers, with most fibres in a perivascular location, and some free in the scar matrix (scale bar = 20 microns). CGRP = calcitonin gene-related peptide; NPY = neuropeptide Y; PGP9.5 = protein gene product 9.5; SP = substance P; v = vessel lumen.
Discussion
We have found evidence of a remarkable plasticity of innervation after ligament injury. The initial sparse distribution of fibres restricted to the epiligament begins to proliferate by 2 weeks and infiltrates the scar by 6 weeks. The number and distribution of nerve terminals then regresses by 14 weeks. Nerve growth into the ligament scar appears to mimic the angiogenic response to injury, which begins in the epiligament, where the greatest number of vessels are found, and then spreads to the midsubstance of the scar.13,14 The similarity between the vascular and neuronal responses to ligament injury is consistent with recent evidence that growing neurons and vessels are subject to similar, and in some instances identical, regulatory signalling pathways (see Carmeliet15 for a review).
Some CGRP- and SP-containing profiles were noted in the scar region as early as the 2-week time point, consistent with the known role of these neuropeptides in promoting inflammation. The expression of PGP9.5, a general nerve marker, parallelled both SP and CGRP neuropeptides at the 2-week time point.
Six weeks after ligament injury, a time when cellular proliferation and new matrix production are maximal, we found that there were numerous SP- and CGRP-labelled nerve fibres in the scar. CGRP and PGP9.5 immunolabelling increased more than SP at the 6-week time point. This likely reflects the fact that CGRP-containing sensory neurons are almost 3 times more prevalent in articular tissues than SP-containing neurons.16 The peak number of labelled profiles for all the examined neuropeptides was at 6 weeks postinjury, which also coincides with the time of peak blood flow and vascular volume in rabbit MCL.13,14 NPY staining in the scars did not change significantly after injury. Because NPY is a marker for sympathetic neurons, whereas SP and CGRP are found in sensory neurons, this observation could indicate that sensory and sympathetic neurons respond differently to ligament injury in this model.
At 14 weeks, there were fewer peptidergic profiles in the scars, consistent with our hypothesis that neuronal responses would parallel the angiogenesis response. The dynamic and highly plastic nature of the neuronal response after ligament injury further supports the idea that innervation plays an important role in promoting the early healing response of these tissues. Our findings are remarkably similar to those recently reported for injured Achilles tendon in rats,17,18 supporting the speculation that neuronal proliferation and activity may be a necessary aspect of wound healing in many tissues and species.
Owing to the small number of animals studied at each time point, the morphological nature of the data, and the amount of interanimal variation, a statistical analysis was not feasible in this study. However, this preliminary study provides clear supporting evidence that an intact peripheral nervous system is dynamically responsive to the early stages of ligament injury. Nerve growth, blood flow and angiogenesis are all substantially upregulated from 2 to 6 weeks after ligament rupture, indicating that these 3 processes likely all influence the healing process in an interdependent manner.
Pain after joint injury is common, severe and disabling, but little is known about the processes and mediators that regulate the activity of the peripheral nervous system in this context. Unlike the phenomenon of angiogenesis, which has received considerable attention in recent years, the factors involved in neuronal sprouting and growth have received little attention outside the realm of direct injury to brain, spinal cord or peripheral nerve. Further research may reveal the molecules and mechanisms regulating the plasticity of neuronal morphology in injured connective tissue. This could lead to important insights and perhaps new avenues for the treatment of these very common, frequently disabling injuries.
Acknowledgements
This work was supported by an operating grant from the Arthritis Society of Canada. Dr. Salo is a Senior Scholar and Dr. Bray a Scientist of the Alberta Heritage Foundation for Medical Research.
Footnotes
Competing interests: None declared.
Contributors: Drs. Salo and Bray and Mr. Ivie designed the study. Mses. Seerattan and Leonard and Mr. Ivie acquired the data, which Dr. Salo, Ms. Beye and Mr. Ivie analyzed. Drs. Salo and Bray and Mses. Beye and Seerattan wrote the article, which Drs. Salo and Bray, Ms. Leonard and Mr. Ivie reviewed. All authors gave final approval for publication.
- Accepted January 22, 2007.