Abstract
Background: Complex abdominal wall reconstruction technique remains controversial. The use of biologic mesh products is also debated in active infection, sepsis prophylaxis and high-risk patients. Differences in biologic mesh technology and cost remain significant. We aimed to compare the efficacy of 2 commonly used biologic meshes in regards to hernia recurrence at 1 year.
Methods: This study was a parallel, dual-arm, double-blind randomized controlled trial involving adult patients undergoing complex abdominal wall reconstruction with a biologic mesh at a quaternary care institution (2017–2020). Patients were randomly assigned to receive Permacol (cross-linked) compared with Strattice (not crosslinked). The main outcome measure was hernia recurrence at 1 or more years following the index repair.
Results: We included 94 patients randomized to undergo reconstruction with 1 of 2 commonly used biologic mesh products (mean age 59.4 yr, standard deviation [SD] 9.9; 51% female; body mass index 32.9, SD 6.8). We found no significant differences between the groups (patient comorbidities, hernia recurrence risk factors, hernia size or infection profiles). Hernia recurrence rates (15%) were similar between groups (median 783 days of follow up, interquartile range 119). We found there was significantly less of a need for a component separation technique in the Strattice group (69% v. 87%). All other secondary outcome measures were equivalent between study arms. Multivariate analysis identified hepatic transplantation (odds ratio [OR] 1.94, 95% confidence intervals [CI] 0.33–4.41), active abdominal wall infection (OR 2.01, 95% CI 0.50–7.01), and more than 1 previous hernia repair (OR 2.68, 95% CI 0.41–5.99) as risk factors for subsequent hernia recurrence; however, there was no difference in recurrence factors between patient study groups.
Conclusion: Given similar clinical performance between the 2 most commonly used biologic mesh products, the most cost effective mesh should be used in cost-conscious health care systems.
Incisional hernias after abdominal operations are unfortunately common for patients (up to 23%) and costly to health care systems.1–6 In patients who pursue surgical repair by way of abdominal wall reconstruction, the quality of the index repair correlates with the subsequent recurrence rate. Furthermore, with each successive hernia repair, the risk of failure increases dramatically.7 Careful patient selection, prehabilitation, precise surgical technique and selecting the optimal procedure from a wide variety of available options (i.e., addressing patient anatomy and the quality of the abdominal wall componentry) are critical to ensure a durable repair.8
In a clean operation without bacterial contamination, a permanent synthetic mesh is typically employed as a reinforcement to lower the risk of recurrence (i.e., by 50%).9–11 This technique has represented the standard of care for decades. Unfortunately, the management of patients with active abdominal wall infections; high risk of postoperative sepsis, such as those who undergo synchronous gastrointestinal procedures (i.e., stoma reversal, enterotomy repair, fistula closure); and/or immunosuppression (i.e., systemic impact of an infection), is fraught with concern over infected synthetic mesh products. These scenarios may require open wounds, repeat debridements, prolonged antimicrobials or even reoperative mesh explantation in some cases.
Biologic meshes have been promoted as resisting infection (i.e., bacterial contamination) and enabling wound healing.12–15 These products are derived from the living tissue of mammals (bovine, porcine and human) and have displayed significant commercial innovation over time. Biologic meshes have been purported to reduce the risk of surgical site infections (SSI) in patients, as well as offer high resistance against infections in animal models.13 Although recent evidence suggests otherwise,16,17 these reports have aligned well with the traditional belief and training that placing synthetic mesh when there is a high risk for infection is contraindicated. Interestingly, the process of cross-linking some mesh products (i.e., supports fibroblast growth and resistance against collagenase enzymes) prolongs structural integrity (i.e., slows degradation) over time. The relative benefits and risks of this manufacturing process are heavily debated.
Given variable manufacturing methodologies (e.g., products that were cross-linked v. products that were not cross-linked), as well as significant cost differences between biologic mesh products, the primary aim of this study was to compare the efficacy of 2 commonly used biologic meshes (the majority of market share) in regards to hernia recurrence at 1 year.
Methods
Study design and participants
We conducted a prospective, double-blind, randomized controlled trial at a single centre in Calgary, Alta., using a parallel-group design and an intention-to-treat analysis. The specific study protocol has been previously published in peer-reviewed format.18 This protocol complied with the Declaration of Helsinki, followed the Consolidated Standards of Reporting Trials (CONSORT) Guidelines and was approved by the Conjoint Health Research Ethics Board at the University of Calgary.18 The study was designed as a pragmatic trial to reflect the full spectrum of clinical practice to maximize both relevance, and the ability to extrapolate findings to other centres and surgeons. Pragmatic trials are particularly helpful for comparing effectiveness and cost in real-world situations.
The study population consisted of adult patients (aged ≥ 18 years) undergoing a major abdominal wall reconstruction between October 26, 2017, and August 31, 2020. All patients potentially eligible for abdominal wall reconstruction underwent routine computed tomography (CT) of the abdomen for operative planning and abdominal wall anatomy assessment. Criteria for biologic mesh use included large abdominal defects with significant contamination/the presence of a stoma, loss of abdominal wall secondary to trauma or necrotizing infection, or abdominal wall reconstruction in patients identified as being at high risk for infection. The suitability of a biologic mesh was determined by way of consensus in the preoperative setting between the 2 surgeons involved in the trial (C.G.B. and A.W.K.), and following the institutional guideline.
Any patient involved in this study who had a Centers for Disease Control and Prevention (CDC) grade IV wound infection was downgraded to grade III before intervention. All operations were performed at the Foothills Medical Centre (quaternary care referral centre) by 2 fellowship-trained surgeons with a special interest in abdominal wall reconstruction. Exclusion criteria were limited to patients who were younger than 18 years or who were unable to provide informed consent. Written informed consent was obtained from all study participants before enrolment. Strattice (Life-Cell Inc.) and Permacol (Medtronic Inc.) were selected as the biologic mesh comparators given their dominant market share for abdominal wall hernias. Porcine dermis (Strattice) that was not cross-linked was available in sheets in various sizes (ranging from 6 cm × 6 cm to 25 cm × 40 cm). Chemical cross-linking in Permacol is completed using a proprietary process with noncalcifying hexamethylene diisocyanate. Permacol was also available in sheets (ranging from 1 cm × 4 cm to 28 cm × 40 cm).
Randomization and intervention
Study participants were block randomized in a 1:1 ratio to receive either Strattice or Permacol biologic meshes (block size was not disclosed to preserve allocation concealment). The surgical team received the product in a blinded manner as allocated by the research staff before the operative case. Participants, outcome assessors, surgical team members, care providers and data analysts were blinded to study allocation status.
Perioperative care and intraoperative technique for both patient study groups followed the routine practice of the surgical team. Each patient was assessed in the outpatient clinic setting with both physical examination and preoperative cross-sectional imaging. Prehabilitation was ensured when necessary (smoking cessation, diabetes optimization, body weight reduction, immunosuppressive medication evaluation and treatment of active abdominal wall infections). Epidural anesthesia was common (61% of patients). Operative preparation included hair removal with clippers, the use of clorhexadine-based skin antiseptic solutions, preoperative intravenous antibiotic (cefazolin 2 g and metronidazole 500 mg if gastrointestinal work was also required; standard re-dosing for longer cases), subcutaneous deep venous thrombosis (heparin) prophylaxis within 1 hour before the skin incision, plastic adhesive skin barrier protection, surgical incision (midline), fixed retractor selection when necessary, paper-based surgical drapes, frequency of glove changes during longer cases and intraoperative use of sponges and saline irrigation to control/evacuate any contamination. The specific technique for abdominal wall reconstruction varied by case. However, the technique was consistent between surgeons and variably included underlay, retrorectus, onlay with transfascial fixation mesh strategies and bridging, depending on specific patient variables (abdominal wall anatomy, hernia defect morphology or risk of infection). No synthetic mesh was positioned intraperitoneally in any patient. Synchronous component separation, as well as uncommonly “modified” component separation, techniques were employed when necessary.19–21 The hierarchy of releases followed a standard pattern (unilateral posterior rectus sheath followed by bilateral posterior rectus sheath, followed by unilateral external oblique aponeurosis, followed by bilateral external oblique aponeurosis). Associated principles included minimizing flaps and dead space, abdominoplasty when necessary, quilting of all onlay meshes to the fascia, use of generous closed suction drainage and multilayer reinforcement for wound closures. All sutures at all sites were absorbable (usually polydioxanone), except for skin closure (i.e., staples and reinforcing nylon mattress sutures v. a minority with subcuticular absorbable sutures).
Definitions and data collection
The primary outcome measure was the incidence of all postoperative hernia recurrences at 1 year after the index reconstruction. This diagnosis was confirmed by both physical examination and cross-sectional imaging. Secondary outcomes included postoperative complications (SSI, need for re-intervention, infected intraabdominal fluid collection (i.e., organ space SSI, either turbid output through surgical or percutaneous drain and need for antibiotic therapy), hemorrhage (requiring reoperation, radiologic or endoscopic intervention), pleural effusion (if symptomatic or required thoracocentesis/ drainage), pulmonary embolism (confirmed by ventilation–perfusion scan or CT), deep venous thrombosis (confirmed by ultrasonography or CT), acute kidney injury, arrhythmias (absence of cardiac sinus rhythm prompting specific medical intervention or patient transfer to a monitored bed), acute myocardial infarction, cerebrovascular accident or transient ischemic attack (as diagnosed by an appropriate medical specialist). Infectious complications were defined by the CDC diagnostic criteria (i.e., pneumonia, urinary tract infections, bloodstream infections and SSI).22–25 Confirmation of the presence of an incisional SSI occurred by way of direct observation at the patient’s bedside (i.e., no chart or database evaluations/ reviews). The patients, as well as wound and complication assessors (study nurse), were each blinded to the study allocation status. Furthermore, no information regarding the study arm allocation was recorded in the chart (clinical notes). All outcome assessors were provided with standardized criteria and instructions regarding assessment. Once patients were discharged, their wounds were assessed in an outpatient clinic setting using the above principles and methods.
Additional secondary variables of record included patient age, sex, presence of diabetes mellitus, chronic obstructive pulmonary disorders, body mass index (BMI), smoking status, American Society of Anesthesiologists score, preoperative albumin level, patient surgical history (i.e., number of preceding hernia repairs), perioperative chemotherapy details, type/date of immunosuppression, details of intraoperative and postoperative fluid administration, type of operation, length of the operation (min), estimated blood loss (mL), length of hospital stay (d), and 90-day postoperative mortality. Specific characteristics of the incisional hernia itself were also noted (length, width, position, volume of abdominal viscera outside of peritoneal domain and quality of abdominal wall componentry). After patients were discharged, they were assessed by their operating surgeon (i.e., physical examination) at about 1, 3, 6, 12 and 24 months. All patients with concern for recurrence of a hernia on physical examination underwent subsequent CT for confirmation and characterization. Given the close patient follow up, as well as the availability of a province-wide, population-based electronic medical record for all patients, data were secured for all patients without loss.
Sample size and statistical analyses
This study was conceived by the statistical and clinical teams with an accrual goal of 90 patients (45 within each study arm) and analyzed as an intention-to-treat trial. This was according to an incidence of postoperative/ reported rate of hernia recurrence after abdominal wall reconstruction with biologic meshes of 15% (i.e., the rate of success was estimated to be 85%).18 Sample size for this equivalence trial was therefore calculated using a significance level (α) of 0.05, a power (β) of 90%, and an equivalence limit of 25%. Two interim analyses were planned and occurred at 33% and 66% of total accrual. These were reviewed by a safety review board comprising a data analyst, clinical researcher and faculty surgeon. Patient demographics and clinical characteristics were compared using the χ2 test or Fisher exact test for categorical variables and Student t and Mann–Whitney U tests for continuous variables. Univariate and multivariate analyses were also completed. A 2-sided, p value threshold of 0.022 for each interim analysis was defined according to the Pocock boundary.26 Statistical analyses were conducted using Stata/IC version 17.0 (Stata Corp. LP) and R version 3.0.1 (available at http://www.r-project.org/). A portion of this study was funded by an unrestricted grant from Medtronic, Inc.
Results
Patients
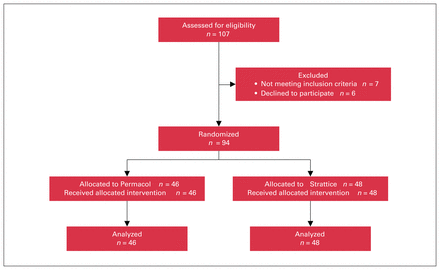
We included 94 patients randomizeed to undergo reconstruction with 1 of 2 commonly used biologic mesh products (Table 1) between October 26, 2017, and August 31, 2020. All patients were followed to at least 1 year after their operative intervention (no patients or data elements were lost to follow up). Forty-six patients were allocated to the Permacol study arm and 48 to the Strattice study arm (Figure 1). The mean patient age was 59.4 years (standard deviation [SD] 9.9), 51% were female, and the mean BMI was 32.9 [SD 6.8]). Comorbidity profiles and hernia recurrence risk factors (smoking status, diabetes mellitus, Charlson Comorbidity Index (CCI) score, BMI, medical immunosuppression, active infections and preceding repairs) were similar between the groups (Table 1). All patients were evaluated by our internal medicine and anesthesiology colleagues in the preoperative assessment clinic for optimization. The wound status was similar between groups across all subtypes (Table 2). Patients with wounds that could be improved (e.g., conversion from dirty toward clean categories) underwent considerable effort in the preoperative setting to achieve these goals. Most patients (60.6%) received an epidural catheter for intraoperative and postoperative analgesia.
CONSORT flow study diagram.
Demographics and clinical characteristics according to study group allocation
Intraoperative and characteristics according to study allocation
Operative data
A midline incision was performed in 98% of patients (2% with a lateral extension). Five patients (5.3%) underwent a bridged repair, while the remaining patients received a typical distribution of other techniques (54% intraperitoneal, 23% onlay with transfacial fixation and 17% retrorectus). The mean duration of surgery was 163 minutes (SD 51), with an associated mean estimated blood loss of 160 mL (SD 195 mL). Subcuticular skin closure was used in the minority of patients (17.0%). In the remaining patients, we used skin staples and reinforcing horizontal mattress nylon sutures. Placement of closed suction drainage was left to the discretion of the individual surgeon, but it was frequently employed (91.5% of reconstructions [86.0% 2 drains; 9.3% 1 drain; 8.1% 3 drains]). Red blood cell transfusions were required in 2.1% of cases. Blood transfusions were intraoperative in 1 of 3 patients in each study arm. Operative data according to the patient study group allocation are displayed in Table 2.
Postoperative outcomes
No immediate postoperative deaths were noted in either group. There were no deaths within 90 days after surgery in either group. The overall complication rate was 55.3% using the strict definitions previously described (Table 3). The specific incidence of complications within the Permacol and Strattice groups was statistically similar (46.0% v. 64.6%; p = 0.06 ). This nonsignificant trend (p = 0.06) was not created by an increased rate of any individual complication compared with others (Table 3). Fourteen (14.9%) patients experienced a hernia recurrence within the follow-up period (median 783 days, interquartile range [IQR] 119). The median time to confirmation of a recurrence was 151 days (IQR 38). Two patients received a biologic mesh hernia repair during an initially unrelated index inpatient stay (1 patient underwent a reconstruction with a synthetic mesh and suffered an unrecognized intestinal injury requiring reoperation and the use of a biologic mesh during the subsequent procedure; 1 patient underwent a second reconstruction after an index repair by a surgeon who was not involved in this study that suffered an early catastrophic abdominal wall repair failure). No readmissions were noted within 90 days after discharge from the index abdominal wall reconstruction. One patient required placement of an outpatient percutaneous drain for an early seroma. All patients with remaining wound care issues were successfully treated as outpatients after the index hospital stay by way of home care nursing, surgeon clinic visits and rarely antimicrobials. The patient length of hospital stay was similar between groups (Table 3). The type of repair (i.e., position of the mesh) (Table 2) did not affect outcome rates or comparisons (p = 0.03).
Number of postoperative complications and outcomes by allocation group
Neither univariate, nor multivariate logistic regression analyses identified any specific preoperative, patient or intraoperative variables that altered the statistical conclusions reported above (including patient age, sex, preoperative patient comorbidities, technique of reconstruction, length of surgery and estimated blood loss), except for the number of preceding surgical repairs, the presence of active abdominal wall infection at the time of reconstruction and previous liver transplantation. Patients with more than 1 previous hernia repair (2–5) displayed an increased risk of recurrence (OR 2.68, 95% confidence intervals [CI] 0.41–5.99). Similarly, the presence of active infection and preceding liver transplantation increased the risk of developing a recurrence (OR 2.01, 95% CI 0.50–7.01 and OR 1.94, 95% CI 0.33–4.41, respectively). All cases of organ space SSI were successfully managed with antibiotics with or without percutaneous drainage (Clavien–Dindo classification, grade III).
Discussion
Significant progress has been achieved in the past 2 decades in patient optimization and surgical savvy, despite the continued debate regarding optimal abdominal wall reconstruction techniques. Although we understand classic risk factors for hernia recurrence include large and complex defects, contaminated surgical fields and repairs without reinforcing mesh products, a general surgeon is required to operate within these scenarios.7,27,28 This has led to the use of advanced, and expensive, biologic meshes in selected cases. Once a patient is optimized preoperatively (weight loss, tight control of diabetes, modification of immunosuppression, clearance of active infections and smoking cessation), the most common subsequent concern is the risk of infection and, more directly, a patient’s ability to both tolerate and clear the associated septic insult (locally and systemically).
Although the role of biologic meshes within infected areas remains controversial, the current use of these products is not uncommon. Most high-volume centres have witnessed cases of both poor performance when biologic meshes become secondarily infected by either bacteria or fungus,29 as well as excellent performance in seemingly similar scenarios. These results have not been clearly related to the development methodology of the product (e.g., cross-linking v. not) or the specific brand. Although it is clear that urgent reoperations on patients who were previously injured with biologic mesh implants is preferred in comparison to those with a preceding intraperitoneal synthetic mesh, the more common challenge of subsequent infection and hernia recurrence merits further study.
Another critical issue in contemporary health care systems remains cost escalation. Given the relatively expensive nature of biologic, compared with synthetic, mesh products, we must ensure careful and cautious use of these devices as active stewards of a single-payer health care system. There is also a significant cost difference between the 2 dominant market leaders (Strattice and Permacol). Although the specific price that a given hospital will be charged can vary according to preexisting arrangements and bulk purchase agreements, the cost of 1 biologic mesh product is generally twice as much as the other (1.8–2.2 times, depending on sizing). Given these discrepancies in manufacturing and cost, the primary aim of this randomized controlled trial was to compare the efficacy of 2 commonly used biologic meshes in regards to hernia recurrence at a minimum 1-year follow up.
The overall recurrence rate noted within the trial was 15% at a median of 783 days (IQR 119). Considering the complexity of these patients (63% had previous hernia repairs; mean CCI 3.5 (range 1.5–4.6); mean BMI 32.9, SD 6.8; 35% preceding transplantation), this result fits within the contemporary literature.1–28 However, it must be noted that recurrences continue to occur as the follow-up interval lengthens. More specifically, at 140 months of follow up, there were 37% of patients with primary ventral hernias compared with 64% of patients with incisional hernias.7 In patients who have undergone 3 previous incisional hernia repairs, this rate is about 73% at 140 months.7 Although the median follow-up in this study was 783 days (2.2 yr, IQR 119), it can be assumed that recurrence rates will increase as these patients are followed in coming years.
Despite the intermediate timeframe of these results, patients with confirmed recurrences mandate a nuanced analysis. Patients with a preceding hepatic transplantation, more than 1 previous incisional hernia repair/active abdominal wall infection, possessed an increased risk of a subsequent hernia recurrence. Although this risk varied from 1.94 to 2.68 depending upon the specific factor, there are a number of underlying lessons that can reemphasized. Although a past medical history inclusive of hepatic transplantation is not typically avoidable, potential modification and optimization of a patient’s required immunosuppressive regime is critical (i.e., by a transplant surgeon or hepatologist). Similarly, adequately treating active abdominal wall infections and eliminating them before reconstruction when possible, is crucial. This may require a 2-stage procedure (i.e., extraction of a grossly infected synthetic mesh followed by a delayed reconstruction once all tissues have healed) in some patients to optimize the performance of any subsequent mesh product and minimize a cyclical devolution into repeated complications. Lastly, the increasing rate of hernia recurrence noted with additional repairs is essential to consider. It reminds us that our first repair is likely the best chance at overall durability, patient satisfaction and a surgeon’s technical success.
Given that the recurrence rate was similar between both biologic mesh products, one must subsequently consider the cost of the product itself. In light of equal clinical performance, the less costly product should be employed in cost-conscious health care systems.
It should also be noted that although there was a nonstatistical trend toward more total complications within the Strattice study arm (p = 0.06), this is likely a byproduct of data fragility. More specifically, despite this study being one of the largest published randomized trials on this topic (and the only one, to our knowledge directly comparing biologic meshes), occurrences of only a few more (or less) events within either patient study group would have potentially altered this finding. Although there is some anecdotal belief that the specific processing of a biologic mesh may affect subsequent liver transplantation graft function (and immunosuppression), the lack of observed statistical variance in complications between study groups aligns well with an absence of confirmed biologic rationale for any true difference.
Another interesting difference surrounds the observation that more patients in the Permacol study arm than in the Strattice study arm required a component separation strategy. Although this did not align with the observed similarity in hernia sizes between the groups, it potentially suggests that patients in the Permacol group had more technically challenging hernias. More specifically, it not only provides insight into a more difficult physical patient landscape (i.e., increased difficulty in achieving primary fascial midline re-approximation), but also a heightened risk of subsequent complications for these patients (i.e., increased risk of postoperative abdominal wall sepsis and morbidity).30 Our study shows these factors may suggest an enhanced clinical performance for Permacol given the reality of potentially more mechanically challenging reconstructions and associated complication risks among patients in this study group.
In observing statistical outcome equivalence between these 2 groups, this trial unsettles claims surrounding the relevance of cross-linking biologic mesh products. Industry marketing has stated that cross-linked products were superior in regards to long-term recurrence rates, whereas products that were not cross-linked were preferred in the shorter term within infected areas. Our study findings suggest that the importance of cross-linking may be overstated in the context of adequate patient preoperative optimization, correct case and product selection and nuanced surgical technique by the operating surgeon.
Limitations
This study has a number of limitations. We cannot comment on the efficacy of other commercial biologic mesh products (according to either cross-linking or performance), as the trial focused exclusively on 2 products. This is particularly relevant to the discussion surrounding indications for the use of biologic meshes in general. Second, because our study groups were limited to biologic mesh implantations, we are restricted in our data extraction to synthetic products. Third, we were unable to obtain formal wound culture data (bacterial and fungal) for all patients, as these diagnostic tests were limited to scenarios with clear clinical indications. Fourth, although the followup timeframe of this trial was significantly longer than most hernia studies within the literature, longer-term recurrences must be expected in coming years. To address this, we will continue to follow these patients beyond the current (median 2.2 years, IQR 119) postoperative interval and report our findings within the peer-reviewed literature. Fifth, there was a nonstatistical shift over the course of the study to using more retrorectus mesh implantation compared with onlay with transfascial fixation (both in the context of this randomized biologic mesh trial and in regards to general practice with synthetic mesh reconstructions distinct from this trial). This is an important consideration that may affect the rate of hernia recurrences in the coming years. Sixth, although the specific type of reconstruction did not lead to varying recurrence rates, this may also change over time. One would expect that bridging repairs, for example, would eventually fail more frequently than the other modalities. Seventh, formal stratification of randomization was omitted but may have reduced potential bias in a small randomized controlled trial. Finally, the concept of “data fragility” must be revisited. Despite this study being one of the largest within the literature on this topic, occurrences of only a few more (or less) events within either study arm could have affected the significance of our findings. This can be addressed only by much larger studies that offer data safety by way of sheer volume.
Conclusion
Our study shows 2 separate biologic mesh products appear to perform in a similar clinical manner despite increased technical challenges in one of the study arms. Given the similar performance, and the substantial cost difference, the lower price option appears safe, durable and preferable within cost-conscious health care systems. A larger, confirmatory randomized trial involving multiple centres is required to confirm findings that lightweight synthetic mesh products may perform adequately in some high-risk patients who currently use biologic meshes for complex abdominal wall reconstruction.
Footnotes
Competing interests: C.G. Ball is coeditor-in-chief of CJS; he was not involved in the review or decision to accept this manuscript for publication. No other competing interests were declared.
Contributors: C.G. Ball and M.J. Rosen designed the study. T.L. Eberle acquired the data, which A.W. Kirkpatrick and T. Stuleanu analyzed. C.G. Ball wrote the article, which T.L. Eberle, A.W. Kirkpatrick, M.J. Rosen and T. Stuleanu reviewed. All authors approve the final version for publication.
Funding: This study was partially funded through a ‘no oversight’ grant from Medtronic.
- Accepted June 13, 2022.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/