Abstract
Background: There are limited published data on population estimates of survival after spinal surgery for metastatic disease. We performed a population-based study to evaluate survival and complications among patients with cancer who underwent surgery for spinal metastases in Ontario, Canada, between 2006 and 2016.
Methods: We used health administrative databases to identify all patients who underwent surgery for spinal metastases in Ontario between Jan. 1, 2006, and Dec. 31, 2016. We assessed overall survival, mortality rates according to primary cancer lesion and complications after surgery. We contrast the results to those for a comparable cohort from 1991 to 1998.
Results: A total of 2646 patients (1194 women [45.1%]; mean age 62.5 yr [standard deviation 12.2 yr]) were identified. The median survival time was 236 (interquartile range 84–740) days. Mortality was highest for patients with melanoma, upper gastrointestinal cancer and lung cancer, with 50% dying within 90 days of surgery. The longest median survival times were observed for primary cancers of the thyroid (906 d) and breast (644 d), and myeloma (830 d). Overall 90-day and 1-year mortality rates were 29% and 59%, respectively.
Conclusion: We identified differential survivorship based on primary tumour type and a shift in the distribution of operations performed for specific primary cancers over the past 2 decades in Ontario. Overall reductions in mortality associated with this shift in treatment may reflect the use of adjuvant therapies and more personalized treatment approaches.
Spread of metastatic disease to the spine causes substantial clinical burden, affecting nearly one-third of all patients with cancer.1,2 Advances in medical treatment for systemic disease has improved survival rates among patients with cancer, which has contributed to an increased incidence of spinal metastases.3,4 Erosion of the bony spine architecture can lead to mechanical instability, with pathological fracture, and epidural disease can lead to spinal cord compression, with associated neurologic injury.5 Quality of life is affected considerably owing to pain, spinal cord injury and loss of functional abilities.6,7
Management of patients with spinal metastases is aimed at alleviating pain, delaying functional decline and maintaining an acceptable quality of life.2,8,9 The multidisciplinary strategies used to achieve these goals and provide palliative care include chemotherapy, radiation and, when appropriate, surgery.10 The goal of surgical intervention in these patients is to maintain or restore stability, provide analgesia and reverse neurologic deficit.2,11 Surgical intervention has now also become an adjuvant for stereotactic body radiation therapy (SBRT) to create separation between the spinal cord and the tumour.12,13 Considerations regarding surgical candidacy include overall medical status, number of spinal levels involved, age, cancer type, radiation status and life expectancy.8,11,14 The importance and challenges of estimating survival and the potential complications of surgery cannot be overemphasized, as the decision to operate must be made with the intent of maximizing the quality of remaining life.15–19
There are limited published data on population estimates of survival after spinal surgery for metastatic disease.2,20,21 Over the past decade, improvements in the management of spinal metastases have included advances in classification systems, surgical innovations with more minimally invasive techniques, image guidance and use of intensity-modulated radiation therapy, improved chemotherapeutic regimens and targeted personalized therapies based on genetic typing of tumour type.22–27 With these advances, it is likely that the demographic characteristics of patients undergoing surgery, as well as survival and complications of surgery, have changed substantially.2,9,10 Thus, we sought to evaluate the survival of patients who underwent surgery for spinal metastasis in Ontario, Canada, between 2006 and 2016. Our secondary objective was to determine postoperative complications and identify risk factors that may lead to poor outcomes. We contrast the current results to those of a comparable cohort from 1991 to 1998.2
Methods
Study design and setting
We performed a population-based, retrospective cohort study of patients who underwent their first surgical treatment for spinal metastases in Ontario between Apr. 1, 2006, and Dec. 31, 2016. Ontario residents have access to universal health care through the Ontario Health Insurance Plan (OHIP), which covers medically necessary hospital and physician services.
Data sources
We used multiple health administrative databases to identify and describe study patients and ascertain outcomes. We obtained patient demographic information and vital status from the OHIP Registered Persons Database. The OHIP Claims History Database provided information on physician services, including diagnoses and procedures. The Canadian Institute for Health Information Discharge Abstract Database provided inpatient and outpatient hospital diagnoses using the enhanced Canadian version of the International Statistical Classification of Diseases and Related Health Problems, 10th Revision (ICD-10-CA), and surgical procedures were defined with the Canadian Classification of Health Interventions codes. We identified emergency department visits using the Canadian Institute for Health Information National Ambulatory Care Reporting System. We used the Ontario Cancer Registry28 to verify cancer diagnoses using International Classification of Diseases for Oncology, Third Revision (ICD-O-3) codes. These data sets were linked by means of unique encoded identifiers and analyzed at ICES.
The use of data in this project was authorized under section 45 of Ontario’s Personal Health Information Protection Act, which does not require review by a research ethics board.
Cohort selection
We identified all people aged 20 years or older who underwent surgery for spinal metastases between Apr. 1, 2006, and Dec. 31, 2016 (see Appendix 1, Table S1, available at www.canjsurg.ca/lookup/doi/10.1503/cjs.000921/tab-related-content, for vertebral surgical procedures for metastatic disease). The cohort included a primary cancer diagnosis of metastatic bone tumour within 6 months before or after the index date of surgery (i.e., International Classification of Diseases, 9th Revision code 198.5 or ICD-10-CA code C795 appearing at any diagnosis level). People who were not residents of Ontario, and those with missing age or sex, or an invalid health insurance number were excluded. We also excluded anyone aged younger than 20 years and those who died on the index surgery date.
Covariates
We captured several sociodemographic characteristics of our cohort, including age, sex, urban versus rural place of residence (defined by postal code) and neighbourhood income quintile (derived from census data). We used the Deyo–Charlson Comorbidity Index (computed from hospital encounters over the 3 years before the index surgical procedure) to characterize comorbidity.29 We also recorded the calendar year of the index procedure and the anatomic site of the primary cancer (see Appendix 1, Table S2 for definitions of primary cancers of interest).
Outcomes
The primary outcome was time from the index surgery to death from any cause, with a minimum of 365 days of follow-up. Secondary outcomes included 30-day all-cause hospital readmission, surgical complications in the 30 days after the index procedure, deep vein thrombosis, pulmonary embolism, a major vascular event and vertebral fracture. The secondary outcome definitions are provided in Appendix 1, Table S3.
Statistical analysis
We described patients’ characteristics at the time of the index surgery using descriptive statistics. We assessed survival and crude mortality rates after surgery overall and by primary cancer site. We used the Kaplan–Meier method to estimate overall survival, measured from the time of the index hospital discharge to death, with censorship at loss of OHIP coverage or the end of the follow-up period (Dec. 31, 2017). Secondary outcomes (complications) were also described.
Results
A total of 2646 patients with cancer (1194 women [45.1%]; mean age 62.5 yr [standard deviation 12.2 yr]) underwent surgery for spinal metastases during the study period (Table 1). The 6 most common primary cancer sites/types were breast (462 patients [17.5%]), prostate (337 [12.7%]), urinary system (286 [10.8%]), myeloma (173 [6.5%]) and lower gastrointestinal tract (165 [6.2%]).
Characteristics of patients at the time of initial surgery for spinal metastases in Ontario, Apr. 1, 2006, to Dec. 31, 2016
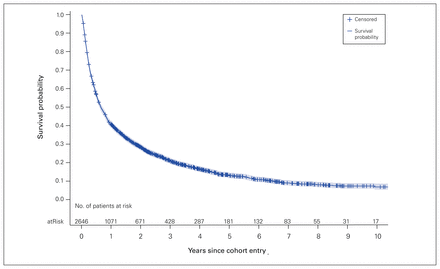
During 4005 person-years of follow-up, 2230 patients (84.3%) died, for a mortality rate of 55.7 (95% confidence interval 53.4–58.0) per 100 person years. The median survival time after surgery was 236 (interquartile range 84–740) days (Table 2 and Figure 1). Overall, 214 patients (8.1%) died within 30 days after surgery, 706 (26.7%) died within 90 days, and 1561 (59.0%) died by 1 year. The shortest median survival times were among patients with melanoma (90 d), upper gastrointestinal cancer (90 d) and lung cancer (95 d), with half of these patients dying within 90 days of surgery. The longest median survival times were observed for primary cancers of the thyroid (906 d) and breast (644 d), and myeloma (830 d).
Kaplan–Meier survival curve for patients who underwent initial surgery for spinal metastases.
Survival by site/type of primary cancer
During the 30-day postsurgery period, 553 patients (20.9%) were readmitted to hospital, and 323 (12.2%) had at least 1 surgical complication or any major outcome event. The most common complication type was a complication of the surgery itself (240 patients [9.1%]). During the entire follow-up period, the most common complications were pulmonary embolism (92 patients [2.6%]), deep vein thrombosis (38 [1.4%]), vertebral fracture (35 [1.3%]) and a major vascular event (31 [1.2%]) (Table 3). There was no important difference in the annual proportion of patients who were readmitted or experienced a complication within 30 days over the study period (Appendix 1, Table S4).
Outcomes
A total of 609 patients (23.0%) received radiation of the spine before surgery. We included 1730 patients for analysis of postoperative radiation; of the 1730, 772 (44.6%) underwent conventional radiation treatment within 8 weeks after surgery, 195 (11.3%) received SBRT (with or without conventional treatment), and 763 (44.1%) had no radiation. Patients who underwent SBRT after surgery were on average younger and more likely to be female than those who received conventional radiation and those who did not receive radiation.
Discussion
In this large population-based cohort study, we identified a median survival duration of 236 days among all patients who underwent surgical interventions for metastatic disease to the spine. We observed survival to be poorest for patients with melanoma, upper gastrointestinal cancer and lung cancer, with half dying within 90 days of surgery. Patients with primary cancers of the thyroid and breast, and those with myeloma had the longest survival times.
There have been several previous reports assessing outcomes after spinal surgery for metastatic lesions.30–33 The number of patients assessed in those reports ranged from 62 to 282, and the overall 30-day mortality rate ranged from 3% to 13%. In our current study, 8% of patients died within 30 days of surgery. Moreover, similar to previous investigators, we observed wide variation in survival times by primary tumour type. Reported complication rates ranged from 8% to 34%.19,33–36 In our cohort, 12% of patients experienced a complication after surgery, and 21% were readmitted within 30 days of operation. The wide variation may be a consequence of differences in definitions, and reporting of major and minor complications.
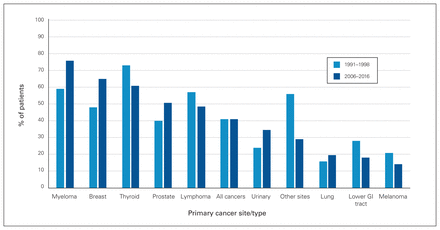
There have been almost 2 decades of treatment advances since our previous report on population-based survival after spinal surgery for metastatic disease.2 In that study, with similar methodology, we had a cohort of 987 Ontario patients who underwent surgery for spinal metastases between 1991 and 1998. We reported 90-day and 1-year mortality rates of 29% and 59%, respectively, which is almost identical to survival in the present cohort. We identified improved survival outcomes for specific cancers compared to our previous report, as well as a shift in the distribution of operations performed for specific primary cancers. We observed improved 1-year survival among patients with myeloma (76% v. 59%), breast cancer (65% v. 48%), prostate cancer (51% v. 40%), urinary system cancer (35% v. 24%) and lung cancer (20% v. 16%) (Figure 2). However, survival appears to have decreased for thyroid and lower gastrointestinal cancer, other cancer sites, lymphoma and melanoma. This says nothing about survivability from the time of diagnosis of the initial cancer, and it should be emphasized that we analyzed survival after surgery, not after diagnosis of disease. Hence, although overall survival may have improved for some cancer types, surgery may have been performed late in the patients’ disease process.
Proportion of Ontario patients who survived 1 year after surgery for spinal metastases, 1991–19982 and 2006–2016 (present study). GI = gastrointestinal.
There is little level I or II evidence and conflicting reports on the comparative effectiveness and risks of SBRT versus conventional radiation for spinal metastases.37–40 Among radiation modalities, conventional radiation was used most frequently in the present cohort, as SBRT was in its infancy during the period used for this study. After we adjusted for confounding factors using multivariable Cox regression, radiation after surgery was not associated with improved survival. However, selection biases and other, unmeasured confounding factors may have contributed to these results. Further study regarding the role of SBRT in survival is indicated.
The results of this study may provide further guidance to surgeons about surgical indications and help quantify risks and benefits based on patient factors. Evidence-based treatment has influenced how metastatic disease to the spine has changed over the past 2 decades.41 Compared to our previous cohort,2 a lower proportion of patients with primary lesions of the lung (3% v. 19%) and those with lymphoma (2% v. 10%) in the current study underwent surgery, and a higher proportion of those with breast cancer (18% v. 13%) had surgery. This suggests that lessons learned (high crude mortality rate in lung cancer), identification of ideal patients for surgery and the use of newer evidence-based treatment algorithms have guided surgeons in patient selection.42
Limitations
A limitation to our study is the absence of important clinical variables such as time between onset of epidural compression or myelopathy and surgery. Prolonged duration of metastasis to the spine before surgery may negatively affect survivorship, as survival of patients with certain cancers appears to have decreased compared to our prior study from 2 decades ago.2 There are conflicting reports on the effect on survival of time to surgery after the onset of spinal metastases.43,44 Our study did not directly evaluate the role of timeliness to surgery, chemotherapy and associated adjuvant strategies for the management of spinal metastases. Furthermore, we were unable to differentiate variability in survival and prognoses based on additional factors that may have had an impact, such as genetic variations in tumour types and patient demographic characteristics.
Conclusion
In this population-based study reflecting real-world patients undergoing surgical management of spinal metastases in Ontario, primary cancer type was the most important factor in determining survivorship. The crude number of operations based on primary tumour type changed between 1991–1998 and 2006–2016, with fewer procedures performed for lung cancer and an increased number performed among patients with breast cancer. Importantly, there were substantial improvements in survival among patients with myeloma and with breast, prostate and urinary cancers in more recent years. These findings have implications for the clinical management of spinal metastases in patients with cancer.
Acknowledgement
The authors acknowledge the Feldberg Chair in Spinal Research for funding this work.
Footnotes
Competing interests: Joel Finkelstein receives consultant fees from Zimmer Biomet Institutional Educational Support. No other competing interests were declared.
Contributors: All authors designed the study. K. Bhanot, J. Widdifield, A. Huang and J. Paterson acquired the data, which J. Widdifield and A. Huang analysed. K. Bhanot, J. Widdifield and J. Finkelstein wrote the manuscript, which all authors critically revised. All authors gave final approval of the article to be published.
Funding: This study received funding from the Feldberg Chair in Spinal Research, awarded to Joel Finkelstein by the Sunnybrook Health Sciences Centre. Jessica Widdifield holds a salary award from the Arthritis Society Stars Career Development Award (STAR-19-0610).
Disclaimer: This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health. Parts of this material are based on data and information compiled and provided by the Canadian Institute for Health Information and Cancer Care Ontario. The analyses, conclusions, opinions and statements expressed herein are solely those of the authors and do not reflect those of the funding or data sources; no endorsement is intended or should be inferred.
- Accepted October 20, 2021.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/