Abstract
Background: Our objective was to identify morphologic trends in elective and emergency endovascular aneurysm repair (EVAR). This work will inform hospitals with endovascular programs about the diameters and lengths of endostents that should be available to efficiently care for patients with these conditions.
Methods: We performed a retrospective review of patients undergoing elective (n = 127) and emergency (n = 17) EVAR. Using computed tomography and 3-dimensional reconstructions, we evaluated the following: diameters of the aneurysm (D3), the aorta at the superior mesenteric (D1) and renal (D2a,b,c; 3 levels) levels, the iliac arteries (D5a,b; right and left) and the aortic bifurcation (D4); lengths from the lowest renal artery to the distal aspect of the aortic neck (H1), to the aortic bifurcation (H3), to the right and left iliac bifurcations (H4a,b); and angles of the origin of the common iliac arteries on the transverse plane (A1). We used descriptive statistics of trends within groups and independent sample t tests.
Results: In elective and emergency aneurysm repair, D2max (26, standard deviation [SD] 3, mm v. 30.7 [SD 3] mm), D5a (16 [SD 4.7] mm v. 19.3 [SD 5] mm), D5b (15.3 [SD 4] mm v. 18.1 [SD 3.6] mm), H1 (25.6 [SD 8.6] mm v. 18 [SD 2] mm), H4a (173 [SD 22] mm v. 189.5 [SD 22] mm) and H4b (174 [SD 25] mm v. 190 [SD 14] mm) were significantly different between the 2 groups (p = 0.001, p = 0.006, p = 0.007, p < 0.001, p = 0.05 and p = 0.01, respectively). H3 (118 [SD 17] mm v. 121.5 [SD 13.5] mm) was not significantly different (p = 0.40). In elective patients, A1 identified the right common iliac more frequently anterior relative to the left common iliac (mean 23°, SD 16°).
Conclusion: Significant anatomic differences between elective and emergency patients will require hospitals to stock separate endovascular devices to treat abdominal aortic aneurysms in both groups.
Abdominal aortic aneurysms (AAAs) are more prevalent among men than among women over 64 years of age (6% v. 1%).1–4 In Canada, AAAs are responsible for about 1658 deaths per year.5 A population-based study has shown that 66% of patients with ruptured AAAs die before undergoing operative repair.6 The risk of rupture increases with AAA size from 1% per year for a diameter of 4.0–4.9 cm to 26% for a diameter greater than 6.0 cm.7–9 Screening, timely treatment and reduction in perioperative mortality will reduce the burden of this disease.5
Until recently, repair of AAAs required invasive open surgery. This resulted in perioperative mortality of 5%–8%10–15 and risk of complications of 15%–20%.16 Endovascular aortic aneurysm repair (EVAR) is an accepted, minimally invasive modality of treatment with reduced peri-operative mortality of 1%–2%16–18 and risk of complications of 5%–10%.16 Although certain anatomic criteria must be satisfied, a significant proportion of patients (40%–60%) is eligible for EVAR.19,20 To fit a patient’s anatomy, EVAR relies on customized modular stents that are deployed at the site of the aneurysm to divert blood flow from the weakened walls of the aneurysm.20–22 The components available for EVAR are so numerous that more than 65 000 combinations may be possible using components from a single manufacturer. This number will increase exponentially if hybrid endografts, made of components derived from multiple manufacturers, are also considered. The cost of these endovascular grafts limits the applicability of this treatment and is a serious consideration for hospitals, which need to stock a large number of components for patients undergoing elective and emergency EVAR.
This study defines trends in aortoiliac anatomic measurements in patients undergoing elective and emergency EVAR. The results may be used by hospitals to improve cost-efficiency by stocking a limited number of devices and permitting expansion of existing endovascular programs or facilitating the development of new ones. In addition, the results of this study may inform the industry to optimize the production of stents in terms of diameters, lengths and configurations, and possibly improve graft hemodynamics.
Methods
We performed a retrospective review of prospectively collected data of consecutive patients undergoing elective and emergency EVAR from October 2004 to December 2007 in a tertiary vascular centre. We defined ruptured aneurysms as displaying extravasation of contrast outside the limits of the aortic wall. This study received institutional ethics approval.
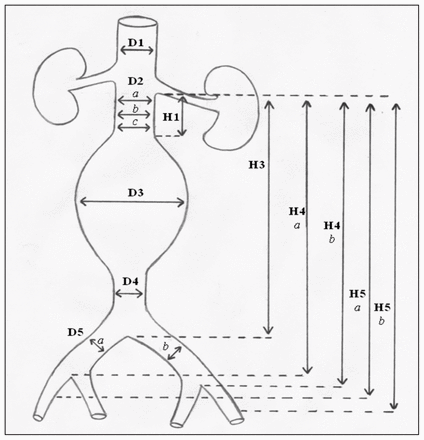
To determine the most common anatomic measurements of elective EVAR patients, we evaluated aortoiliac diameters, lengths and angles. We measured aortic diameters at the superior mesenteric (D1), proximal, middle and distal levels of the infrarenal aortic neck (D2a,b,c), the widest section of the abdominal aneurysm (D3) and the region immediately proximal of the aortic bifurcation (D4). We assessed diameters of the right and left common iliac arteries (D5a,b) at the estimated landing zone. We measured lengths from the lowest renal artery to the distal aspect of the aortic neck (H1), the aortic bifurcation (H3), the right and left iliac bifurcations (H4a,b) and the right and left external iliac arteries (H5a,b) in patients who required landing in the external iliac artery (Fig. 1). Using the proximal, middle and distal measurements (D2a,b,c), we defined the shape of the aortic neck as cylindrical, flared, reversed flare or hourglass. We used a change in diameter of greater than 10% between the most proximal and most distal measurements as the threshold for classification. A flared neck implied that the distal diameter was larger, whereas a reversed flare assumed the opposite configuration. For patients in the emergency EVAR group, we did not measure D1, D3, D4, H5a and H5b given the emergency of the settings and the fact that these variables were not relevant to choose the appropriate stent graft.
Aortoiliac anatomic measurements. Note: D1 = diameter of the superior mesenteric artery; D2a,b,c = diameter at the proximal, middle and distal levels of the infrarenal aortic neck, respectively; D3 = diameter at the widest section of the abdominal aneurysm; D4 = diameter at the region immediately proximal of the aortic bifurcation; D5a,b = diameters of the right and left common iliac arteries, respectively; H1 = length from the lowest renal artery to the distal aspect of the aortic neck; H3 = length from the lowest renal artery to the aortic bifurcation; H4a,b = length from the lowest renal artery to the right and left iliac bifurcations, respectively; H5a,b = length from the lowest renal artery to the right and left external iliac arteries, respectively.
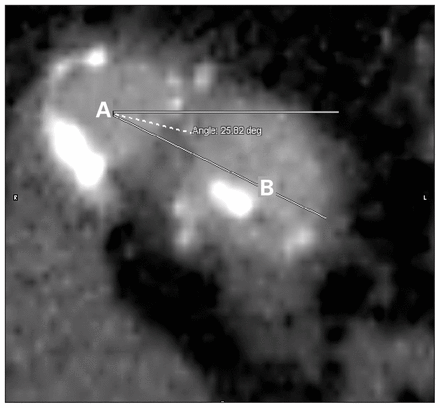
In addition, we described the position of the origin of the common iliac arteries relative to each other and expressed the relation as an angle on the transverse plane (A1). To accomplish this measurement, we established the estimated centre of the right common iliac artery as the origin of the angle. From this origin, 2 lines, one horizontal and one through the estimated centre of the left common iliac artery (LCIA), were drawn. The position of the LCIA was defined in degrees of an angle. We described the angle as positive if the LCIA was anterior, and negative if the LCIA was posterior in relation to the right common iliac artery (RCIA). We defined an angle of 5° or less in either direction as an angle of zero, and we considered both arteries to be in the same plane (Fig. 2).
A computed tomography scan showing the angle used to define the relative position of the iliac arteries: centre of the right common iliac artery (A) and centre of the left common iliac artery (B).
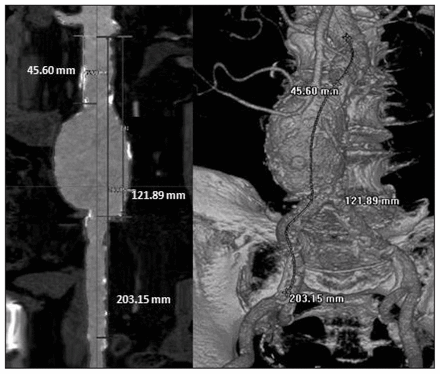
Between October 2004 and September 2006, we took measurements of elective EVAR from axial scans with slice thickness ranging from 5 to 3 mm, and reconstructions created with 1.5-mm overlap. Starting in September 2006, we assessed measurements using computed tomography (CT) scans obtained with a 64-slice scanner and postprocessing with an Aquarius workstation v. 3.5 (TeraRecon Inc.). The senior vascular surgeon (C.S.C.) obtained all measurements, which were entered in a specifically designed planning form. We performed measurements of diameters from adventitia to adventitia, and we estimated lengths using centre line reconstructions (Fig. 3).
Centre line measurements in a stretched view of the aorta (left) and in 3-dimensional rendering (right): H1 (neck of the aneurysms) is 45.60 mm; H3 (distance between the lowest renal artery and the aortic bifurcation) is 121.89 mm; and H4a (distance from the lowest renal artery to the iliac bifurcation) is 203.15 mm.
We obtained emergency EVAR measurements from axial CT scans generated from 8- to 64-slice scanners while the patient was being prepared for surgery. In each case, the senior vascular surgeon (C.S.C.) evaluated the appropriate variables. We assessed measurements using different CT readers according to availability. In this group of patients, the diameter measurements could usually be assessed accurately from the available imaging techniques. Lengths were often estimated by a combination of tomographic imaging and intraoperative angiography.
Three investigators entered measurements into a database. At least 2 investigators were present at each session to minimize errors associated with entering the variables and to discuss any disagreements that arose during the process. Investigators resolved any remaining disagreements through discussion with the senior author (C.S.C.). Excluded from the analysis were thoracic (n = 25) and thoracoabdominal (n = 12) aneurysms. We conducted statistical analysis using SPSS software v.15.0. We expressed diameters, lengths and angles in millimetres and as means and standard deviations (SDs), and we used the maximum and minimum values to represent the range of measurements for each variable. We calculated the mode, with the number of counts it appeared for each measurement, to determine the most frequent dimensions.
We calculated missing data points for elective patients by linear interpolation for all variables excluding D1.23 This involved averaging the values closest to (e.g., above and below) the missing data point to provide an estimate of the missing value. We interpolated the missing H5a,b values when the patients’ operative report showed an external iliac landing zone. We estimated missing D1 values using the largest of the 3 aortic neck diameters D2a,b,c (D2max). We deemed this a more accurate technique over interpolation owing to the proximity of the 2 measurements in relation to the patient’s anatomy.
We used independent sample 2-tailed t tests to compare variables when the assumption of equality of variances (Levene test) was satisfied, and we applied an appropriate correction when this assumption was not satisfied. We used a Fisher exact test to evaluate categorical variables and considered p ≤ 0.05 significant.
Results
Overall, 158 patients underwent EVAR. Data were not available for 12 patients undergoing elective EVAR and for 2 patients with ruptured aneurysms, leaving 144 patients for evaluation (127 elective and 17 emergency). One hundred elective patients were selected for the analysis of A1.
Among the included elective patients, missing values per variable were as follows: D1 = 56, D3 = 22, D4 = 21, D5a = 8, D5b = 5, H3 = 14, H4a = 4, H4b = 9, H5a = 2, H5b = 2. There were no missing values for D2 and A1. No missing values were present among the emergency patients. Table 1 and Table 2 describe the variables for elective and emergency patients, respectively. Table 3 contains the results of the independent samples t tests comparing the 2 groups. Among elective patients, D1 had a mean diameter of 26.3 (SD 3.2, range 21) mm. The most frequent measurement of 28 mm occurred 22 times among the sample population. The average largest aneurysm diameter (D3) in the elective group was 60 mm (SD 8.7, range 55, mode 55 mm, count = 20). D4 mean was 28.9 mm (SD 7.9, range 45; modes 22 and 25 mm, each with a count of 11).
Descriptive statistics for patients undergoing elective endovascular abdominal aortic aneurysm repair
Descriptive statistics for patients undergoing emergency endovascular abdominal aortic aneurysm repair
Comparison of results of independent sample t tests in patients undergoing emergency v. elective endovascular abdominal aortic aneurysm repair
Maximum aortic neck diameter (D2max) and diameter of right and left common iliac arteries (D5a,b)
D2max was smaller in the elective compared with the emergency group (26.2, SD 3.0, range 21 mm v. 30.7, SD 3.4, range 14 mm; p < 0.001). The most frequent D2max measurements were 28 mm and 31 mm, occurring in 23 elective and 6 emergency patients, respectively. No significant difference was found between D1 and D2max measurements in the elective group (p = 0.90).
In elective patients, D5a and D5b were 15.9 (SD 4.7, range 34) mm and 15.4 (SD 3.8, range 30) mm, respectively. The mode for both D5a and D5b was 15 mm, with counts of 18 and 21, respectively. Diameters were greater in the emergency group: 19.3 (SD 4.97, range 18) mm (p = 0.006) and 18.1 (SD 3.6, range 13) mm (p = 0.007), respectively. Among emergency patients, the most frequent measurements were 19 (count = 3) and 20 mm (count = 4) for D5a and D5b, respectively.
Lengths of aortic neck (H1)
H1 was greater in the elective than in the emergency patients (25.7 [SD 8.6, range 46] mm v. 18.8 [SD 2.25, range 7] mm; p < 0.001); modes were 20 mm (count = 29) and 19 mm (count = 4), respectively.
Length from lowest renal artery to aortic bifurcation (H3)
H3 was 118.1 (SD 17.3, range 111) mm in the elective and 121.5 (SD 13.5, range 50) mm in the emergency group (p = 0.40); modes were at 110 (count = 17) and 111 mm (count = 3), respectively.
Lengths from lowest renal artery to right and left iliac bifurcations (H4ab)
There was no difference between H4a and H4b among elective patients, as the analysis yielded measurements of 173.4 (SD 21.6, range 132) mm and 174.4 (SD 25, range 230) mm, respectively. The mode for H4a was 160 mm (count = 11) and 2 modes were present for H4b, 160 and 170 mm (each with a count of 12). In the emergency group, H4a and H4b were greater than in the elective group: 189.5 (SD 22.3, range 86) mm (p = 0.05) and 190.2 (SD 14.1, range 43) mm (p = 0.012), respectively. In the emergency group, the mode for H4a was 187 mm (count = 3), and 2 modes were present for H4b, 176 and 210 mm (each with a count of 3).
Lengths from lowest renal artery to right and left external iliac landing zones (H5ab)
H5a and H5b were 235.8 (SD 31.9) mm and 227.8 (SD 23.4) mm among elective patients, respectively. Modes are not presented since measurements occurred only once per variable.
Angle of origin of common iliac arteries (A1) and shape of the neck
In the elective group, the most common configuration regarding the angle of origin of the common iliac arteries involved the RCIA being anterior relative to the LCIA (23.1°, SD 16°, range 61°). No patients were found to have the opposite configuration.
In elective patients, a cylindrical configuration of the neck was the most commonly represented. This distribution was reversed in the emergency group, in which the flared neck was represented in greater number than in the elective (p ≤ 0.001) (Table 4).
Shape of neck distribution in patients undergoing elective and emergency endovascular abdominal aortic aneurysm repair
Discussion
Accurate assessment of aortic and iliac morphology is important in endovascular graft design. A review of 18 centres in Australia has shown that aneurysm morphology can be predictive of outcome, finding aneurysm neck lengths of less than 10 mm to be associated with endoleaks (odds ratio 8.3, 95% confidence interval 8.0–8.6).24
We have shown that elective patients undergoing EVAR frequently have neck diameters of 26–28 mm. Hence, endovascular stents with a diameter of 28–32 mm are likely to cover a large range of anatomies. In cases of ruptured aneurysm, the size of the patient’s neck is larger and the length shorter than in cases of elective EVAR. Most emergency patients were found to have a neck diameter in the 30–31 mm range, requiring larger stents of 35–36 mm in diameter. Although there was a slight increase in the length of H3 for ruptured aneurysms, this did not reach statistical significance. Iliac arteries were of greater length and diameter in the ruptured group compared with the elective group. The relevance of these findings is that main bodies with similar lengths may be kept in inventory for both groups, whereas leg extensions to deal with iliac arteries of 70–80 mm in length and 19–20 mm in diameter are necessary for ruptured aneurysms.
Previous studies have described aortic and iliac trends in patients with AAAs and have compared the quality of different imaging techniques used to measure anatomic variables in these individuals. There have been a limited number of studies evaluating these variables in ruptured AAAs and even fewer that undertook a comprehensive anatomic evaluation of all relevant variables using dedicated postprocessing software of CT images.
Beebe and colleagues,25 using a retrospective review of CT images and cut plain traditional contrast angiography, conducted an anatomic study in 50 patients with AAAs who did not undergo EVAR. They found that average neck diameters were 29 (SD 0.7) mm by CT and 26 (SD 0.56) mm by angiography. The difference in measurements is explained by the fact that angiography is only able to image the inner lumen of the vessel. The average H3 was significantly longer than in our study (156 [SD 23.8] mm).25 The magnitude of this difference is difficult to explain: the type of population studied (ours was only of patients who underwent EVAR) and the different technique of measurements used (they used neither centre line reconstructions of CT images or graduated catheters and traditional angiography) may in part account for it.
Sprouse and colleagues26 compared interrater agreement for anatomic measurements in 5 patients from dimensional reformatted CT reconstruction versus axial CT scans. The average neck diameter with 3-dimensional reconstructions was 26.5 versus 29.3 mm with axial scans. With axial scans, larger diameter may result if measurements are taken along the major axis of oval images derived from angled aortic necks cut obliquely. In addition, H1 measurements were on average 23 and 21 mm, respectively. The authors concluded that 3-dimensional reconstructions provide accurate results, with a greater interrater agreement for measurements compared with axial CT scans (κ = 0.81 v. 0.59).26
Resch and colleagues27 studied anatomic variables in elective EVAR candidates and collected measurements using CT and angiography. Diameters were not measured adventitia to adventitia, and lengths were calculated by summing the number of 3-mm CT scans. The authors reported average diameters of 23 (SD 3.5) mm for D2 and 17 (SD 7) mm for D5a,b when measured by CT. Also using CT, the authors found H1 and H3 lengths to be 26 (SD 12) mm and 120 (SD 13) mm, respectively.27
Two studies examined the anatomic variables in ruptured and elective aneurysms. Wilson and colleagues28 conducted a retrospective study of 50 elective and 51 ruptured aneurysms in patients who did not undergo EVAR. Using calipers and physical references, they obtained measurements from CT images with 3–5 mm cuts. Measurements were not obtained using centre line reconstructions, and the smallest vessel diameters were recorded. The authors found that the average neck diameter was 26 (SD 4) mm cases of elective EVAR and 26 (SD 9) mm in cases of ruptured aneurysms (p = 0.90). Neck lengths were shorter in the ruptured group compared with the elective group (17 [SD 12] mm v. 22 [SD 11] mm). Ruptured aneurysms were also found to have larger diameters at the iliac landing zones, but this trend did not reach statistical significance.28
Lee and colleagues29 examined D2, D3 and H1 in patients who underwent traditional and endovascular aneurysm repair and compared a retrospective group of elective patients (n = 100) with a prospectively collected group of emergency patients. The emergency group comprised patients with ruptured (n = 10) and symptomatic (n = 18) aneurysms.29 Caliper measurements were used to evaluate anatomic measurements. Similar to our study, the authors found the diameters of the patients’ necks to be larger in those undergoing urgent surgery than in the elective group (25 [SD 4] mm v. 23 [SD 3] mm, p = 0.04) and the length shorter (16 [SD 10] mm v. 23 [SD 14] mm, p = 0.017).29
Reliability of measurements from CT images using mechanical means has been shown to be poor. Velazquez and colleagues30 found a 2-fold increase in the number of additional iliac extensions when aortic dimensions were measured with manual calipers compared with digital centre lumen techniques. If the values from a Bland–Altman analysis from one study31 are transformed, the standard error of measurement for experts using electronic calipers is 0.52 mm when measuring the aortic neck and 1.30 mm for the aneurysm sac. The reliability of using CT as the sole modality has been further proven in a study that found a high correlation (R = 0.96) when comparing postprocessing 3-dimensional reconstructions with intravascular ultrasound, the gold standard.32
Computed tomography angiography and postprocessing software have improved from the capabilities of first-generation technology,33 and CT scans are now used in most centres as the primary modality for preoperative assessment.34,35
The inference that may be drawn from our study is strengthened by several characteristics: the large sample size of consecutive patients undergoing elective and emergency EVAR, the use of dedicated postprocessing software specifically designed for this purpose, the comprehensive evaluation of all pertinent anatomic variables and the fact that all measurements were derived by a single rater with expertise in endovascular aneurysm planning and repair. The prospective nature of the study, the large sample size and the homogeneity of the groups render the results generalizable to similar patients presenting to tertiary vascular centres. The use of dedicated postprocessing software and the expertise of the rater increase the internal validity of this study. The former allows centre line reconstructions and increases the precision of length and diameter measurements. Although there is no specific definition for what is required to achieve expertise in endovascular planning, Forbes and colleagues36 have indicated that expertise, defined as the occurrence of complications less than 10%, requires at least 60 procedures. The senior author (C.S.C.), in addition to obtaining specific training, has performed more than 400 EVAR and planned infrarenal, complex fenestrated and branched devices.
Limitations of this work include the need for interpolation of missing data, the lack of a test–retest reliability measure, the relatively small sample size for ruptured aneurysms and the fact that almost one-half of the measurements were not obtained with dedicated postprocessing software.
The results of this work may be used by health care providers to stock a limited number of commonly used stents and to provide timely endovascular treatment for elective and emergency aneurysms. In addition, data relating to the angle of the origin of the 2 iliac arteries may have future implications with respect to stent delivery and design.
Research implications may extend to the engineering of endovascular stents. The morphometric anatomic variables described may help the industry to tailor graft design to patients’ anatomies.
An analysis of aortoiliac dimensions among elective and emergency patients for endovascular repair has revealed significant differences among all anatomic variables, except the length from the lowest renal artery to the aortic bifurcation. Given that measurements tended to be greater among emergency patients, health care providers will need to stock different devices in order to effectively treat abdominal aortic aneurysms in both groups.
Acknowledgements
The authors thank Brandy Cochrane for her assistance in constructing the database of elective EVAR patients.
Footnotes
Competing interests: None declared.
Contributors: Dr. Cinà designed the study. Mr. Kilian and Mr. Dang acquired and analyzed the data. Mr. Kilian and Dr. Cinà wrote the article. All authors reviewed the article and approved its publication.
- Accepted July 13, 2008.