Abstract
Rectal adenomas and cancers occur frequently. Small adenomas can be removed colonoscopically, whereas larger polyps are removed via conventional transanal excision. Owing to technical difficulties, adenomas of the mid- and upper rectum require radical resection. Transanal endoscopic microsurgery (TEM) was first designed as an alternative treatment for these lesions. However, since its development TEM has been also used for a variety of rectal lesions, including carcinoids, rectal prolapse and diverticula, early stage carcinomas and palliative resection of rectal cancers. The objective of this review is to describe the current status of TEM in the treatment of rectal lesions. Since the 1980s, TEM has advanced substantially. With low recurrence rates, it is the method of choice for resection of endoscopically unresectable adenomas. Some studies have shown benefits to its use in treating early T1 rectal cancers compared with radical surgery in select patients. However, for more advanced rectal cancers TEM should be considered palliative or experimental. This technique has also been shown to be safe for the treatment of other uncommon rectal tumours, such as carcinoids. Transanal endoscopic microsurgery may allow for new strategies in the treatment of rectal pathology where technical limitations of transanal techniques have limited endoluminal surgical innovations.
Rectal adenomas and cancers are common. In 2007, 6721 new cases of rectal and rectosigmoid cancers were reported in Canada, representing an annual incidence of 20.4 per 100 000.1–3 In the United States, 40 000 new diagnoses of rectal cancer and 51 370 deaths from colon and rectal cancers were estimated for 2014.4 As premalignant lesions, adenomas are thought to transform into adenocarcinoma in 2.5% of patients after 1 year and in 8% after 5 years.5,6 In the lower rectum, small adenomatous polyps can be removed colonoscopically, whereas larger polyps are removed via conventional transanal excision (TAE) using anal retractors and diathermy. Adenomas of the mid- and upper rectum are much more difficult to visualize and remove transanally, thus requiring radical, transabdominal resection in most cases, including anastomosis where possible.
In the early 1980s, Buess and colleagues7 developed a transanal operating proctoscope with modified laparoscopic instruments as an alternative treatment for these lesions using a technique known as transanal endoscopic microsurgery (TEM). Following animal model experiments,7 the group described the use of TEM to remove a rectal adenoma in a human in 1983.8 The method was conceptually designed to facilitate removal of endoscopically unresectable sessile polyps using a minimally invasive technique. After this early success with benign disease, the technique has been subsequently used to remove early stage carcinomas and carcinoids of the rectum, for palliative resection of advanced rectal cancers and to surgically correct rectal prolapse and rectal diverticular disease.9
The objective of this review is to describe the current status of TEM in the treatment of rectal lesions.
Technique
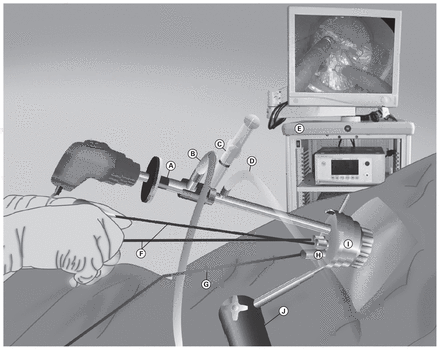
The TEM technique involves 3 main components: an operating proctoscope, a laparoscopic camera and modified laparoscopic instruments. The operating proctoscope is typically 4 cm in diameter and varies from 12 cm to 20 cm in length. The proctoscope maintains an airtight seal at the anus once inserted in the rectum and is held in place by the obligate articulating arm, which fixes the proctoscope to the operating table. The proctoscope has a port for inflow (typically CO2) and outflow, which facilitates smoke evacuation during cauterization. The faceplate on the proctoscope has 4 ports through which a camera and 3 modified laparoscopic instruments facilitate the full-thickness excision of rectal lesions up to 25 cm from the anal verge.10 In conventional laparoscopy, levering of the instruments is the primary strategy for dissection and retraction, but in the confines of the rectum while operating transanally the surgeon is restricted to more rotational movements; thus, the operating ends of the instruments are angulated to improve the operator’s range of motion (Fig. 1).
Transanal endoscopic microsurgery (TEM) operating room set-up. (A) Camera, (B) light source, (C) lens clearing fluid, (D) insufflation tubing, (E) laparoscopy tower, (F) operating instruments, (G) suction, (H) ports, (I) operating anoscope, (J) TEM scope holding arm.
Learning curve
Owing to the technically challenging nature of TEM, limited indications for the procedure and cost of the equipment, the learning curve is steep.10,11 However, in hospitals where surgeons are experienced in minimally invasive operations, the technique is acquired more rapidly.12,13 Decreased complication rates, hospital stay, blood loss and duration of surgery after gaining experience with TEM have been reported in several studies.14–16 Koebrugge and colleagues17 investigated a learning curve effect for TEM in their series of 105 patients from 2002 to 2007. Dividing patients into 2 groups (2002–2004 and 2005–2007), they observed a significant decrease in duration of surgery, hospital stay and postoperative complications in the latter group. These findings are consistent with those of studies that examined the learning curve in laparoscopic colorectal procedures and laparoscopic cholecystectomy.12,18–20
Duration of surgery, length of stay and cost
In centres with more extensive TEM experience, average durations of surgery range from 45 to 113 minutes (Table 1), depending on the size and location of the lesion and the surgeon’s experience.14,17 Also, a number of studies have reported significantly decreased duration of surgery for TEM compared with radical resection, with reductions ranging from 46 to 140 minutes.21–24
Duration of surgery for resection of rectal neoplasms using TEM
Most patients who undergo TEM experience short hospital stay and early return to routine activities, even after resection of very large lesions, often with full thickness excision. Most TEM case series report mean postoperative hospital stays of 0–5 days.25–28 Shorter hospital stay and lower rate of postoperative complications after TEM also contribute to a considerable reduction in operation costs. In an American study by Cocilovo and colleagues,25 the cost-effectiveness of TEM compared with radical resection was analyzed. The average cost per patient (excluding device cost) for TEM was reported as $7775 compared with $34 018 for low anterior resection. These cost savings are mitigated by the relatively high initial cost of the instrumentation. However, in centres performing high volumes of TEM procedures, the per procedure costs are substantially reduced.29
Functional outcomes
Anorectal function after TEM has been addressed in several studies.30–34 As a result of the dilation of the anal canal by the proctoscope and possibly prolonged operations, it has been suggested that damage to the anal sphincter could cause postoperative fecal incontinence.35 Although manometeric studies have shown decreased anal sphincter pressures and compliance after surgery, no remarkable detrimental long-term clinical impact on continence after TEM has been observed.35,36
Kreis and colleagues32 investigated functional outcomes after TEM in 42 patients using manometry and standardized interview scales. They observed decreased anal sphincter resting pressure 3 months and 1 year postoperatively (both p < 0.01). However, the reduced squeezing pressure 3 months after surgery improved at 1 year. Kreis and colleagues suggested that these findings could be explained by the elderly group of patients undergoing TEM or by the direct effect of the tumour itself rather than the surgical technique. Similarly, in a prospective study of 41 patients Cataldo and colleagues33 found no deleterious consequences on fecal continence after TEM. They did not find any significant difference between pre- and postoperative mean Fecal Incontinence Severity Index (FISI) score (2.4 v. 2.4), mean Fecal Incontinence Quality of Life (FIQL) scores (lifestyle: 3.6 v. 3.5; coping: 3.7 v. 3.5, depression: 3.9 v. 3.8, embarrassment 3.6 v. 3.4), number of bowel movements per day (mean 2.4 v. 1.5) and ability to defer defecation. In a recent study of 50 patients Doornebosch and colleagues34 found significantly improved FISI and FIQL scores after TEM (all p < 0.05). Also, patients reported improved quality of life after surgery. Allaix and colleagues37 studied the long-term functional outcomes and quality of life measures after 5 years of follow-up in 93 patients who underwent TEM. The authors assessed manometric and clinical values preoperatively and at 3, 12 and 60 months after surgery. Similar to previous studies, manometric values, including anal resting pressure, rectal sensitivity threshold, maximum tolerated volume and urge to defecate threshold, declined at 3 months but returned to preoperative level at 12 months after surgery. Compared with preoperative levels, there were no significant changes in anal squeeze pressure after surgery. Wexner incontinence scores and general quality of life scores, which were increased in the early postoperative period, returned to preoperative levels at 5 years. They also reported that tumours larger than 4 cm caused a significant decline in rectal sensitivity, urge to defecate thresholds and maximum tolerated volume at 3 months (p = 0.008). Tumor size was the only factor predicting postoperative urgency in this series. The best available evidence suggests that, independently, the TEM procedure seems to have no permanent deleterious effect on fecal continence.
Outcomes and complications
Adenoma
Since its introduction, TEM has been used primarily for resection of large adenomas of the rectum.25,26,38,39 Several studies in recent years have shown TEM to be a safe alternative to TAE in the treatment of rectal adenomas.
Transanal endoscopic microsurgery excision of adenomas results in low adenoma recurrence rates. In the 2893 TEM procedures for adenomas reported in the literature, the reported local recurrence rates vary from 2% to 16% in individual series (Table 2).9,24,27,38,40–52 Several studies have compared local recurrence of adenomas treated by TEM and TAE. De Graaf and colleagues42 compared outcomes in 248 patients with rectal adenomas: 208 treated with TEM and 40 treated with TAE. They resected 216 adenomas in the TEM group and 43 adenomas in the TAE group. On pathology evaluation, patients who underwent TEM were significantly more likely to have negative margins on the adenoma excision than those who underwent TAE (88% v. 50%, p < 0.001). The authors reported recurrence rates of 6.1% and 28.7% after TEM and TAE, respectively (p < 0.001). Similarly, in another study of 78 patients with adenoma treated with TEM (n = 40) or TAE (n = 38), Moore and colleagues52 reported more frequent negative margins (83% v. 61%, p = 0.030) and a lower recurrence rate in the TEM group (3% v. 32%, p = 0.003). The higher rate of negative margins achieved in adenoma resection using TEM seems to account for the lower rate of recurrence associated with this procedure. In a series of 117 patients Speake and colleagues43 used TEM to remove rectal adenomas in 80 patients. No recurrence developed in patients with negative margin resections. However, the authors reported a 10% recurrence for the positive margin specimens (p = 0.001). The importance of achieving negative margins in preventing local recurrence in patients undergoing TEM is highlighted in several studies. McCloud and colleagues48 investigated the predictors of early recurrence in patients with adenomas after TEM. They found significantly different recurrence rates between complete and incomplete excision groups after a median follow up of 31 months (4.3% v. 35.7%, p < 0.001). In subgroup analysis, their 75 patients were divided into 3 groups based on the largest tumour dimension: 0–50 mm (n = 45), 51–100 mm (n = 27) and 101 mm or larger (n = 3). Recurrence increased as tumour size increased (8.9% v. 25.9% v. 33.3%, p = 0.020). The authors surmised that large size of a polyp is likely related to an inability to completely excise with clear margins, thus leading to recurrence. Close monitoring of patients with positive margins or incomplete excision through endoscopic follow-up is highly recommended.
Local recurrence in patients undergoing TEM for resection of rectal adenoma
Complications after TEM for adenoma have been evaluated in several studies. Guerrieri and colleagues53 evaluated 588 patients with rectal adenoma at 6 centres in Italy. Overall, 8.1% of these patients experienced minor complications (e.g., suture site leakage, soiling and minor postoperative bleeding) and 1.2% experienced major complications (e.g., rectal hemorrhage requiring intervention, rectovaginal fistula requiring ileostomy, rectovesical fistula, suture site leakage treated with second TEM) within 30 days of surgery. Said and Stippel38 studied 280 patients with rectal adenoma, 3.4% of whom had postoperative complications including perforation, bleeding and fistula. They reported 1 (0.3%) postoperative death due to a recurrent thromboembolic event. In another study, Whitehouse and colleagues50 performed 146 TEMs in 143 patients. Six (4.1%) patients had postoperative bleeding and received transfusions. Two of them had repeat TEM to stop the bleeding. The authors reported 1 procedure-related death due to intraperitoneal perforation and leakage. They also found nonsignificant radiologic leaks in 3 patients (2%) who were managed nonoperatively.
In summary, for adenomas TEM has shown excellent results, low recurrence rates and a remarkably favourable complication profile compared with TAE54–56 and radical resection.23,57 The evidence supports TEM as the preferred approach to rectal adenoma resection when colonoscopic removal is not possible.
Adenocarcinoma
Early rectal adenocarcinomas
Transanal excision for early rectal cancer is controversial. Recent data from well-designed observational studies have suggested high local recurrence rates and worse survival in patients treated with TAE compared to those treated with radical resection (Table 3).58–62 Bentrem and colleagues60 completed a study of 319 patients who were treated for T1 rectal adenocarcinomas by either TAE (n = 151) or radical resection (n = 168) over a 17-year period. The authors compared local and distant recurrence, disease-free survival and overall survival between the 2 groups. Survival analysis was based on a follow-up period of 51 months. Their results indicated significantly lower 5-year local and overall recurrence rates in the radical resection group. Nonetheless, they observed similar outcomes in terms of disease-free and overall survival between the 2 procedures. While these data are concerning, it is possible that high local recurrence rates associated with TAE may be partially related to the technical challenges of the procedure. Comparative studies of TEM and TAE for early rectal cancers demonstrated fewer resections with positive margins and lower local recurrence rates with TEM. In a study of patients with early rectal cancers, Christoforidis and colleagues63 found a lower local recurrence rate in patients undergoing TEM than in those who had TAE, although the difference was not significant (12% v. 22%, p = 0.37). They also found better tumour resection in patients who underwent TEM, with fewer positive margins in these patients than in those who underwent TAE (37% v. 19%, p = 0.001). Langer and colleagues23 also confirmed that TEM is superior to TAE with respect to resection margin results, with higher R1 and Rx resections in patients who underwent TAE (37% and 16% v. 19% and 5%, respectively, p = 0.001). Furthermore, conventional TAE is usually limited to resection of the rectal wall, whereas TEM facilitates excision of some mesorectal, fat and perirectal lymph nodes, which can be retrieved and sampled with the specimen.
Studies comparing transanal excision versus radical resection in treatment of rectal cancers
For rectal cancer, the gold standard therapy is radical resection. In the Dutch total mesorectal excision (TME) trial,64 patients with T1 cancer treated with TME alone had 1.7% local recurrence at 2 year follow-up. Use of TEM in patients with early rectal adenocarcinomas is usually limited to patients with low-risk T1 rectal adenocarcinomas that are small (< 4 cm); well differentiated; have no lymphatic, vascular or perineural involvement; and are located within 15 cm of the anal verge. Early rectal cancer is defined as invasive adenocarcinoma confined to the submucosal layer. Once the cancer spreads beyond the mucosal layer, it is possible for it to metastasize to local lymph nodes or even distant organs. Kudo65 proposed a submucosal (sm) classification (sm1 refers to infiltration into the upper third, sm2 into the middle third and sm3 into the lower third of the submucosal layer) that describes the level of penetration of the tumour into the submucosa. It has been used previously to predict lymph node metastasis and to select the proper treatment approach in patients with early rectal cancer.66,67 Studies report that the risk of lymph node involvement correlates with the depth of tumour invasion into the submucosa.68–70 Kikuchi and colleagues66 reported that the overall incidence of lymph node metastasis was 5% in patients with sm2 and 25% in those with sm3 tumours. However, they found that none of their patients with sm1 and pedunculated sm2 tumours had lymph node metastasis. For pedunculated polyps, sm1 classification is similar to Haggit level 1 classification, and sm2 includes Haggit levels 2 and 3. As described by Haggit and colleagues,71 level 4, which refers to the spread of cancer cells above the muscularis propria, is the most important risk factor for lymph node metastasis in patients with such tumours. This level of invasion is similar to Kudo’s sm3 classification. Kikuchi and colleagues66 found no sm3 level of invasion in their patients with pedunculated polyps. Therefore, they suggested that since lymph node metastasis occurs rarely in patients with sm1 and pedunculated sm2 submucosal rectal cancers, these tumours could be removed by local excision. However, if margins are positive, radical options are more favourable in this group of patients. In cases of greater submucosal invasion (sm3), an sm2 sessile tumour or when vessels are involved, radical resection or adjuvant therapy after local resection should be considered. On the other hand, more recently, in a retrospective study, Choi and colleagues67 reported a higher incidence of lymph node metastasis in patients with sm2 and sm3 rectal cancers (21.3% and 38.5%, respectively). They also reported that 4 patients with sm1 invasion showed evidence of lymph node metastasis (4.2%). The authors suggest that even in the case of sm1 invasion, local resection should be reserved for meticulously selected patients. In any event, precise preoperative staging is critical in the selection of appropriate patients to optimize results.72
With the advancement of magnetic resonance imaging (MRI), the accuracy in staging rectal cancer has increased significantly. Studies report MRI accuracy of 65%–100% in the staging of rectal cancer.73–78 This variability might cause difficulty, especially when staging borderline tumours. The role of MRI in the staging of early rectal cancer is not yet evident owing to its limited ability to distinguish between mucosal and submucosal layers of the rectal wall.74 However, MRI has been shown to be effective in the evaluation of mesorectal fat and fascia, which are important features in treatment planning, especially for patients with more advanced cancers.79 Endorectal ultrasonography (ERUS) is usually used to assess T stage and has been reported to have an accuracy of up to 94%.80,81 However, ERUS is not reliable in assessing lymph node positivity.82–85 Nonetheless, the combination of digital rectal examination, ERUS and flexible sigmoidoscopy with biopsy is the recommended preoperative staging and tumour assessment strategy when considering TEM. In otherwise appropriate candidates who have an ERUS suggestive of T2 or T3 disease but in whom there is clinical suspicion of early cancer, TEM can still be used to confirm T stage in patients who are reluctant to undergo radical resection; early salvage surgery can still be performed postoperatively if advanced rectal cancer is identified.86 Moreover, there are a number of reports that immediate reoperation after local excision of rectal carcinomas demonstrates oncologic results comparable to primary radical resection.62,87–89
Several groups have reported their experience with TEM in patients with early rectal cancers (Table 4).38,39,44,45,86,90–99 Baartrup and colleagues86 retrospectively studied 72 patients with pT1 cancers. They reported a recurrence rate of 13% (9 of 72), 5-year cancer-specific mortality of 6% (4 of 72) and 5-year total survival of 76%. Allaix and colleagues91 studied 300 patients with rectal neoplasms treated with TEM between 1993 and 2007. Three of 38 patients with pT1 cancers received postoperative radiotherapy. The authors observed no recurrence; the overall and disease-free survival rates were 100% at a mean follow-up of 60 months in this group of highly selected patients.
Transanal endoscopic microsurgery outcomes in rectal adenocarcinoma
Four studies have compared oncologic results of TEM and radical surgery exclusively in patients with T1 rectal cancers (Table 5).22,24,100,101 Only 1 was a prospective randomized study. In this study, Winde and colleagues22 found no difference in oncologic outcomes between the treatment groups. Similarly the other 3 studies reported no difference in 5-year cancer-free and overall survival rates. De Graaf and colleagues24 reported a significantly lower local recurrence rate in the radical resection group (24% in the TEM group v. 0% in the TME group, p < 0.001). Positive margin rates were shown to be higher with TEM than radical resection by Palma and colleagues100 and Heintz and colleagues;101 however, they observed similar local recurrence rates in both the TEM and radical resection groups. All of these studies suggest that patients treated with TEM had significantly shorter duration of surgery, less blood loss, shorter hospital stay, lower analgesic demand and lower morbidity.
Studies comparing TEM and radical resection in the treatment of early rectal cancers (T1)
As such, the available evidence suggests that TEM alone seems to be a reasonable alternative to radical resection in patients with low-risk T1 adenocarcinomas. However, further therapeutic steps (salvage surgery or chemoradiation [CRT]) should be considered in patients with positive margins, as suggested in the literature. Furthermore, scrutinous preoperative patient selection and disclosure of the limited evidence to the patient is critical.
Advanced rectal adenocarcinomas
The limited role of TEM in the treatment of more locally advanced tumours is more defined. Lymph node involvement ranges from 12% to 28% in patients with T2 tumours and from 36% to 66% in those with T3 tumours.62,102 High rates of local recurrence for T2 (up to 50%) and T3 tumours (up to 100%) with conventional local excision alone have been reported.102–109
Transanal endoscopic microsurgery alone is not a reasonable treatment for fit patients who have rectal cancer that is local stage T2 or deeper. There is recent data to guide the use of TEM in patients with T2 and T3 rectal cancers. Borschitz and colleagues110 found reduced local recurrence in patients with T2 rectal carcinomas who underwent immediate reoperation after TEM as opposed to those who had TEM alone (12% v. 35%, p value not reported). Similarly Tsai and colleagues44 reported their results of TEM resection of rectal cancer in a retrospective series of 269 patients (111 patients with adenocarcinomas: 58 with T1, 26 with T2 and 11 with T3 tumours). Patients with less than 6 months of follow-up, those who had previous resection, patients with metastases at the time of presentation and those who underwent radical surgery after TEM were excluded from the recurrence analysis. The mean follow-up was 42.8 (range 9–116) months for patients with T2 and 44.7 (range 8–73) months for those with T3 tumours. The authors reported a local recurrence rate of 23.5% (4 of 17) and 100% (4 of 4) for patients with pT2 and pT3 adenocarcinomas, respectively. Two of 4 patients with recurrent T2 tumours underwent abdominoperineal resection (APR) with curative intent (both patients were alive at the last follow-up: 1 was disease-free 11 mo after recurrence and 1 was alive with disease 47 mo after recurrence). Of the remaining 2 patients with T2 tumours, 1 had repeat TEM (this patient was alive and disease-free after 27 mo) and the other was awaiting low anterior resection (LAR) after CRT (this patient was alive with disease 3 mo after recurrence). The authors reported 1 (20%) recurrence in 5 patients with T2 tumours who had adjuvant therapy after TEM. The remaining 4 patients were alive and disease-free at the time of last follow-up. All 4 patients with recurrent T3 tumours died of cancer: 1 had LAR for recurrence, but the other 3 refused further surgery after recurrence (mean follow-up after recurrence was 4.75 mo). Thus, evidence suggests that in patients with more invasive rectal carcinomas TEM cannot be done with curative intent and seems to be appropriate only in patients who refuse radical resection or in elderly patients in whom the medical risks prohibit surgery.
A combination of local excision and adjuvant therapy for high-risk patients with T1, T2 and more advanced tumours, especially when patients are not fit enough to undergo radical surgery, has been investigated in several studies. Duek and colleagues111 reported the outcomes of TEM in 21 patients with T2 rectal adenocarcinomas with a median follow-up of 3 years. They included patients with T2 tumours who were not fit for surgery or refused to undergo radical resection and 1 patient whose cancer was staged as T1 preoperatively but who was restaged as T2 based on the postoperative pathology report. Of the 16 patients who had clear margins, 12 received adjuvant radiotherapy; all of them were disease-free after a median follow-up of 3 years. Of the remaining 4 patients who refused radiotherapy, 2 experienced a recurrence at a mean of 12 months; 1 died at 10 months and 1 had liver metastasis at 15 months. Five patients who had positive margins underwent salvage surgery (2 APR, 3 LAR); 3 of them were disease-free at the end of the follow-up period, 1 had lymph node metastasis after LAR and died 18 months later, and 1 was disease-free at 2 years of follow-up. In a similar study, Ramirez and colleagues112 studied 88 patients who underwent TEM: group A included low-risk patients with pT1 tumours (n = 54), group B included high-risk patients with pT1 tumours (n = 6) and low-risk patients with pT2 tumours (n = 24), and group C included patients with T2 tumours (n = who were staged pT3 and a patient with pT2 disease with positive margins (R1; n = 1). Group A had no further surgery after TEM. Group B received adjuvant radiotherapy. Two patients from group B decided to have immediate radical surgery after TEM. Group C underwent salvage surgery immediately after TEM (1 LAR and 3 APRs). Survival study was done for groups A and B. After a mean follow-up of 71 (36–128) months, the local recurrence rate in group A was 7.5%, with 5-year cancer-specific survival of 98%. Local recurrence in group B (10.7%) was higher than previously reported for radical resection of T2 tumours, but the 5-year cancer-specific survival rate (93%) was comparable to that for radical resection.
Transanal endoscopic microsurgery after neoadjuvant therapy for downstaging of advanced tumours is being investigated and has demonstrated promising results.21,96,113 Lezoche and colleagues21 randomly assigned 70 patients with T2N0 rectal cancer to TEM and laparoscopic resection (LR) procedures after CRT. Patients were restaged after neoadjuvant therapy. Those in the TEM group had significantly better results in terms of hospital stay, blood loss and duration of surgery than those in the LR group, although there was no difference in complication rates between the groups. Oncologic results after TEM and LR were comparable in terms of local (5.7% v. 2.8) and distant (both 2.8%) recurrence rates (combined local and distant recurrences 9% v. 6%) and probability of disease-free survival (both 94%). However, lower morbidity, shorter hospital stay and faster return to normal activities in the TEM group may suggest that this technique is favourable for selected patients with T2 disease without nodal involvement or distant metastasis, though more evidence is required. Guerrieri and colleagues114 studied 137 patients with rectal adenocarcinoma treated with TEM. Fifty-four patients with T2 and 46 with T3 disease underwent preoperative radiotherapy. Twenty-eight patients with T1 disease who had favourable differentiation did not receive preoperative radiation. Likewise, 9 patients with T2 and T3 disease did not receive preoperative radiotherapy owing to bleeding, treatment refusal and pre-operative diagnosis of adenoma. Among those who received pre-TEM radiation, 2 patients with T2 (3.7%) and 2 patients with T3 disease (4.3%) had local recurrence at a mean of 46 (range 6–115) months follow-up. This is compared with 3 local recurrences (33%) in patients who did not have radiation preoperatively. Eighteen patients preoperatively staged as having T2 (n = 12) and T3 (n = 6) disease who received radiotherapy before the operation had no residual malignancy (pT0) in the final pathology assessment.
In a recent prospective, multi-institutional phase II trial, Garcia-Aguilar and colleagues115 reported the preliminary results of the American College of Surgeons Oncology Group (ACOSOG) in patients with T2N0 rectal cancer. Seventy-seven patients, completed neoadjuvant CRT followed by local excision within 4–8 weeks. Following CRT 56% of the patients showed a complete clinical response defined as “complete disappearance of tumour on proctoscopic exam.” A positive resection margin was observed in 1 patient (1%), and 1 patient with pT3 disease who underwent TME following LE was shown to have lymph node metastasis after CRT. In 49 patients (64%) tumours were down-staged to pT0–1. Pathologic complete response was observed in 32 patients (44%) and complete clinical response reached a sensitivity of 85% and specificity of 67% in prediction of pathologic complete response in this study. Perioperative complications occurred in 45 (58%) patients, with rectal pain being the most common. Overall this study demonstrates the effectiveness of CRT in the nonradical approach to rectal cancer in selected patients; however, further adjustments need to be made to CRT protocols to reduce the relatively high rate of adverse effects associated with CRT.
Carcinoids
Mentges and colleagues116 were the first to report the use of TEM in the resection of rectal carcinoid tumours. Subsequently, several authors presented their experience with TEM and carcinoids.9,117–121 Carcinoids eligible for local excision are typically smaller and less technically challenging than rectal adenocarcinomas.118 Kinoshita and colleagues117 reported their results in 27 patients with carcinoid tumours treated with TEM. Fourteen patients had TEM for primary excision, and the remaining 13 underwent TEM for excision of prior incomplete endoscopic resection of rectal carcinoids. Duration of surgery, blood loss and histopathologic results did not differ between these 2 groups. The authors reported no positive margins in their specimens, and there was no local recurrence or carcinoid-specific mortality after a follow up period of 70.6 months. Araki and colleagues118 reported on 12 patients with rectal carcinoid treated by TEM. Ten of these patients’ submucosal tumours were less than 10 mm in size; the pathology demonstrated negative resection margins, and there were no local recurrences. Two patients in whom pathology showed lymphatic involvement were treated with subsequent LAR. Ishikawa and colleagues119 compared the results of carcinoid removal between conventional excision (n = 11) and TEM (n = 17). After a mean follow-up of 23.8 (range 6–49) months for the TAE group and 47.1 (range 12–96) months for the TEM group, the authors reported no local or distant recurrence.
Despite limited data in the literature, it appears that TEM is a safe and effective way to remove small rectal carcinoids either primarily or after incomplete endoscopic removal. However, further study is still needed.
Role of TEM in other diseases
The application of TEM for a variety of other rectal lesions has been described in case series and case reports. Serra Aracil and colleagues122 and Zoller and colleagues123 reported 4 cases of retrorectal cysts excised using TEM. They reported no significant complications and complete excision of the cysts. Other reported, albeit rare, indications for TEM include excision of gastrointestinal stromal tumours,91,124 repair of colorectal anastomotic strictures125–127 and rectovaginal or rectourethral fistula closure.128,129
Future directions of TEM
Use of TEM as a portal for natural orifice transluminal surgery (NOTES) has been discussed widely.130–132 However, most of the studies in this field are still in preclinical and experimental phases. One of the main concerns about the transanal or transcolonic access is the risk of fecal spillage into the abdominal cavity. Extensive experience from TEM studies suggests that full-thickness excision of rectal tumours and peritoneal entry is not associated with postoperative complications.133,134 Theoretically, TEM should be a safe platform for a transanal NOTES procedure.
A number of feasibility studies have shown successful completion of transrectal NOTES on porcine and human cadavers using TEM.135,136 Nonetheless, as TEM instruments were developed for intraluminal resection, technical difficulties, such as inadequate length of instruments to reach the mid-and upper abdominal structures, inability to effectively mobilize intra-abdominal organs and safely fashion anastomoses, are the barriers to clinical implementation of transanal NOTES procedures using TEM. To address these technical issues, in an experimental bovine model Bhattacharjee and colleagues137 demonstrated a redesigned TEM instrument that is longer and more manoeuvrable. Using the modified instruments, they achieved easier dissection of mesenteric vasculature and colonic mobilization.
The first clinical human studies were performed as hybrid laparoscopically assisted TEM NOTES procedures.138,139 These studies have been shown to be promising in terms of ease of access, feasibility of the technique, postoperative course, lymph node retrieval and achieving clear margins. However, it is crucial to follow up the long-term outcomes to better evaluate the safety and efficacy of this approach in comparison to open or laparoscopic colorectal procedures.
Overall, TEM provides an ideal access portal for NOTES. It offers a stable platform, capability to safely close the enterotomy and maintenance of adequate pneumoperitoneum. Further modification of the instruments, however, is required to overcome its technical challenges.
Conclusion
Transanal endoscopic microsurgery is a technological advance that has been refined and improved significantly since its introduction in the mid-1980s. For rectal adenomas, TEM is the preferred method of removing lesions that are not amenable to colonoscopic excision and has resulted in low recurrence and complication rates. In patients with early (T1) rectal cancers, the use of TEM is controversial. However, small studies comparing TEM to radical resection suggest that there is a role for TEM in select patients. For those with T2 and T3 lesions, TEM should be considered palliative (in patients with prohibitive medical risks for radical resection) or experimental. Patients with T2 or deeper lesions should be treated only with TEM in the context of a clinical trial if they are fit and willing to have radical resection. For the treatment of other uncommon rectal tumours, such as carcinoids, TEM should be used where previously conventional transanal excision was the preferred treatment strategy. Finally, TEM may allow for new strategies in the treatment of rectal pathology where technical limitations of transanal techniques have limited endoluminal surgical innovations.
Acknowledgements
We thank Mr. Bashir Heidary for his valuable efforts in the creation of Figure 1.
Footnotes
Competing interests: None declared.
Contributors: B. Heidary, T.P. Phang, M. Raval and C.J. Brown designed the study. C.J. Brown acquired the data, which B. Heidary, M. Raval and C.J. Brown analyzed. All authors wrote and reviewed the article and approved the final version for publication.
- Accepted April 26, 2013.