Deep venous thrombosis and pulmonary embolism are known complications of Crohn disease, but arterial thrombosis is extremely rare. We present the case of a man with Crohn disease with extensive primary thrombosis of abdominal aorta who died of diffuse visceral ischemia. Thus the possibility of large-vessel screening for thrombosis in patients with severe active Crohn disease may be considered.
Case report
A 41-year-old man who had received a diagnosis of Crohn disease 18 months previously was referred to our hospital on postoperative day 6 for continuing care as he had undergone 2 laparotomies 5 days apart, the first for ileal gangrene and the subsequent one for anastomotic dehiscence. Six months before this he had undergone another laparotomy for jejunal perforation owing to Crohn disease. On examination, the patient was sick-looking and wasted but normotensive and afebrile. Cardiopulmonary exam results were within normal limits except for bilateral decreased basal air entry. Abdominal examination revealed mild diffuse tenderness, sluggish bowel sounds, a functioning ileostomy and an abdominal drain having serosanguinous output. Results of laboratory tests showed a leukocyte count of 26 × 109/L, hemoglobin of 90.7 g/L and partial thromboplastin time of 64.7 s. His potassium was 2.7 mmol/L, bicarbonate 20.6 mmol/L, urea 26.8 mmol/L and creatinine 281 μmol/L. His bilirubin level was 29.0 μmol/L and lactate was 3.7 mmol/L. Thrombophillic workup was normal.
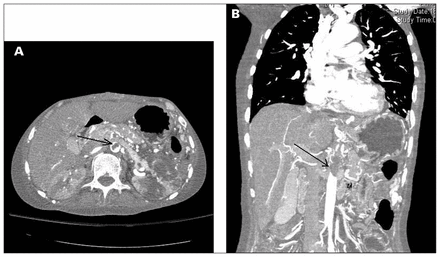
He had a normal electrocardiogram with chest radiograph showing bilateral minimal pleural effusions. Lower limb and carotid Doppler findings were normal and transthoracic echo showed free-floating thrombus in the arch of aorta. A computed tomography (CT) aortogram (Fig. 1) showed extensive aortic thrombus extending from the celiac artery to the ostia of renal arteries.
(A) Cross-sectional image of the abdomen on a computed tomography (CT) aortogram demonstrating thrombosis of the abdominal aorta (arrow) associated with infarction of the left kidney. (B) Coronal reformatted CT scan showing extensive thrombus in the abdominal aorta (arrow), which had blocked the ostia of celiac, superior mesenteric and left renal arteries. A number of collateral vessels could be seen.
The patient received intravenous fluids, broad spectrum antibiotics, heparin infusion protocol and total parenteral nutrition, and he was transferred to the intensive care unit with renal failure, visceral ischemia, sepsis, metabolic acidosis and dyselectrolytemia. He was not fit for major surgical intervention because of diffuse visceral ischemia, and the interventional radiologist failed to cannulate the ostia of celiac, superior mesenteric and left renal artery on attempted thrombolysis, but the right renal artery was stented. On his second day in the intensive care unit, the patient had worsening sepsis with acidosis, pulmonary edema and severe renal failure. Despite full organ support, he died of severe septic shock on his eighth day in intensive care.
Discussion
We describe the case of a patient with primary extensive aortic thrombosis of the abdominal aorta and descending aortic arch in association with severe Crohn disease that had no identifiable risk factor for thrombophilia, presenting quite late when no treatment was successful.
Patients with inflammatory bowel disease (IBD) are 3 times more likely to experience thromboembolic complications than the general population, and such events complicate the conditions of 1%–8% of patients with IBD. They are most often venous in origin rather than arterial, with deep venous thrombosis pulmonary embolism being most common.1 More than half of these cases occur during disease exacerbations or in the presence of complications like fistulae and abscesses.2,3
Aortic mural thrombosis is extremely rare, and after extensive review of literature, we could only find 12 cases reported in English literature.2 The characteristics of these cases, together with our patient’s case, are presented in Table 1. Possible factors that led to thrombosis in our patient were postoperative state and severe active disease.
Characteristics of patients with inflammatory bowel disease and aortic thrombosis, including the patient presented in this report (n = 13)
The exact cause of thrombophilia in patients with IBD is unknown. All components of coagulation cascade are thought to be involved in pathogenesis.2,3 As most of the patients with IBD and aortic thrombosis have active disease or complication, endotoxemia has been incriminated as it is able to activate coagulation cascade, and further systemic endotoxemia has been detected in active and fistulating Crohn disease.4 The increased levels of proinflammatory cytokines (tumour necrosis factor and interleukin-6) and enhanced expression of CD40 ligand on platelets are thought to play an important role by promoting a prothrombotic state. In addition, platelet leukocyte aggregates, which cause microinfarction and thrombus formation by enhancing the production of tissue factor have also been implicated, and they are increased in patients with IBD.5
Treatment should involve management of underlying bowel disease and prevention or correction of acquired risk factors leading to thromboembolism. Therapeutic anticoagulation may be an effective alternative to surgical thrombectomy and also prevents recurrence. Prophylactic doses of low molecular weight heparin, as recommended by the British Society of Gastroenterology, should be used in patients with IBD, especially with evidence of increased disease activity and gastrointestinal complications in the postoperative period.1–3 The outcome of acute mural thrombi of the aorta ranges from complete resolution to amputation to death.
We conclude that aortic thrombosis is a devastating complication of IBD. The most important risk factors for aortic thrombosis in IBD are severe active disease, postoperative status, smoking and hormone use. We suggest possible screening of the abdominal aorta in patients with severe, active IBD, particularly in the presence of these risk factors.
Footnotes
Competing interests: None declared.