Abstract
Background: As populations age, more elderly patients will undergo surgery. Frailty and complications are considered to increase in-hospital cost in older adults, but little is known on costs following discharge, particularly those borne by the patient. We examined risk factors for increased cost and the type of costs accrued following discharge in elderly surgical patients.
Methods: Acute abdominal surgery patients aged 65 years and older were prospectively enrolled. We assessed baseline clinical characteristics, including Clinical Frailty Scale (CFS) scores. We calculated 6-month cost (in Canadian dollars) from patient-reported use following discharge according to the validated Health Resource Utilization Inventory. Primary outcomes were 6-month overall cost and cost for health care services, medical products and lost productive hours. Outcomes were log-transformed and assessed in multivariable generalized linear and zero-inflated negative binomial regressions and can be interpreted as adjusted ratios (AR). Complications were assessed according to Clavien–Dindo classification.
Results: We included 150 patients (mean age 75.5 ± 7.6 yr; 54.1% men) in our analysis; 10.8% had major and 43.2% had minor complications postoperatively. The median 6-month overall cost was $496 (interquartile range $140–$1948). Disaggregated by cost type, frailty independently predicted increasing costs of health care services (AR 1.76, 95% confidence interval [CI] 1.43–2.18, p < 0.001) and medical products (AR 1.61, 95% CI 1.15–2.25, p = 0.005), but decreasing costs in lost productive hours (AR 0.39, p = 0.002). Complications did not predict increased cost.
Conclusion: Frail patients accrued higher health care services and product costs, but lower costs from lost productive hours. Interventions in elderly surgical patients should consider patient-borne cost in older adults and lost productivity in less frail patients.
Trial registration NCT02233153 (clinicaltrials.gov).
In the developed world, aging populations are increasingly in need of surgical interventions. Currently, 16% of Canadians are older than 65 years.1 By 2050, 22% of all North Americans will be considered elderly.2 Health care spending represented 17% of the American gross domestic product (GDP) and 10% of Canadian GDP in 2014.3,4 Costs are expected to increase as the population ages. Improved medical technology, experience and more aggressive surgical treatment criteria have allowed a greater number of elderly and more frail patients to become surgical candidates.5,6
Patients older than 60 years accounted for more than 58% of Canadian surgical bed utilization in 2005,7 and this number will only increase as our population ages. Older patients are at highest risk of postoperative complications, prolonged hospital admissions and increased dependency or institutionalization.8,9 Alongside increased system cost, patients may also require support in the home, home modifications, outpatient medications and caregiver support. These services may represent a substantial out-of-pocket burden for patients and their families.
Reducing postdischarge costs requires identification of high-cost patients and potentially modifiable risk factors. However, most cost prediction models do not effectively predict cost after surgery. Of the 6 preoperative risk stratification tools used in cardiac patients, none reliably predicted costs after surgery;10 all models relied on age, and none incorporated frailty. Additionally, none of the 6 tools assessed differences in type of cost following discharge.
Frailty, defined as a poor physiologic reserve limiting response to acute physiologic insult, and postoperative complications have predicted increased postoperative morbidity and mortality,5,6,11–15 in-hospital costs, costs following discharge15–17 and outpatient medical costs over 6 months.18 However, using the full economic impact following surgery to develop models may have better predictive strength. Only 1 study has incorporated frailty as a predictor for cost after surgical discharge in elderly patients.15 To our knowledge, no studies have assessed cost in older patients receiving emergent surgery or the type of costs following discharge in this population, including patient-borne costs.
This present study aimed to identify independent predictors of overall cost and types of costs accrued by older patients within 6 months of discharge after acute abdominal surgery. Overall costs included costs for health care services utilization, medical products and productive hours lost. This information will be useful to physicians as they consider the financial burden experienced by their patients and attributes that may be associated with higher cost.
Methods
Population and baseline data collection
Our cohort includes patients who were enrolled during the preimplementation period of a controlled before-and-after care transformation, the Elder-Friendly Approaches to the Surgical Environment (EASE) study19 (clinicaltrials.gov identifier: NCT02233153). Patients were prospectively recruited at 2 tertiary referral teaching hospitals in Alberta, Canada, with 1450 inpatient beds combined and more than 1 million unique patient visits per year (University of Alberta Hospital and Foothills Medical Centre). Patients were enrolled during index admissions between January 2014 and September 2015 if they required emergency abdominal surgery and were aged 65 years or older. Exclusion criteria were elective, trauma or palliative surgery, transfers from out of jurisdiction or other hospital services, and preoperative dependence in 3 or more activities of daily living. The heath research ethics boards at each site approved our study procedures (Pro00047180). All study participants provided informed consent.
We collected demographic and clinical characteristics by performing detailed chart reviews or conducting follow-up interviews. We assessed frailty before admission, as defined by the revised Canadian Study of Health and Aging Clinical Frailty Scale20 (CFS; scores ranging from 1 [very fit] to 6 [moderately frail], as severely frail or terminally ill patients [CFS scores of 7–9] were ineligible for this study). For each patient, we calculated the Charlson Comorbidity Index21 and the Clavien–Dindo classification of surgical complications22 (grades III–V refer to major complications and grades I–II refer to minor complications). Complications that occurred postoperatively were included, but those that occurred on postoperative day 0 that were related to the admission diagnosis were excluded. Surgical complications were independently and blindly assessed by 2 clinicians (G.E. and R.K.), with disagreement resolved by consensus. All-cause readmissions within 6 months of discharge were also sought and collected from the provincial electronic medical database.
Outcomes
The primary outcomes were overall cost and costs for health care services, medical products and lost productive hours in the 6 months following discharge after acute abdominal surgery. Costs were calculated based on a modified version of the self-reported Health Resource Utilization Inventory (HRUI) that has previously been validated with administrative data.23 Overall, very few resource utilization surveys are available that have been validated exclusively among elderly patients,24 and none are relevant to the present study. The HRUI includes patient-reported utilization of health care services (e.g., readmission, emergency department visits and any interactions with allied health care providers, including physicians, nurses, physical therapists and acupuncturists), medical products used or purchased (e.g., walkers, ostomy supplies, diapers, wheelchairs) and productive hours (i.e., lost wages in paid employment or volunteering) within 6 months after discharge. Eyeglasses, dentures and hearing aids were considered unrelated to surgery; the costs for these were excluded. The cost for medical products and lost productive hours were considered patient-borne costs. Prescription medications for pain or sedation were additionally sought, as these are commonly prescribed upon discharge after surgery, and were considered health care services. We did not assess the cost of inpatient rehabilitation programs or outpatient laboratory tests; these are covered by the single-payer public insurance program in the jurisdiction of the participating sites.
The HRUI was administered by telephone 6 months after discharge. Cost of health care services, including medication, were calculated from reimbursement schemes by the Alberta Aids to Daily Living (AADL) program25 or using market rates when required. Cost of physician or dental visits were based on published fee schedules26–28 and allied health costs were based on local market rates. Costs for medical products were calculated from government25 and commercial sources. Total productive hours lost in paid employment or volunteer positions were multiplied by the mean hourly wage ($CAD29.2729) in Alberta.30 All costs are reported in Canadian dollars ($USD1 = $CAD1.32) and correspond to January 2016 reimbursement or market rates.
Statistical analysis
We performed descriptive analyses of demographic and clinical data, including χ2, Fisher exact and t tests. Study sites were compared and pooled for analysis. Clinical characteristics and cost categories were assessed for statistical significance in univariate analyses (Kruskal–Wallis tests for ordinal data, Fisher exact or χ2 tests for categorical data, and t tests for continuous variables). As cost data were skewed, outcomes were log-transformed. Data distribution was determined by visual inspection. Overall cost, medical product cost and health care services cost were analyzed using a general linear regression model (GLM).31 Gaussian distribution was used for overall cost and medical product cost; γ distribution was used for health care services cost in the GLM model. Cost for lost productive hours was analyzed using zero-inflated negative binomial (ZINB) regression,32–34 which generates 2 separate models and then combines them. A logit model is generated to assess which patients were a “certain zero” (i.e., where there was no chance an individual could experience any cost because they weren’t working or volunteering before admission). Then, a negative binomial model is used to predict the adjusted cost for patients who are not certain zeros (i.e., patients who are not predicted “certain zeros” are included in the nested negative binomial model). Finally, the 2 models are combined. The ZINB model was compared with a traditional negative binomial model using the Vuong test. The GLM and ZINB models report b-coefficients for log count of cost. Variables were sequentially added to models and kept if p < 0.20. Age and site was forced into each model. To ease interpretation, b-coefficients were inversely transformed and can be interpreted as adjusted ratios (AR).
In sensitivity analyses, we considered 6-month readmission in models for medical products and productivity costs, but not elsewhere, as readmission forms part of the dependent variable (health care services utilization). We assessed model fit using the Bayesian information criterion (BIC), which penalizes for additional variables. Lower BIC indicates a more plausible model given the data. Outliers were retained within the models. Health care costs are driven by outliers that represent a disproportionate percentage of overall expenditure. Removing outliers would result in excluding patients who use a sizable portion of the health care budget. Analyses were performed using STATA software version 14 (StataCorp LP). We considered retuls to be significant at p < 0.05, 2-tailed.
Results
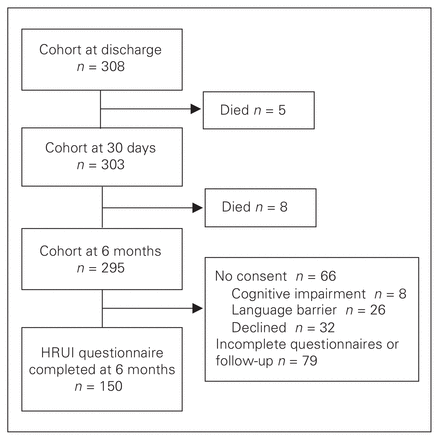
Of the eligible participants (n = 308), 66 were unable or unwilling to participate; 242 patients enrolled. Thirteen patients died within 6 months of enrolment, and 79 were lost to follow-up (Fig. 1). Overall, 65.5% of enrolled participants who were alive at 6 months (n = 150) completed 6-month assessments (Fig. 1). The median age of patients was of 73.7 (range 65–96.5) years, and 54.1% were men. Nearly all patients (93.9%) were living independently before admission and had a median CFS score of 3 (range 1–6); 10.8% had major and 48% had minor complications postoperatively (Table 1). When comparing those who completed the survey and those who were lost to follow-up, there was no difference in age, BMI, sex, marital status, ostomy creation, Charlson Comorbidity Index score, American Society of Anesthesiologists (ASA) classification, preadmission dementia diagnosis, postoperative complications or number of readmissions. Among those who were lost to follow-up, frailty was greater (p < 0.001), length of stay was longer (p = 0.014), and there were more visible minorities (p < 0.001).
Flow of patients through the study. HRUI = Health Resource Utilization Inventory.
Baseline demographic and clinical characteristics of study participants (n = 150)
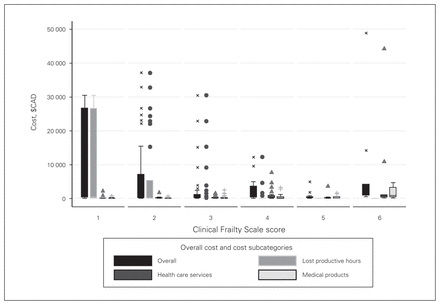
The mean 6-month overall cost was $3921 ± $8582 (max: $48 893) and the median was $496 (interquartile range [IQR] $140–$1948; Table 2). After log transformation there was no skewness (p = 0.13). Stratified by frailty, patients deemed to be well (CFS = 2), to be managing well (CFS = 3) or to be mildly frail (CFS = 5) had the lowest 6-month overall costs (Table 2). In multivariable analysis, increasing age predicted slightly decreasing overall cost (AR = 0.96, p = 0.047), whereas being admitted to the University of Alberta Hospital predicted a 2-fold increase in overall costs (AR 2.14, p = 0.024) within 6 months of discharge, after controlling for postoperative level of care, frailty, ASA class and comorbidities (Table 3).
System and patient-borne costs within 6 months of discharge, according to frailty (n = 150)*
Total and subgroup costs at 6 months after discharge
In general, health care services accounted for the bulk of postdischarge costs ($138, IQR $65–$332). Log transformation resulted in persistent skewness (p = 0.003). Stratified by frailty, costs for health care services were greatest among the moderately frail group (CFS = 6; Table 2). In multivariable analysis, a 1-category increase in frailty independently predicted a 76% increase in health care services costs (AR 1.76, 95% confidence interval [CI] 1.43–2.18, p < 0.001) within 6 months of discharge (Table 3).
Most patients did not accrue costs for medical products within 6 months (Table 2). Log-transformed data did not contain skewness (p = 0.07). Stratified by frailty, the cost of medical products was highest among moderately frail patients (CFS = 6; Table 2). In multivariable analysis, increases in frailty independently predicted a 61% increase in cost for medical products (AR 1.61, 95% CI 1.15–2.25, p = 0.006); marital status, age, ostomy creation or modification, length of stay and site also predicted increased cost (Table 3).
Lost productive hours were analyzed using a ZINB model. The ZINB model fit our data considerably better than a negative binomial model (p = 0.002, Vuong test) and the log-transformed data were not skewed (p = 0.36) after removing zeros. Most patients (115 of 150) also did not accrue costs for lost productive hours within 6 months (Table 2). Stratified by frailty, lost productivity was predominantly observed in very fit (CFS = 1) or well (CFS = 2) patients, but was also seen in patients managing well (CFS = 3) and in vulnerable patients (CFS = 4; Table 2 and Fig. 2). In the ZINB analysis, a 1-category increase in frailty independently predicted a 2-fold increase in probability that the patient was not working or volunteering before admission (AR 2.13, 95% CI 1.38–3.30, p = 0.001; Table 3) and predicted decreased cost for lost productive hours for those who were working or volunteering before admission (AR 0.39, 95% CI 0.21–0.71, p = 0.002; Table 3). Male sex also independently predicted a more than 2-fold increase in lost productive hours (AR 2.28, 95% CI 1.05–4.99, p = 0.042; Table 3) within 6 months of discharge. Age was associated with more often reporting no lost productive hours (AR 1.11, 95% CI 1.03–1.19, p = 0.004; Table 3).
Overall and subcategory costs according to frailty. Boxes represent the interquartile range (IQR), while whiskers define 1.5 times the IQR. Outliers are indicated by symbols.
Sensitivity analyses
On additional assessment, all-cause 6-month readmission independently predicted increased cost for medical products but not for lost productive hours. Inclusion in the model improved fit (Table 4). Notably, univariate logistic regression of major complications did not identify significant interactions with overall cost or cost associated with lost productive hours. It did identify increased costs for medical products (AR 1.09, p = 0.002) and health care services (AR 3.51, p = 0.040); however, these results were not significant in multivariable analysis. Minor complications did not predict a change in any category. The cost of lost productive hours was initially assessed using a multivariable GLM; frailty (AR 1.50, p < 0.001) and age (AR 2.47, p = 0.009) were significant. However, the fit of the GLM was much worse than that of the ZINB model (BIC = 1006 v. BIC = 1). The ZINB model is also specifically designed to account for data sets with high numbers of zeros and consequently was chosen as the superior model.
Estimated total and subgroup cost 6 months following discharge with readmission
Owing to the high number of patients with no cost in the medical product cost category, we also performed a ZINB analysis. We felt that GLM regression was a more appropriate analysis of this sort of cost data (all patients could experience this cost), but we wanted to test the robustness of our findings. Increasing frailty remained a significant predictor of cost in those who experienced cost (AR 1.48, 95% CI 1.24–1.77, p < 0.001). Ostomy creation also predicted higher cost (AR 1.95, 95% CI 1.05–3.63, p = 0.035); increasing age (AR 0.97, 95% CI 0.94–0.999, p = 0.042) and male sex (AR 0.62, 95% CI 0.38–0.999, p = 0.05) predicted lower cost. Increasing frailty (AR 0.60, 95% CI 0.43–0.84, p = 0.003) and age (AR 0.92, 95% CI 0.87–0.97, p = 0.002) also predicted a lower probability of experiencing no cost. Overall, the ZINB model fit much worse than the GLM model (BIC = 1268 v. BIC = 110). Length of stay and marital status were not robust, but the effect of frailty and ostomy creation remained large and statistically significant.
When minor or major complications were included in the multivariable GLM regression and ZINB models there was no significant interaction. Major complications were retained only in health care services and remained nonsignificant.
Discussion
Elderly surgical patients incur both system and patient-borne costs after discharge. Health care services costs account for the majority of postdischarge costs experienced by patients in our study, as most patients did not experience costs for medical products or lost productivity within 6 months of discharge. The costs experienced by patients following discharge depends on their premorbid health state before surgery and clinical course. Frailty was associated with higher health care services utilization and greater medical product use. Increasing frailty predicted that patients were less likely to be working or volunteering before admission; however, increasing frailty in patients who had been working or volunteering before admission predicted lower cost of lost productive hours. Additionally, in those who were working or volunteering before admission, increasing age was associated with decreased cost from lost productivity. This is likely explained by older or more frail patients working fewer hours per week before admission, which decreases the maximum economic loss they could experience if they were no longer able to work after discharge. This resulted in a nonlinear distribution of cost. Patients with frailty scores of 1 and 6 experienced the highest mean costs.
Cost analysis of postdischarge costs typically account for direct medical costs while ignoring the wider economic impact of recuperation. The only other study to examine postdischarge costs in general surgery patients was conducted in older patients undergoing elective colorectal surgery.18 It showed that increased cost following discharge is associated with increasing frailty, as measured by an unvalidated assessment of frailty domains. The study did not incorporate lost wages, use of complementary health care providers (e.g., massage therapists, chiropractors) or other disposable health care products used.
Overall cost was significantly influenced only by age. A 4% decrease in cost was associated with each year increase in age. This was likely a statistical error, as there was a significant increase in medical product cost (9%) with age and a significant decrease in cost of lost productive hours (5%). We measured several different sources of cost, which responded to our measured variables in different ways. Some of our measured cost categories increased with increasing frailty (medical products and health care services cost), whereas other measured variables decreased (lost productivity). This results in a nonlinear association between frailty and total cost. Patients who were well (CFS = 1) and those who were frail (CFS = 6) experienced higher cost than those who were in between. This resulted in frailty having no statistically significant influence on overall cost in our linear model and most predictors having no significant effect on overall cost.
Frail patients are more likely to be readmitted to hospital, resulting in increased emergency department utilization, which is included in our health care services cost category. Overall, only frailty predicted increased cost after controlling for age, length of stay and other clinical factors. It predicted a significant increase in the cost of health care services. This is consistent with previous findings. Addressing frailty with targeted interventions may help reduce these costs.
Medical product cost was influenced by several factors. Ostomy creation resulted in a 5-fold increase in patients’ medical product costs. After controlling for frailty, each additional day of admission and each year of life also resulted in significant cost increases. Moreover, in addition to being costly in its own right, hospital readmission may also be a clinically important driver of patient-borne costs, as readmitted patients may require additional medical products or services.
Paid work and volunteering are treated as economically equivalent in economic analysis. As people age they become less likely to work; however, volunteerism among older adults remains quite common. Our analysis found that increasing age and frailty predicted decreased employment or volunteerism before the index admission. Conversely, younger and less frail patients experienced higher costs (up to $37 000) over 6 months. This is because those who worked or volunteered more hours before admission experienced a higher economic loss if they were unable to return to their work or volunteer activities following surgery.
Previous studies have shown significant costs associated with inpatient care following postoperative complications.16,17 However, we did not find a statistically significant association between major or minor complications in any cost category after controlling for age, frailty and other clinically relevant factors. This may be due to the increased risk of complications associated with increased age and frailty. Adding complications to the model may not add any explanatory power to the model beyond the variables already in the model.
Many of the factors we have identified that influence cost are not modifiable. However, identifying frailty allows for improved assessment and implementation of frailty-specific care plans. The use of comprehensive geriatric assessment (CGA) to evaluate seniors has been shown to improve outcomes in a Cochrane review of acute medical admissions.35 A systematic review of economic evaluations of CGA in a surgical setting has shown improved outcomes while also reducing cost in patients with hip fracture, 36 and a Cochrane review of CGA in surgical patients is currently underway.37 We are currently investigating the effect of CGA and an elder-friendly care program in an acute general surgery patient population19 to see if we can improve outcomes in frail seniors.
Limitations
To our knowledge, this study is the first to examine a range of costs, including lost wages, in elderly patients following discharge after emergency surgery. However, though we conducted in-depth assessment of patient-reported sources of cost after discharge using a validated questionnaire, data collection did not include overnight care in rehabilitation programs or outpatient laboratory tests. Moreover, despite the size of the overall sample, subgroup analyses were limited by small sample sizes, as many patients declined to respond to questionnaires or were unavailable. It is possible that nonresponders included patients with increased frailty and length of stay, who were more likely to be transferred to higher levels of care at much higher cost; this would make follow-up more difficult and would result in our analysis generating conservative estimates for costs. It is also possible that we did not identify any significant effect from complications because those who experienced complications (and consequently had longer lengths of stay) did not answer our questionnaire. We were also unable to assess cost associated with death postdischarge, as our survey was conducted at the final follow-up for the surgery or in patients who were managed conservatively by a surgical team because they did not meet our inclusion criteria. Further microcosted analysis of all enrolled patients is planned when the EASE study is published. Despite collecting and assessing a comprehensive list of clinical and operative variables in-hospital, lower R2 and BIC values suggest that much of the variation between costs results from factors not controlled for in our models. Finally, some of our cost subgroups had only a small number of respondents who experienced a cost. This is commonly seen in cost analysis and has been controlled for within our analysis but does limit the interpretability of our results and may be a source of low R2 in our models.
Conclusion
Understanding predictors and types of cost accrued following surgical discharge in older patients is important to sufficiently address rising health care expenditure and understand the economic impact of surgery in elderly patients. Previous economic models to predict postoperative costs have mostly been unsuccessful.10 Our findings will be useful to physicians and policy-makers as they consider the financial burden experienced by patients and the attributes associated with higher cost. First, frailty should be considered in prediction models of postdischarge costs. Second, interventions designed to reduce perioperative morbidity, and consequently length of stay and readmission, should consider varying degrees of frailty and consider system costs as well as patient-borne costs among older adults, including lost productivity. Finally, this study highlights a need for further investigation of whether targeted interventions can reduce inpatient and postdischarge costs.
Footnotes
Funding: Financial support was provided by Alberta Innovates Health Solutions — Partnership for Research and Innovation in the Health System and the Canadian Frailty Network.
Competing interests: None declared.
Contributors: G. Eamer, F. Clement, T. Churchill and R. Khadaroo designed the study. G. Eamer and J. Pederson acquired the data, which all authors analyzed. G. Eamer, J. Pederson and R. Khadaroo wrote the article, which all authors reviewed and approved for publication.
- Accepted July 12, 2017.