Abstract
Background: The number of displaced midshaft clavicle fractures treated surgically is increasing, and open reduction and intramedullary fixation is an emerging surgical treatment option. The study quality and scientific levels of published evidence in which possible complications of this treatment are presented vary greatly.
Methods: We performed systematic computer-based searches of EMBASE and PubMed/MEDLINE. Studies included for review reported complications after intramedullary fixation alone or in comparison to either treatment with plate fixation and/or nonoperative treatment. The Level of Evidence rating and Quality Assessment Tool were used to assess the methodological quality of the studies. Included studies were ranked according to their levels of evidence.
Results: Six articles were eligible for inclusion and final quality assessment; 3 studies were graded the highest level of evidence. Major complications like bone-healing problems and deep infections requiring implant removal were reported at a rate no higher than 7%. Reported rates for minor complications, such as wound infection and implant irritation that could be resolved without further surgery, were as high as 31%.
Conclusion: The noted rates for major complications requiring additional surgery were low, but implant-related problems that require additional surgery might present with high prevalence. Owing to routine implant removal, treatment with intramedullary fixation often requires an additional surgical procedure.
Fractures of the clavicle account for 5% of all fractures, and 80% of these fractures are located in the middle third of this S-shaped bone.1–4 The incidence of surgical treatment for completely displaced midshaft clavicle fractures (DMCF) is rising owing to poor results reported after nonoperative treatment for this specific subset of patients in recent years.5–10
Intramedullary fixation has emerged as a promising alternative to traditional open reduction and internal plate fixation.11 Advantages of this minimally invasive treatment option include maintaining the fracture hematoma and keeping the periosteum intact, which positively influences bone formation and improves cosmetics owing to the small incisions used.11,12 Different techniques and examples of intramedullary fixation devices have been reported and include the Hagie,9 Knowles13 and Rockwood14 pins and titanium elastic nails (TEN).15
A spectrum of possible postoperative complications, including pin migration, implant failure, deep and superficial infections, refractures and mal- or nonunions has been reported.11,16 In the current literature, however, the scientific level of evidence for reported complications from intramedullary fixation of DMCF and the quality of the studies reporting them varies. The aim of this systematic review was to answer the following questions:
What is the incidence of major and minor complications associated with intramedullary fixation of acute DMCF?
What interventions are available for resolving major or minor complications?
What is the value of reported complications in terms of the scientific level of evidence and the quality at which they are presented?
Methods
Search strategy
A computer-based search for relevant studies was carried out on June 1, 2011, on EMBASE and PubMed/MEDLINE online databases. We consulted the Cochrane Handbook (www.cochrane-handbook.com) for an optimal search strategy. Using the first 2 phases of this strategy in combination with a subject-specific search, a study database was obtained. To ensure inclusion of all relevant studies, reference and citation tracking were performed.
Inclusion and exclusion criteria
Studies were included for quality assessment if they reported on acute, isolated, DMCF treated with intramedullary fixation. Comparative studies in which intramedullary fixation was compared with other treatment modalities were also eligible for inclusion.
Exclusion criteria were studies written in languages other than English, German, Dutch or French. Also, case reports, biomechanical studies, surgical technique studies, review papers and studies involving fewer than 10 patients were excluded. Studies reporting on intramedullary fixation as a treatment for open fractures, pathological fractures, multitrauma, floating shoulders, nonunions or malunions and without a clear distinction in complication rates between these and isolated DMCF were also excluded.
Selection of studies
After completion of the initial search strategy, 2 researchers (F.-J.G.W., O.A.J.v.d.M.) screened the titles and abstracts of potential studies against the inclusion criteria. Studies were then thoroughly read to assess them for final eligibility. Disagreement between the reviewers was resolved by discussion with another independent reviewer (R.M.H.).
Quality assessment
Quality assessment was performed by 2 researchers (F.-J.G.W., O.A.J.v.d.M.) on the final study selection without masking the source or authorship of trial reports. Studies were first labelled according to their level of evidence (Oxford Centre of Evidence Based Medicine, www.cebm.net). The level of evidence rating is divided into 4 levels: level I indicates high-evidence studies, level II moderate, level III low and level IV very low-evidence studies.
To further interpret methodological study quality, we used the modified version of the Cochrane Bone, Joint and Muscle Trauma Group’s former quality assessment tool (QAT, www.cochrane-handbook.com). The QAT is a tool with 11 items (7 on internal validity and 4 on external validity) that can be used to score a study on methodological qualities. Scores can range from 0 to 24. Disagreement between the reviewers about the quality assessment was resolved by discussion with another independent reviewer (R.M.H.).
Data extraction and analysis
Studies were ranked according to their level of evidence. The study characteristics, including design, type of intramedullary fixation and follow-up time were also noted. Complications of intramedullary fixation were identified and categorized for each study. The categories included implant-related problems (medial/lateral protrusion, visible or palpable presence of the implant, migration/telescoping, displacement of intramedullary device with or without displacement of the fracture parts), infection (wound or deep), bone-healing problems (nonunion, delayed union and symptomatic malunion), mechanical failure (angulation or breaking of intramedullary device and corresponding irritation), refractures, nonroutine intramedullary device removal and other complications.
These categories were further subdivided into 2 groups: major and minor complications. Major complications were those requiring additional (nonroutine) surgery to either remove or revise intramedullary fixation devices as a result of the complication presented. Major complications included nonunions, symptomatic malunions, deep infections requiring implant removal, breakage of the implant, angulation of the implant with persistent symptoms requiring removal and refractures after device removal. Minor complications were those not requiring additional (nonroutine) surgery but requiring nonoperative treatment to resolve (i.e., antibiotics in case of an infection). These included wound infections, deep infections not requiring implant removal or irrigation/debridement, irritation, migration and telescoping, angulation of the implant without persistent symptoms and neurovascular problems. Irritation, migration and telescoping of the intramedullary device can often be resolved by minimally invasive shortening of the device under local anesthesia.11 Neurovascular problems tend to be self-limiting over time.11,17,18 To avoid misinterpretation, the definitions of various complications stated in the reviewed studies were used in our analysis as much as possible.
Results
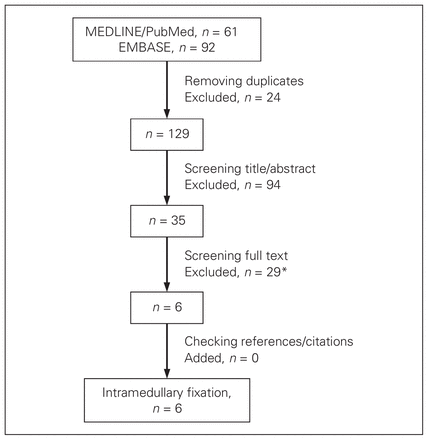
The search in PubMed/MEDLINE identified 61 articles; the search in EMBASE identified 92 articles. After checking for double entries, we excluded articles based on title and abstract content; 35 articles remained for full text screening. Finally 6 articles were eligible for inclusion and final quality assessment (Fig. 1). There was no disagreement between the reviewers about the selection of the final 6 articles.
Literature search and final article selection. The search was conducted on June 1, 2011. Excluded were 1 review paper,19 4 studies in which there was no clear difference in postoperative complications made between closed and open clavicle fractures13,20–22 and 11 studies that included additional morbidity (e.g., floating shoulders, rib fractures) but in which the authors made no distinction between complications after isolated clavicle fractures and fractures with additional injury.14,23–32 In addition, 6 studies included treatment of nonunions or delayed unions and did not make a distinction with the treatment of acute fractures when describing the complications,16,33–37 2 studies were surgical technique papers,38,39 1 study reported on plate fixation,40 1 study lacked a definition for dislocation,41 1 study did not make a distinction between angulation and comminution,42 1 study included fewer than 10 patients15 and, in 1 study,11 authors reported on a previously reported patient population.43
Level of evidence
Three studies were assigned the highest level of evidence (Table 1).18,43,44 The rates of major complications like bone-healing problems and refractures were no higher than 7% in these 3 studies.18,43,44 Judd and colleagues18 reported 1 nonunion that required open reduction and plate fixation and 1 refracture after pin removal that was treated successfully with nonoperative treatment. Two studies reported a total of 3 delayed unions that all healed without further intervention.18,43
Level I evidence studies
Regarding minor complications, Judd and colleagues18 reported an implant-related irritation rate of 31% resulting in the early removal of 3 intramedullary devices owing to wound infections (10%), but the authors made no mention of further treatment for the remaining 21% of patients. Smekal and colleagues43 reported a rate of 20% for device protrusion, which was treated with shortening of the intramedullary device under local anesthesia.
The study by Witzel45 was assigned level II evidence (Table 2). There were no major or minor complications reported in that series of 35 patients. One retrospective cohort study was assigned level III evidence (Table 3).17 This study reported 1 nonunion, 2 delayed unions and 4 malunions, but the authors did not describe treatment for these major complications. In addition, a 30% radiographic migration rate of the intramedullary fixation devices was reported in this study; most patients, however, remained asymptomatic and did not require any additional treatment.17
Level II evidence study
Level III evidence study
Finally, 1 retrospective case series was assigned level IV evidence (Table 4).46 In this study, Chen and colleagues46 reported a rate of 7% for malunions, which did not require additional surgery. Most of the complications encountered were minor and involved a 20% rate of irritation, which was treated by device shortening under local anesthesia.
Level IV evidence study
Finally, in 4 of the 6 included studies, all of the intramedullary devices were routinely removed according to the standard treatment guidelines in the authors’ practices.17,18,44,45
Discussion
Our systematic review aimed at answering several questions regarding complications after intramedullary fixation of DMCF. What is the incidence of major and minor complications? What interventions are available for resolving major or minor complications? And, finally, what is the value of reported complications in terms of the scientific level of evidence and the quality at which they are presented?
Reported rates for major complications like bone-healing problems and deep infections requiring implant removal were no higher than 7%.17,18,43–46 In addition, only 4 refractures after pin removal were reported in a total of 3 studies.17,18,43 Therefore, overall, the rate of major complications requiring additional surgical treatment was low. Most complications were implant failures, breakages, irritations or implant migrations. Reported rates of minor complications were as high as 31%.
Major complications require an additional surgery (e.g., corrective osteotomy). Minor complications are resolved with antibiotics for wound infections or shortening of the intramedullary device under local anesthesia. Treatments for minor complications are non- or minimally invasive and therefore easy to resolve. Irritation is one of the main effects of migration, telescoping or protrusion. Three interventions seem to resolve most of these irritation-related complications: shortening of the intramedullary device, removal of the device or revision osteosynthesis. Migrating, telescoping or protruding devices may remain asymptomatic without requiring additional treatment, yet most intramedullary fixation techniques require routine surgical device removal once fracture healing has occurred.18,44–46 The vast majority of patients require additional surgical interventions, although these interventions are minor. An option for reducing medial protrusion (and thus irritation) might be the use of medial end caps. Frigg and colleagues47 reported a reduction in medial protrusion rates by using an end cap for TEN.
The level of evidence and quality assessment tools were used to assess studies on methodological quality. Only 6 studies met our inclusion criteria, 3 of which provided the highest possible level of evidence. Therefore, studies on this specific subject with high-level evidence are scarce.
The optimal surgical technique to accomplish intramedullary fixation is anecdotally harder to master than more traditional surgical fixation techniques. Reported operative outcome may be influenced by learning curves of the involved surgeons. Only 1 study included in our review mentioned the involved surgeon’s experience with intramedullary fixation.45 In addition, depending on the intramedullary device used, the degree of open fracture reduction varies. Theoretically, this may negatively affect infection rates due to increased exposure, cosmetic outcome due to an increase in incision length and fracture healing due to disruption of the periosteum and fracture hematoma. For instance, fixation with the Rockwood pin, requires open reduction,44 whereas TEN fixation reportedly allows for closed reduction in 60%–85% of cases.11,35 In addition, the type of intramedullary device may influence the rate of complications. Unfortunately, the current numbers available are too small and study designs too different to permit a thorough analysis.
Discussion of whether more complex fractures should then be treated with intramedullary fixation may arise. The included studies did not answer that question. However, other studies that did not meet our inclusion criteria indicated that intramedullary fixation might also be suitable for fixation of more complex fractures.11,24,32
In 3 of the studies included in our review, the intramedullary devices were routinely removed upon fracture healing.18,44,46 Device removal requires additional surgery with corresponding risks and complications (e.g., infection, refracture). However, it should be noted that these additional surgical procedures only require small incisions and have short durations.17,44
Limitations
The study design carries the risk of certain limitations. Only the PubMed/MEDLINE and EMBASE databases were used for our search. Proceedings of annual meetings were not taken into account; therefore, valuable information might have been lost. However, usually only preliminary results are presented at (annual) meetings and may differ from the final study results.
Owing to the different study designs and characteristics, data could not be pooled and were summarized separately per study. Available studies were only included if they reported on the treatment of isolated clavicle fractures. This strict inclusion criterion was chosen to avoid the possible influence of comorbidities on reported outcomes and complications. It has to be noted that studies reporting on a combination of clavicle fractures and lower extremity or rib pathology35,36 were accordingly excluded, although the impact of the mentioned comorbidities on the outcome of the clavicle fracture treatment may not be severe.
Several different complication groups were defined for analysis of the selected studies. However, there are differences in definitions of complications among the studies (e.g., deep, superficial and wound infections may have been present). Future studies would benefit from improvements in defining complications when determining outcome parameters. Furthermore, our review focused solely on the spectrum of complications of intramedullary fixation; therefore, no comparisons with complications after open reduction and internal plate fixation or nonoperative treatment were made.
Finally, the applicability of the quality assessment tools used should be addressed. With regards to the levels of evidence, a “high” level of evidence referred to the manner in which different treatment options were compared, but not necessarily to the way in which data were collected. In general, prospectively collected data are more reliable than retrospective data. However, when drawing conclusions on the incidence and consequences of adverse events of a single therapeutic intervention, data can be extracted from both case series and comparative studies. It is uncertain that the level of evidence of data from comparative studies is greater than that of data from prospective case series. Therefore, the quality assessment tool was added and used to strengthen the quality assessment of different studies included in our review.
Conclusion
Reported rates of major complications requiring additional surgery (e.g., corrective osteotomy) after intramedullary fixation were low, but implant-related problems that also required additional surgery might present with high prevalence. Owing to (routine) implant removal, treatment with intramedullary fixation often requires an additional surgical procedure.
Footnotes
Contributors: F.-J.G. Wijdicks, P.J. Millett and O.A.J. Van der Meijden designed the study. F.-J.G. Wijdicks and O.A.J. Van der Meijden acquired the data. F.-J.G. Wijdicks, R.M. Houwert, E.J.J.M. Verleisdonk and O.A.J. Van der Meijden analyzed the data. F.-J.G. Wijdicks, R.M. Houwert and O.A.J. Van der Meijden wrote the article. All authors reviewed the article and approved its publication.
Competing interests: None declared for F.-J.G. Wijdicks, R.M Houwert and E.J.J.M. Verleisdonk. P.J. Millett is a consultant and receives payments from Arthrex and owns stock in Game Ready and options in VuMedi. O.A.J. van der Meijden’s research position was supported by Arthrex. This research was supported by The Steadman Philippon Research Institute, which is a 501(c)3 nonprofit institution supported financially by private donations and corporate support from the following entities: Smith & Nephew Endoscopy, Arthrex, Siemens Medical Solutions, Ortho Rehab, Ossur Americas and Conmed Linvatec.
- Accepted January 9, 2012.