Abstract
Supine anteroposterior chest radiography is an insensitive test for posttraumatic pneumothoraces. Computed tomography often detects pneumothoraces that were not diagnosed on chest radiography (occult pneumothoraces). Whereas the incidence of occult pneumothoraces approximates 5% of all trauma registry patients, this value approaches 15% among injured patients undergoing computed tomography. Up to 76% of all pneumothoraces may be occult to supine chest radiography with real-time interpretation by trauma teams. Although the size and intrathoracic distribution (anterior) of overt and occult pneumothoraces are similar, significantly more patients with occult pneumothoraces undergo tube thoracostomy compared with those with overt pneumothoraces. This pattern extends both to patients receiving mechanical ventilation and those with penetrating trauma. As an early clinical predictor available during the resuscitation of a trauma patient, only subcutaneous emphysema is predictive of a concurrent occult pneumothorax. The majority of patients with occult pneumothoraces (85%) do not have subcutaneous emphysema, however. Thoracic ultra-sonography, as part of a bedside extended focused assessment with sonography for trauma examination, detects 92%–100% of all pneumothoraces and represents a simple extension of the clinician’s physical examination. The final remaining question is whether clinicians can safely omit tube thoracostomy in some patients with occult pneumothoraces concurrent to positive pressure ventilation. This omission would avoid subjecting patients to the 22% risk of major chest tube–related insertional, positional and infective complications.
Occult pneumothoraces are a relatively recent radiological phenomenon.1–5 They are defined as pneumothoraces detected with thoracic or abdominal computed tomography (CT) that were not diagnosed on preceding supine anteroposterior chest radiography.1–5 With the increase of both CT and thoracic ultrasonography as the initial screening modalities for the investigation of blunt thoracoabdominal trauma,1–14 these entities are becoming extremely common. Because of the importance of spine immobilization in the acutely injured patient, it is also evident that the technological limitations of supine anteroposterior chest radiography performed in the trauma bay will not be overcome in the near future. This is compounded by the fact that supine chest radiography is the least sensitive of all the plain radiographic techniques for demonstrating a pneumothorax.6,15–18 These factors make the accurate, rapid and reliable diagnosis of pneumothoraces a significant challenge.
Although occult pneumothoraces are regularly identified in the daily practice of caring for the injured patient,1,19 clinicians have had minimal objective evidence to guide their management decisions until recently. During the past 5 years, many of the inaccurate assumptions regarding occult pneumothoraces have been debunked. Studies have confirmed the incidence of occult pneumothoraces (unpublished data, 2009),19–25 and have commented on their intrathoracic distribution,23,26 risk factors,19,20 outcomes19,23 and appropriate treatment.19,23,27 Recent literature also discusses the utility of thoracic ultrasonography in identifying occult pneumothoraces,7–13 and its evolution as a crucial diagnostic tool in the assessment of patients with blunt injuries in the trauma bay. Still unclear, however, is the optimal treatment for patients with occult pneumothoraces requiring concurrent mechanical ventilation.1 It is also unknown which modality is most appropriate for subsequent monitoring for occult pneumothoraces in patients whose conditions are stable and who do not require mechanical ventilation (chest radiography, CT, dependent ultrasonography or clinical observation). This review discusses all recent literature pertaining to occult pneumothoraces, as well as the remaining controversies. We also include our diagnostic and management algorithm for patients with blunt thoracic trauma.
Definition of occult pneumothoraces
The definition of an occult pneumothorax is uncontested. It is a pneumothorax that was not suspected on the basis of clinical examination or plain radiography, but is ultimately detected with CT.1–5 It was originally defined as a pneumothorax noted on abdominal CT that was not seen on preceding supine anteroposterior chest radiography.2–5 This definition has been expanded to include occult pneumothoraces identified with thoracic CT as well.1 It is also reasonable to include patients who have undergone initial investigation with upright chest radiography (unpublished data, 2009). Although erect chest radiography is superior to its supine colleague for detecting pneumothoraces (sensitivities of 92% and 50%, respectively), it is not possible to attain upright views in all patients with blunt or even penetrating injury (unpublished data, 2009).1–6,15–20
More recently, the trauma literature has also supported the use of thoracic ultrasonography in the initial assessment of injured patients.8–13 This technique of early bedside thoracic imaging to detect pneumothoraces has a sensitivity of 92%–100% among patients with blunt trauma.8,9,11,12 Whereas this test performance is impressive, it is unclear whether the definition of an occult pneumothorax should be expanded to include patients whose pneumothoraces were occult to supine anteroposterior chest radiography, but are subsequently identified on extended focused assessment with sonography for trauma (eFAST) examination.14 We believe that if chest radiography is performed before a definitive test (thoracic ultrasonography or CT), then the pneumothorax should be defined as occult. More commonly the pneumothorax is first identified on an eFAST examination performed early in the resuscitation phase, and hence before the completion of radiography. In this setting, the pneumothorax is only occult to supine anteroposterior chest radiography because of the test’s poor sensitivity.
Finally, the concept of a secondary occult pneumothorax has been recently introduced.26 This occurs when supine anteroposterior chest radiography fails to detect a residual pneumothorax following tube thoracostomy (i.e., before definitive CT). Secondary occult pneumothoraces can be large in size (20 CT images) and are common in frequency (57% of all postinsertion patients).26
Incidence of occult pneumothoraces
The published incidence of occult pneumothoraces has remained remarkably constant during the past 5 years. It continues to reflect the inception cohorts involved (unpublished data, 2009).19–25 Although original literature describing the incidence of occult pneumothoraces varied from 3.7% in injured children presenting to an emergency department,28 to 64% in multitrauma patients after tube thoracostomy with an average injury severity score of 30,29 most publications approximated 5% among all injured patients. Recent publications have confirmed these values with incidences of occult pneumothorax ranging from 1.8% using International Statistical Classification of Diseases and Related Health Problems, ninth revision, codes in a diagnostic level-II study of 21 193 injured patients,23 to 4% among patients with minor blunt thoracic trauma (level III),25 to 6% of all patients with blunt injuries in a given trauma registry (level III).19 When analysis is restricted to patients who underwent CT, the incidence is significantly higher. In a retrospective level-III study of 2326 patients who received full thoracic CT, Plurad and colleagues27 identified an incidence of 4.4%. Additional publications incorporating 88 (level III), 27 (level III) and 338 (level III) patients with blunt injuries noted incidences of occult pneumothorax of 8%,24 11%22 and 15%,19 respectively. This rate also depends on the extent of CT used. With a recent report that 16% of all patients with occult pneumothoraces had evidence of the diagnosis solely on selected images of the thoracic component of their CT scan,19 it is unclear how many more patients would have been diagnosed had all studies included chest CT. Clearly the frequency of occult pneumothoraces depends directly on the imaging modalities applied in any particular study.
The reported proportion of pneumothoraces that are occult compared with those actually seen on supine anteroposterior chest radiography remains variable among recent publications. Level-III and -IV studies of varying quality identified occult pneumothorax rates from 30% to 55%.8,19,22–24 On closer examination, however, it is often unclear who is actually making the diagnosis of an occult pneumothorax at these centres. Most studies employ board-certified radiologist dictations of the initial supine anteroposterior chest radiograph to determine the rate of occult pneumothoraces. Although this methodology is the most precise way to identify the true incidence of occult pneumothoraces, it may not be the most accurate with regard to the actual treatment decision for a given patient. In a 17-month prospective level-II study, the incidence of occult pneumothoraces appeared as high as 76% when interpreted by the trauma service at the time of admission.20 Although the team may have good interpretive skills, they do not routinely have the luxury of prolonged interpretation times, a perfectly lit environment and premium digital monitors like our radiology colleagues.21 Although this is concerning, it is also a more realistic view of patient treatment in a real-time environment. It is these initial interpretations, not the delayed dictations, that result in tube thoracostomy, and therefore exposure to the 22% risk of major complications.30–33
Although the incidence of occult pneumothoraces among patients with blunt injuries is now well described, the frequency of this entity in penetrating trauma was previously unknown. Up to 17% of all pneumothoraces in patients injured by penetrating mechanisms were occult in a recent level-III study of 5552 admissions at Grady Memorial Hospital (unpublished data, 2009). In the subset in which only supine anteroposterior chest radiography was available, this rate approached 80%. Fortunately, upright chest radiography detected 92% of all pneumothoraces and were possible in most patients (87%) (unpublished data, 2009).
In summary, the incidence of occult pneumothoraces in patients with blunt trauma approximates 2%–15% depending on whether all patients in a given registry, or only those that underwent CT, are included. This value may increase substantially when the initial chest radiography interpretation is done by the trauma team. The frequency of occult pneumothoraces in victims of penetrating trauma approaches 17%, but may be reduced when upright chest radiography is used. These new data once again highlight the poor test performance of supine anteroposterior chest radiography for detecting pneumothoraces, and the requirement for complete thoracic CT if the diagnosis of an occult pneumothorax must be ruled out in a given trauma patient.
Distribution of occult pneumothoraces
Clinicians often state that the reason a given pneumothorax is occult to supine anteroposterior chest radiography is because of its extremely small size or “atypical” intrathoracic distribution. In a level-IV study of 89 patients with 103 CT-identified pneumothoraces, the size and distribution of overt and occult pneumothoraces were determined to be statistically similar, however.26 Regardless of type (overt, occult or secondary occult), most pneumothoraces were anterior in the supine patient.26 Furthermore, the mean number of CT images with evidence of an overt or occult pneumothorax, as well as their comparative size index (number of images multiplied by the maximum width of a given pneumothorax), were similar.26 As mentioned previously, it is important to note that 16% of all occult pneumothoraces were evident only on selected thoracic images of the chest CT.19 This reaffirms the belief that if it is essential to rule out an occult pneumothorax, complete CT of the thorax must be performed. In a recent unpublished audit from our institution, 12 of 20 (60%) patients with evidence of an occult pneumothorax solely on chest CT (i.e., not abdominal CT) could have also been diagnosed using the pulmonary window settings during evaluation of the cervical spine component of the CT imaging series.
Risk factors for occult pneumothoraces
In an effort to compensate for the poor diagnostic sensitivity of supine anteroposterior chest radiography,6,15–18 a series of readily apparent clinical markers should ideally be available to the clinician early in the resuscitation of a severely injured patient. These risk factors could then be used to make treatment decisions for patients with a high probability of having an occult pneumothorax. A level-III retrospective study of 338 severely injured patients identified subcutaneous emphysema (odds ratio [OR] 5.47), pulmonary contusions (OR 3.25) and rib fractures (OR 2.65) as clinical risk factors for the presence of an occult pneumothorax.19 Unfortunately, on a subsequent prospective level-II evaluation by the same group, only subcutaneous emphysema remained independently predictive of occult pneumothoraces.20 Although 15% of patients with occult pneumothoraces have concurrent subcutaneous emphysema, virtually all patients with subcutaneous emphysema have an underlying overt or occult pneumothorax.19,20 As a result, the absence of subcutaneous emphysema is insufficient to rule out an occult pneumothorax.24 Lastly, in a level-III study of minor blunt thoracic trauma, Misthos and colleagues25 also identified the presence of associated chest wall muscle contusions in 79% of patients with occult pneumothoraces.
In addition to clinical variables available in the trauma bay, it was also unclear how particular physicians might affect the incidence of occult pneumothoraces. As previously noted, there has been some suggestion that the radiologic skills of the admitting trauma team may be inferior when compared with delayed interpretations by board-certified radiologists.20,21 It was also unknown whether the ability to diagnose occult pneumothoraces via an examination with such poor test characteristics (i.e., supine antero-posterior chest radiography) varied among radiologists themselves. A simple level-III study using 3 heterogeneous blinded radiologist cohorts to reinterpret 55 supine antero-posterior chest radiographs found that nearly one-quarter of all occult pneumothoraces might have been inferred from subtle radiologic findings missed by the initial radiologist.21 Although the “miss rate” did not depend on the mix or number of interpreting radiologists, poor agreement was displayed across all evaluations.21 Furthermore, up to 25% of all supine anteroposterior chest radiographs were considered “inadequate” for reliable interpretation.21 With this high level of disagreement among radiologists, it is not surprising that trauma team members practising in challenging and distracting settings observe more occult pneumothoraces.20 This rate of “missed” occult pneumothoraces of 20% was recently confirmed by Lamb and colleagues22 in a study of 134 blunt trauma patients (level III). The most common missed radiologic findings were deep sulcus and crisp cardiac silhouette signs.21,22
Treatment of occult pneumothoraces
Although current level-II and -III evidence indicates that observation of small occult pneumothoraces (i.e., that do not cross the midcoronal line) without tube thoracostomy in trauma patients not receiving mechanical ventilation is likely safe (unpublished data, 2009),1–6,15–27 actual clinical practice remains varied. Whereas 65% of injured patients received a tube thoracostomy in cases of overt pneumothoraces, only 31% underwent chest tube insertion when their pneumothoraces were occult (level III).19 In spite of data confirming overt and occult pneumothoraces as similar in size and distribution,26 this variance in practice reflects the difficulty clinicians have in determining the appropriate therapy for occult pneumothoraces. When patients receiving mechanical ventilation are included, the rate of chest tube insertion is better delineated. In the largest retrospective publication (level III), 102 occult pneumothoraces were identified during a 7-year period.27 Only 12 (12%) of these patients underwent chest tube insertion, irrespective of mechanical ventilation.27 This group reported no complications and therefore cast doubt on the practice of tube thoracostomy for all but the largest occult pneumothoraces. The overall rate of chest tube insertion varies depending on the patient cohort. Rates of 25% in patients with “minor” chest trauma (level III),25 40% in a small level-III emergency medicine study,22 59% in a large retrospective level-II review of 21 193 injured patients,23 and 82% in patients with concurrent occult hemothoraces (level III)24 have recently been published. On inspection of the patients receiving mechanical ventilation as a separate entity, more patients with overt pneumothoraces (95%) also receive chest tube therapy than those with occult pneumothoraces (76%).19 This supports observed insertion rates of 83%–100% in other recent studies and is again intriguing when one considers the similar size and distribution of all pneumothorax types.22,23,26
The highly variable rate of chest tube insertion for pneumothoraces in blunt trauma is also evident among victims of penetrating injury. In spite of no observed difference in hemodynamic stability or patient characteristics, the rate of tube thoracostomy was 95% and 56% in patients with overt and occult pneumothoraces, respectively (level III) (unpublished data, 2009). Similar to blunt trauma, we suspect it relates to the antiquated notion that occult pneumothoraces “must be smaller” than overt pneumothoraces. The concept of overtreatment is particularly important with tube thoracostomy as it is associated with an up to 22% rate of major complications (level III).30 These include insertional (intercostal artery or intra-parenchymal lung injuries), positional (requiring reinsertion) and infective (empyema or wound infection) issues.2–4,30–33 This risk of complications can be objectively compared with recent level-III studies describing patients who were closely observed and for whom tube thoracostomy was initially avoided.19,22,25 None of these patients had a major complication. Similarly, a minority of patients required placement of a subsequent chest tube after progression of a pneumothorax during observation (9%–11%).19,25 The need for chest tube insertion in patients undergoing mechanical ventilation also remains unclear. Although retrospective studies indicate that tube thoracostomy may not be required,1,19 only 2 level-I randomized trials (36 patients) are available. Unfortunately, they arrive at opposite conclusions.4,5
Finally, a unique level-II publication has constructed a new formula comprised of the largest diameter of the air pocket and its relationship to the pulmonary hilum.23 Whereas this study appears to predict the placement of chest tubes by clinicians for occult pneumothoraces in a retrospective fashion, it has yet to be validated as an accurate prospective predictor.
Taken as a collective, there is still clinical equipoise regarding treatment of occult pneumothoraces. Whereas it appears that small to moderate occult pneumothoraces in patients not receiving mechanical ventilation can be observed,1,34 recent literature does not definitively identify which subset of patients receiving mechanical ventilation can safely avoid tube thoracostomy. This likely explains the variation in rates of chest tube insertion, as well as in opinion, among clinicians. Fortunately a prospective multicentre randomized trial is now underway to study this issue.
Thoracic ultrasonography for the detection of occult pneumothoraces
Bedside thoracic ultrasonography, as part of the eFAST examination for trauma patients,14 appears to overcome many of the limitations of supine anteroposterior chest radiography. The diagnostic sensitivity of this modality for detecting pneumothoraces ranges from 92% to 100% among patients with blunt injuries.8,9,11,35 It also allows maintenance of spine precautions. Although the superiority and utility of ultrasonography in this setting is seldom debated,10 the absence of lung sliding is virtually pathoneumonic for an occult pneumothorax in a previously normal chest.11 Potential pitfalls such as pleural adhesions and emphysematous bullae36 are also less common in populations with trauma injuries. Even in these situations, however, other ultrasonography signs can be helpful in diagnosing injuries.36–38 Thoracic ultrasonography has also been shown to be accurate with respect to identifying the size and extension of occult pneumothoraces.8,9,11 When compared with gold standard thoracic CT, ultrasonography confirmed the size of a pneumothorax within 1.9 and 2.3 cm in level-I studies of 25 and 56 patients, respectively.8,9 Unfortunately this accuracy is lost 24 hours after chest tube insertion (level II).39 The authors postulate this is a result of intrapleural adhesions.39 It is unclear if this also applies to patients with occult pneumothoraces who do not receive a tube thoracostomy.
Suggested algorithm for the diagnosis of thoracic injuries and occult pneumothoraces
The extent of CT required for patients with blunt trauma is a contested topic. Whereas some authors believe that the diagnosis of all occult injuries via routine “pan scans” is critical, others believe the pendulum has swung too far. Plurad and colleagues27 recently identified a 24-fold increase in the number of occult thoracic injuries diagnosed during the past 6 years as a direct result of a 10-fold increase in chest CT (level III). Because few of these occult injuries required changes in management, they called for continued refinement of the indications for thoracic CT after negative chest radiography.
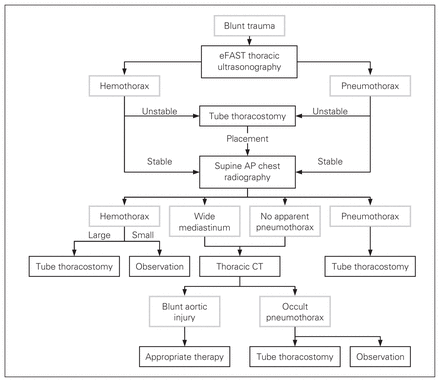
Based on the recent publications summarized in this review, as well as the risk of radiation exposure associated with CT,40–42 our diagnostic algorithm for blunt chest trauma is included (Fig. 1). Although no sequence is 100% sensitive, this approach is limited only by the rare occult pneumothorax or blunt aortic injury missed by thoracic ultrasonography (0%–8%)8,9,11,35 and supine chest radiography (2%–7%),43,44 respectively. This algorithm assumes competency in the performance and real-time interpretation of thoracic ultrasonography. It also recognizes the immense overall screening value of supine chest radiography. Although we consider the chest radiography and eFAST examination to be complementary, we conceptualize the eFAST examination as a simple extension of the clinician’s physical examination. As a result, the eFAST examination is employed as the initial diagnostic modality (Fig. 1). Should the patient ever become hemodynamically unstable, tube thoracostomy is completed without any further imaging. The use of eFAST should not delay the chest radiography, as an organized resuscitation can obtain both images simultaneously if a radiologic technician is present. If overt pneumothorax or hemothorax are identified a chest tube is inserted. Thoracic CT is reserved for the presence of a wide mediastinum, or for cases of potentially false-negative chest radiographs for a pneumothorax (Fig. 1). All occult pneumothoraces must be treated with tube thoracostomy if close clinical observation is not possible.
Diagnostic algorithm for blunt thoracic trauma. AP = anteroposterior; CT = computed tomography; eFAST = extended focused assessment with sonography for trauma.
Conclusion
Recent literature has answered many of the remaining questions surrounding the increasingly common diagnosis of an occult pneumothorax. Whereas the incidence of occult pneumothoraces approximates 5% of all trauma registry patients, this value is likely an underestimate based on the limited proportion of thoracic CT performed among database studies. The use of board-certified radiologist dictations, rather than the real-time trauma team interpretations, also minimizes this value. The size and intrathoracic distribution of overt and occult pneumothoraces appears to be similar. This clouds the rationale for the higher rates of chest tube insertion observed among patients with overt pneumothoraces compared with those with occult pneumothoraces. This pattern extends to both patients recieving mechanical ventilation and those with penetrating trauma. It is clear that only subcutaneous emphysema is predictive of a concurrent occult pneumothorax in the early resuscitation phase. Unfortunately, it cannot be relied on as the sole clinical risk factor for an occult pneumothorax because most of these patients do not have subcutaneous emphysema. Thoracic ultrasonography, as part of a bedside eFAST examination, detects nearly all pneumothoraces and should be incorporated into a clinician’s diagnostic algorithm for chest trauma. The final remaining question in the occult pneumothorax literature is whether a predictable cohort within the group of patients undergoing mechanical ventilation can be safely observed without tube thoracostomy. This would avoid the high risk of chest tube–related complications. With the completion of a prospective multicentre trial this last query should be elucidated.
- Accepted April 28, 2008.