Summary
Biologic mesh is preferred over synthetic mesh for complex and contaminated abdominal wall repairs; however, there are very little data on the risks and complications associated with its use. We report the case of a 67-year-old man with failed synthetic mesh repair for recurrent ventral hernia, who subsequently required an abdominal wall reconstruction (AWR), including the intraperitoneal sublay of noncrosslinked biologic mesh. His postoperative course was complicated with catastrophic sepsis and sustained hemodynamic instability, responding only to mesh explantation. The biologic mesh was subsequently noted to be histologically infected with invasive Candida albicans. Although noncrosslinked biologic mesh is a valuable adjunct to AWR, it is not infection-resistant. Although it is rare, infection of any foreign tissue, including biologic mesh, can occur in the setting of complex ventral abdominal wall repairs. Clinicians should be watchful for such infections in complex repairs as they may require biologic mesh explantation for clinical recovery.
Abdominal wall hernias are a generally underappreciated but morbid complication of abdominal laparotomy. In high-risk settings, the incidence rate of developing postoperative hernia can be as high as 69%.1 Unfortunately, simple primary tissue repair of these hernias is fraught with poor results, and worsens every time another operation is performed.2 Thus, in contemporary practice, mesh is typically used to augment the fascial reapproximation, as its use is associated with a reduced rate of hernia recurrence.3,4 This benefit is such that prophylactic use of mesh in elective surgery to prevent potential future ventral hernias is recommended.5 Synthetic meshes are now used ubiquitously in most surgical settings involving elective groin and ventral abdominal wall repairs. While synthetic mesh has been a tremendous advancement, benefitting countless patients, these products have unfavourable characteristics, particularly when infected. Although rare, synthetic mesh infections are disastrous for patients when they occur. Therefore, when an abdominal wall repair is either contemplated or necessitated in a contaminated operative field, biologic meshes are used as an alternative. Biologic meshes are derived from either porcine or bovine tissue, can be either crosslinked or not, and have been purported to be more resistant to infection.6–9 Further, it has been suggested that noncrosslinked meshes have reduced infection rates compared with crosslinked biologic meshes.10 The same finding has previously been reinforced by our own group, and we thus feel especially obligated to caution surgeons to not perceive biologic mesh to be “infection-resistant” or “infection-proof.”8 We report a devastating case of infiltrative infection of a noncrosslinked biologic mesh (Strattice) with Candida albicans following a ventral abdominal wall repair, where clinical improvement was seen only after explantation of the infiltrated mesh and treatment with systemic antifungal medications.
Illustrative case
A 67-year-old man was referred to our service for concerns of ongoing abdominal pain related to multiple recurrent ventral hernias originating from an umbilical hernia repair. He had previously undergone 3 ventral hernia repair surgeries, where his fascial defects were repaired repeatedly using synthetic prosthetic mesh implantation. Physical examination and computed tomography (CT) showed a 6.3 cm periumbilical incisional defect and possible incarcerated small bowel. After multiple preoperative consultations, he was eventually scheduled for an elective abdominal wall reconstruction (AWR). His comorbidities included hypertension, obesity (body mass index 47), type 2 diabetes mellitus, dyslipidemia, chronic obstructive pulmonary disease and sleep apnea. The patient was a distant ex-smoker and had no known history of coronary artery disease at the time of presentation.
The patient subsequently underwent open AWR with near-total explantation of the previously placed synthetic mesh, except for the edges of the mesh that were already fully incorporated into the wall of the small bowel. The new ventral defect was then reconstructed by implanting an intraperitoneal sublay of a 25 cm × 15 cm noncrosslinked porcine dermal matrix biologic mesh, over which the native fascia was primarily approximated without tension, using a unilateral external oblique over internal oblique component separation musculocutaneous flap. No respiratory support was required postoperatively, and the patient was admitted to the surgical ward. On postoperative day 2, the patient developed rapid atrial flutter with 2:1 block and hypotension, which resolved with cardioselective medications and fluid management. An abdominal CT scan obtained at the time revealed no abdominal wall concerns and showed well-approximated fascia. Though initially slow to mobilize, the patient’s well-being improved substantially over the next 5 days. However, the night before anticipated discharge, the patient had catastrophic cardiovascular deterioration, characteristically representative of profound septic shock. He was hypotensive, anemic and anuric, with an elevated white blood cell count of 30.1 × 109/L (normal range 4.5–11.0 × 109/L), requiring massive fluid resuscitation. Repeat CT showed a 7 cm × 8 cm × 9 cm hematoma and dehiscence of the implanted biologic mesh. The hematoma was emergently evacuated in the operating room, but the partially dehisced biologic mesh was left in place on one half of the abdominal wall, in hopes of using it later to reconstruct the abdominal wall in a delayed fashion. The biomesh was carefully examined by 2 experienced abdominal wall reconstructive surgeons (A.W.K. and C.G.B.) and was felt to be unremarkable and fully viable at this time. The small bowel was very dilated and edematous, and abdominal fascia closure could not be achieved; instead, wound vacuum therapy was initiated, and the patient was transferred to the intensive care unit for ongoing vasopressor support.
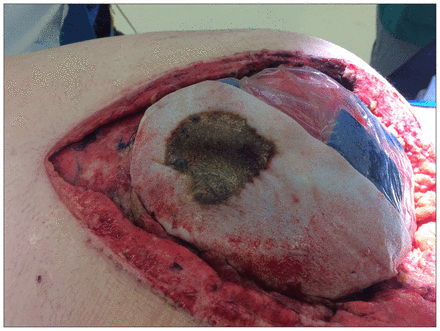
Because of ongoing hemodynamic instability and concerns of a missed source of sepsis, a repeat laparotomy the next day showed edematous bowel with no signs of necrosis or ischemia. A repeat CT scan showed mesentery edema with stranding, indicating mesenteric vascular occlusion. During the third laparotomy, however, the mesh was intraoperatively found to be visibly discoloured and blackened, and the decision was made to completely débride it (Fig. 1). Temporary abdominal closure was reapplied at the time. Within hours of mesh removal, the patient became afebrile and his white blood cell count decreased rapidly; he was also gradually weaned off the vasopressors and ventilator support. As the inflammation resolved, the edema mitigated and eventually a primary closure of abdominal fascia was completed without mesh reinforcement.
Grossly discoloured bioprosthetic mesh in a 67-year-old man who underwent abdominal wall reconstruction.
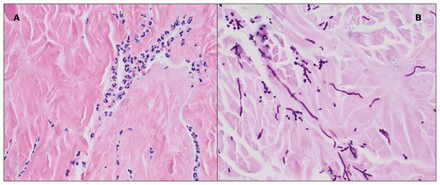
The first culture back from the explanted biologic mesh was positive for C. albicans and the patient was started on empiric treatment with micafungin. Peritoneal swabs taken during laparotomies, as well as débrided abdominal wall tissue, were also positive for C. albicans, whereas multiple blood cultures remained negative for yeast throughout the course of events. Once the detected Candida species was known to be sensitive to fluconazole and amphotericin, the patient was titrated to fluconazole therapy and remained on antifungal treatment for 4 weeks after discharge. We also deemed it important to do a detailed microscopic examination of the débrided biologic mesh, which showed acute inflammatory neutrophilic infiltrates and Candida species yeasts within the mesh, indicating fungal infection rather than contamination of the mesh itself (Fig. 2). At 1-year follow-up, the patient was doing well with no evidence of hernia recurrence or infection.
(A) Biologic mesh, consisting of collagenous tissue with an acute inflammatory infiltrate composed of neutrophils. (B) The fungal stain (Periodic Acid-Schiff stain with diastase) highlights the presence of fungal yeasts and pseudohyphae morphologically consistent with Candida species. Overall, the constellation of these findings is consistent with fungal infection rather than contamination. Magnification ×40.
Discussion
While rapid advances in tissue engineering have led to remarkable developments in dermal matrices becoming available for AWR, well-designed scientific studies to define their appropriate role in clinical practice have yet to be conducted. The direct advantages of biological mesh hence remain unproven despite their widespread usage.11 Low-level evidence and opinion has inferred that biologic meshes are superior to synthetic prosthetic meshes in repairing complex and contaminated abdominal wall defects, including work published by our own group,3,4,8,9,12,13 but long-term data on the safety and durability of biologic mesh in infected fields remains limited. A retrospective study by Rosen and colleagues reported an overall wound complication rate of 48% when biologic mesh was used in contaminated or complex abdominal fields using a single-staged approach.14 Of these, no patients required complete biologic mesh excision or developed mesh infections postoperatively.14 Another retrospective review of 27 patients, who underwent abdominal wall hernia repair with Strattice to replace infected synthetic mesh, showed an infection rate of only 19% when a multi-staged approach was used, with only 1 patient requiring mesh explantation.15 Though these results are somewhat similar to the findings of our reported case, authors reported mesh infection with typical bacterial species, unlike the fungal species detected in our patient.15
During our literature search, we were able to find only 2 reported cases of mesh infections with fungal species. The first case was a patient with strangulated epigastric hernia who underwent emergent repair and deteriorated postoperatively with positive wound cultures for C. albicans. After being treated with fluconazole for 20 days, a biologic mesh was placed in situ, but when consecutive wound cultures remained positive for Candida species, the mesh was removed and replaced. After implantation of the new mesh, Candida krusei (known to be resistant to fluconazole) was detected in wound cultures and, despite aggressive treatment with micafungin, the new mesh had to be explanted for source control.16
The second reported case involved fungal infection of a synthetic mesh (Proceed) in a woman with emergent laparascopic paraumbilical hernia repair. Postoperative CT revealed a communicating fluid collection containing gas around the mesh, which was drained and cultured for C. albicans. Authors reported successful conservative treatment with only routine washouts and antifungal fluconazole, disputing the need for surgical débridement or mesh removal.17 However, neither of these cases involved hemodynamically significant sepsis in the patients.
Conclusion
Even though biologic mesh is considered relatively infection-resistant, our reported case highlights a rare but dramatic postoperative complication of invasive tissue infection, when operating on high-risk patients undergoing complex AWR. The lead surgeon was hesitant to explant the biologic mesh at the first reoperation, as the team was still considering definitive reconstructive options. However, when the patient remained catastrophically septic without any other obvious source of infection, this decision had to be reconsidered. Microscopic examination revealed that the biologic mesh was the source when it was found to be fully infiltrated from within (and not just superficially contaminated) with C. albicans. Thus, it is critical that all involved in AWR recognize that biologic meshes may not be as durable and resistant to infection as previously thought. Furthermore, we remind clinicians to always maintain a high degree of suspicion for fungal infections when severe sepsis occurs in patients with known risk factors such as diabetes and obesity. Depending on the clinical situation, it may also be worthwhile to consider fungal assays for ventral abdominal wounds, as well as possible successive explantation of the biologic mesh, if needed.
Footnotes
Competing interests: C.G. Ball is coeditor-in-chief of CJS. He was not involved in the review or decision to accept this manuscript for publication. No other competing interests were declared.
Contributors: All authors contributed substantially to the conception, writing and revision of this article and approved the final version for publication.
- Accepted January 7, 2020.