Abstract
Although inherited bleeding disorders (IBDs) affect both females and males, this review of the preoperative diagnosis and management of IBDs focuses on genetic and gynecologic screening, diagnosis and management of affected and carrier females. A PubMed literature search was conducted, and the peer-reviewed literature on IBDs was evaluated and summarized. Best-practice considerations for screening, diagnosis and management of IBDs in female adolescents and adults, with GRADE (Grading of Recommendations, Assessment, Development, and Evaluations) evidence level and ranking of recommendation strength, are presented. Health care providers need to increase their recognition of and support for female adolescents and adults with IBDs. Improved access to counselling, screening, testing and hemostatic management is also required. Patients should be educated and encouraged to report abnormal bleeding symptoms to their health care provider when they have a concern. It is hoped that this review of preoperative IBD diagnosis and management will enhance access to women-centred care to increase patients’ understanding of IBDs and decrease their risk of IBD-related morbidity and mortality.
The pathway to high-quality and safe care for people with inherited bleeding disorders (IBDs) continues to be challenging. Patients at risk may encounter obstacles in accessing appropriately resourced, high-quality comprehensive care centres for innovative evidence-based treatment.1 The IBDs that affect female surgical care include inherited von Willebrand disease (vWD) and X-linked hemophilia A and B carrier status, as well as rare platelet disorders.
Focused genetic and hematologic education related to IBDs is required for the surgical health care provider. Enhanced medical and nursing training curricula and public awareness are necessary as well.2,3
Although IBDs affect both female and male patients, this review focuses on preoperative surgical screening, diagnosis and management of affected or carrier females. The review provides an evidence-based diagnostic and genetic classification and management pathway to help provide a framework for reducing IBD morbidity in affected adolescent and adult females undergoing scheduled surgical procedures.
Methods
A scoping review methodology was used. The review was focused primarily on IBDs in adolescent and adult females and pregnant people with vWD, hemophilia A and B carrier status, and inherited platelet disorders. Keywords, report titles, peer-reviewed articles with reference review and author names were used in the search strategy. PubMed was searched for relevant articles in English with the keywords “von Willebrand disease,” “hemophilia A,” “hemophilia B,” “inherited platelet disorders,” “screening criteria,” “diagnostic criteria,” “genetic testing,” “laboratory testing,” “major surgery,” “minor surgery,” “pregnancy,” “menorrhagia” and “prophylaxis.” The scoping review identified adequate data sources for this evidence-based focused review of the topic.
Present health care status of females with inherited bleeding disorders
The impact of IBDs is substantial. The clinical issues of bleeding severity, determination of the patient’s individual risk related to surgery and pregnancy, disease transmission, testing and reproductive decisions contribute to health-related quality of life.4–17 Compared to females without IBDs, those with IBDs have lower values for bleeding control, vitality, and physical and social functioning, and increased pain (joint bleeding/heavy menstrual bleeding) and psychosocial problems (guilt, anxiety, depression, work and school absenteeism, isolation and difficult social relationships).4–17 Women’s occupational opportunities are substantially affected, related to the person’s disease severity and, in the case of familial hemophilia, the potential need to undertake a role as caregiver.4–17 Patient access to specialist care is commonly reported as an obstacle.4–17
Knowledge translation regarding male hemophilia A and hemophilia B outcomes is important for informed-consent counselling of female hemophilia carriers. Both hemophilia A and hemophilia B have transitioned from a disease of childhood to one of adulthood. Life expectancy has increased from 11 years in the mid-1900s to the present 55–63 years owing to management with coagulation factor concentrates. There are long-term clinical concerns associated with hemophilia outcomes, including chronic viral infections with liver disease, joint impairment with bone disease, cardiovascular disease, cancers, chronic kidney disease, covert cerebral microbleeds, impaired sexuality and development of immune factor inhibitors.18
Primary risk screening in female adolescents and adults with an abnormal bleeding history
Health care providers need to increase their recognition of and support for female adolescents and adults with IBDs. Improved access to counselling, screening, testing and hemostatic management is also required. Patients should be educated and encouraged to report abnormal bleeding symptoms to their health care provider when they have a concern.
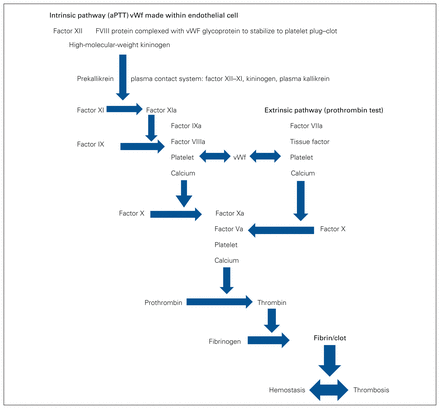
The genetic impact, definition and coagulation testing factors of IBDs affecting female adolescents and adults are summarized in Table 1.2,19–32 The coagulation cascade summarizes the factors and their interactions to achieve hemostasis–thrombosis balance33 (Figure 1).
Traditional coagulation cascade model.33 aPTT = activated partial thromboplastin time; vWF = von Willebrand factor.
Genetic impact, definition and coagulation testing of inherited bleeding disorders
Menorrhagia is the most common symptom reported by female adolescents and adults with IBDs. The clinical descriptions that indicate serious menorrhagia are soaking through menstrual pads or tampons within 1 hour, soaking through bedclothes, decreased ferritin level and anemia (microcytic; hemoglobin level < 10 g/dL) and, as an objective measurement, a score less than 100 on a pictorial bleeding assessment chart (PBAC).34
Health care providers must consider a bleeding disorder in adolescent females with bleeding problems, as 50% of patients have some form of coagulopathy.35,36 Many females diagnosed with an IBD as an adult will have been missed as an adolescent.
Evaluation
Evaluation of females with an IBD consists of the following elements:22,23
History and physical examination
♦ Current clinical status (characterize bleeding phenotype, complete a bleeding assessment tool)
♦ History of excessive bleeding (determine whether history is lifelong)
♦ History of joint pain/swelling (identify undiagnosed hemarthrosis)
♦ History of chronic diseases (identify nongenetic causes)
♦ Reproductive history (assess fertility)
Family history and pedigree
♦ Family history from reliable relatives (determine inheritance patterns)
♦ Laboratory results of affected relatives (determine severity in males)
Verbal review of the use of nonwarfarin and over-the-counter medications, as certain agents have an associated bleeding risk (Table 2).33
Medications and over-the-counter medicines that entail a risk of increased bleeding33
The screening criteria used to identify a possible diagnosis of IBD are as follows:34,35
Adolescents
♦ Menses lasting longer than 7 days, bleeding through a pad or tampon in 1 hour, clots greater than 3 cm in diameter, or bleeding that results in anemia or low iron status
♦ Bleeding requiring a transfusion
♦ Unmanageable heavy menstrual bleeding
♦ Prolonged bleeding from small wounds lasting more than 15 minutes or recurrent bleeding after 7 days
Adults: the above criteria as well as the following:
♦ History of heavy or prolonged bleeding after a procedure or surgical intervention (e.g., tooth extraction, surgery, delivery)
♦ Two or more of the following conditions: epistaxis once or twice per month, epistaxis lasting more than 10 minutes, frequently bleeding gums or family history of bleeding symptoms
♦ Unexpected postsurgical bleeding, history of hemorrhagic ovarian or corpus luteum cyst
♦ Failure of response to conventional hormonal management of menorrhagia (combined estrogen–progesterone or progesterone alone).
Validated tools are available for clinical bleeding history scoring, including a bleeding assessment tool (BAT) and a pictorial scoring system for pads and tampons (PBAC) used during bleeding episodes.36–40 The ISTHBAT (International Society on Thrombosis and Haemostasis – Bleeding Assessment Tool) comprises 14 categories for assessing bleeding symptoms retrospectively; a high bleeding score is associated with the presence of an IBD.37,38 The PBAC is a prospective scoring system developed as a semiquantitative evaluation of menstrual blood loss that considers the number of sanitary products used, the degree to which these products are soiled with blood, the number and size of blood clots passed, and the number of flooding episodes.39,40 These tools provide useful methods for establishing the amount of bleeding, which can then can be compared after therapy initiation.
Hematology and laboratory studies
Hematology screening of plasma should include the following elements:33–35
Complete blood count
Prothrombin time
Activated partial thromboplastin time
Factor VIII (FVIII) level decrease:
♦ vWD profile (rule out vWD)
♦ Factor V level (rule out factor V and FVIII deficiency)
♦ FVIII binding to von Willebrand factor (vWF) (rule out vWD type 2N)
♦ FVIII inhibitor (evaluate for possible acquired hemophilia)
Factor IX (FIX) level decrease: factor II, factor VII, factor X (rule out genetic or acquired combined vitamin K–dependent clotting factor deficiency)
FIX inhibitor (evaluate for possible acquired hemophilia)
von Willebrand panel (vWF activity–ristocetin cofactor test, vWF antigen level, FVIII level)
Fibrinogen level
Possible platelet function and/or aggregation studies, and thyroid function testing if the above screening gives normal results (normal platelet count 150–450 000 × 109/L; normal values in Alberta: thyroid-stimulating hormone level 0.20–6.50 mIU/L, free T4 level 10.0–25.0 pmol/L, free T3 level [age ≥ 18 yr] 3.0–6.5 pmol/L)
A hematology consultation may be required based on the laboratory screening results.
Additional genetic studies after identification of primary etiology20,23,26
Paternity testing (rule out nonpaternity)
Karyotype with high resolution of X (identify X chromosome abnormality)
Sequencing of FVIII and FIX (identify point mutations)
Multiplex ligation-dependent probe amplification (identify gene deletion or duplication)
X chromosome inactivation studies (identify nonrandom inactivation).
Common etiologies and diagnoses identified in females with inherited bleeding disorders
von Willebrand disease2,19,20,41–44
von Willebrand disease is the single most common IBD, with a laboratory risk-defined prevalence of 1% and a symptomatic prevalence of 0.1%. Autosomal dominant and recessive inheritance patterns are dependent on the vWD classification (Table 1).
The classical vWD triad association consists of mucocutaneous bleeding, family history of bleeding and a laboratory finding of a vWF deficiency. Directed questioning for pregnancy-related bleeding, epistaxis and heavy menstrual periods should be considered. The most commonly reported symptoms are heavy menstrual bleeding (75%–100% of patients), excessive bruising (60%–80%), oropharyngeal bleeding (64%), epistaxis (56%) and bleeding after dental surgery (24%–26%).44
In vWD, the bleeding tendency is due primarily to the inherited deficiency or dysfunction of the multimeric glycoprotein vWF, causing abnormal platelet–vessel wall interactions and defective formation of the platelet plug (primary hemostasis).
Diagnosis
There are pre-analytical variables that can affect the result of screening for an increased vWF level, such as delays in transportation or processing, estrogen level, pregnancy, physical or mental stress, and other medical conditions. Testing is required to evaluate quantitative and qualitative “diagnostic” results based on specific laboratory controls. The screening and diagnostic tests are prothrombin time, activated partial thromboplastin time, FVIII coagulant activity, ristocetin cofactor activity and vWF antigen concentration.2,19,20,41,42
Pregnancy is a prothrombotic physiologic event, and the hyperestrogen environment can increase vWF and FVIII levels by up to 300%–400%. These physiologic changes may minimize potential bleeding events, but pregnant people with vWD still have an increased risk for peripartum hemorrhage and the need for red cell transfusion. In addition, there is an increased risk of recurrent miscarriage.43
X-linked hemophilia A and B carrier status
Hemophilia is an X-linked IBD, and females can be mutation carriers with variable levels of FVIII (hemophilia A) or FIX (hemophilia B). Female hemophilia carriers can be classified as obligate (daughters of affected male, women with > 1 affected son, women with 1 affected son and an affected brother or uncle) or possible (women with 1 affected son and no family history, daughters of an obligate carrier and woman with a maternal family history of hemophilia). A de novo germ cell X chromosome mutation is found in 30%–50% of newborn boys with hemophilia21–26,45–47 (Table 1).
There are important characteristic differences between hemophilia A and B, such as hemostatic functional impact (cofactor in hemophilia A, enzyme in hemophilia B), genetic variant impact (null variant in 80% of cases of hemophilia A, missense variant in > 60% of hemophilia B cases) and risk of development of severe treatment-related inhibition of coagulation (in 35%–40% of cases of hemophilia A and 4%–5% of cases of hemophilia B).46
Genetic mechanisms explain most of the reported clinical variability. Possible causes of FVIII and FIX deficiency in women and girls are23,26,48–51
Homozygosity (2 identical hemophilia alleles) by consanguinity, chance or de novo mutation(s)
Compound heterozygosity (2 different hemophilia alleles) due to unrelated hemophilia families, de novo second mutation or 2 de novo mutations
Hemizygosity (1 hemophilia allele and 1 normal allele) by single X chromosome 45,X/46,XY mosaicism with complete androgen insensitivity or complete gonadal dysgenesis, or X chromosome deletion of the FVIII or FIX gene
Heterozygosity (1 hemophilia allele, 1 normal allele) by inheritance in hemophilia-affected family, or 1 new mutation followed by skewed X inactivation (unknown or random), preferential X inactivation due to X chromosome abnormality, cell viability disorder or specific allele of the X-inactive-specific transcript gene, or other inherited skewed X inactivation
Other genetic causes: vWD, particularly type 2N *autosomal recessive or dominant; factor V and FVIII deficiency (autosomal recessive); combined vitamin K–dependent clotting factor (II, VII, IX, XI) deficiency (autosomal recessive)
Nongenetic cause: acquired hemophilia due to FVIII or FIX inhibitors, or vitamin K deficiency.
Diagnosis
Female hemophilia A carriers have an FVIII deficiency, mild in 94% of cases, moderate in 3% and severe in 3%. The circulating plasma FVIII concentration observed in hemophilia A carriers ranges from normal to levels close to those of males with hemophilia, with a median level of 55% of normal (range 4%–136%).52
Female hemophilia B carriers have an FIX deficiency. The median FIX level is 45% of normal (range 19%–114%); 30% of patients have levels less than 40% of normal. The data for hemophilia B carrier morbidity are limited compared to those for hemophilia A carrier status.25
A new nomenclature for female hemophilia carriers aimed at improving diagnosis and management has been defined.53 It distinguishes 5 clinically relevant categories: women/girls with mild, moderate or severe hemophilia (FVIII/FIX level > 0.05 IU/mL and < 0.40 IU/mL, 0.01–0.05 IU/mL and < 0.01 IU/mL, respectively) and symptomatic and asymptomatic hemophilia carrier status (FVIII/FIX level ≥ 0.40 IU/mL with and without a bleeding phenotype, respectively). With the new nomenclature, women attending US hemophilia treatment centres were identified as having mild (FVIII 16.0%, FIX 23.7%), moderate (FVIII 1.8%, FIX 0.86%) or severe (FVIII 0.49%, FIX 0.42%) hemophilia.23 The proportion of women receiving hemophilia treatment in each category was 17.8%, 1.4% and 0.5%, respectively.
Inherited platelet disorders
Inherited platelet disorders consist of a broad spectrum of medical conditions that include quantitative platelet disorders, which can present as increased or decreased platelet numbers, and qualitative platelet disorders, which affect platelet function. Inherited functional platelet disorders are rare, are commonly autosomal recessive and present usually in consanguineous populations29–31,54–57 (Table 1). A history of mucocutaneous bleeding or periprocedural hemorrhage with no other secondary etiology or family history should raise clinical suspicion of an inherited platelet disorder.
An important distinction is that acquired disorders of platelet function are more common than inherited disorders. Antiplatelet drugs are the most common etiology of acquired disorders. Clinical conditions associated with acquired platelet dysfunction are uremia, hepatic cirrhosis, myeloma, polycythemia vera and essential thrombocythemia.58
Diagnosis
The diagnosis is challenging and commonly delayed. The defects in platelet aggregation and activation can be from platelet receptors, platelet signalling, platelet granule release, platelet cytoskeletal remodelling or platelet hematopoiesis. Molecular studies have identified up to 40 genes as being causative of inherited thrombocytopenia, but in 50% of cases, no genetic basis has been identified.31 Whole blood electronic counters are used to evaluate platelet count and size, and a blood smear (with or without staining technology) is also required. For platelet function assessment, the classic testing, such as aggregometry, secretion assays, flow cytometry, Western blotting and electron microscopy, is time consuming and expensive, and interpretation of the results is complex.31
Quantitative platelet disorders associated with an inherited thrombocytopenia etiology are a large heterogeneous group of rare platelet disorders characterized by low platelet count, leading to bleeding and syndromic and systemic manifestations. The prevalence is estimated at 1 per 100 000, but many patients are undiagnosed.30
Inherited qualitative (functional) platelet disorders affect normal platelet function at different stages of initial hemostasis: adhesion (Bernard–Soulier syndrome), activation, granule secretion (Hermansky–Pudlak syndrome, Quebec platelet disorder), signal transduction, aggregation for platelet thrombus formation (Glanzmann thrombasthenia) and coagulation factor activation (Scott syndrome).30
Coagulation cascade and the drugs, medications and blood products used in management
Understanding the coagulation cascade is important for IBD care in order to implement individualized management choices. Figure 1 summarizes the intrinsic and extrinsic systems that result in the development of fibrin-based clotting, with normal balance between the thrombosis and hemostasis states.
Female adolescents and adults with IBDs have a spectrum of bleeding risk based on the genetics and physiology of the coagulation factor imbalance. The deficiency factors include vWF, coagulation factors VIII, IX, XI, VII, XIII and V, and platelet abnormalities. These deficient coagulation factors can be harvested from donated human plasma sources, through technology for recombinant products and with specific HLA platelet selection techniques.2,33,59
The products used for deficiency replacement are as follows: FVIII, recombinant FVIII concentrate; FIX, recombinant FIX concentrate; plasma-derived factors V, VII, XI, XIII concentrate; fresh frozen plasma (can be used if factor XI concentrate is unavailable, as it contains all the coagulation factors); vWF concentrate; tranexamic acid; and desmopressin (Table 3).2,59
Traditional antithrombotic therapies (anticoagulation, antiplatelet) may be required in females with IBDs needing other surgical and medical treatments.2,55–57,59–63 The available anticoagulation drugs are used to reduce the risk of thrombosis but result in a concomitantly increased risk of bleeding (Table 4).61,62 Although perioperative management of patients who are receiving antiplatelet therapy is a common clinical occurrence, there are minimal high-quality studies to direct, continue or interrupt antiplatelet therapy, as well as concerning the role of bridging therapy.63
Summary of traditional antithrombotic therapies for use in elective surgical management of inherited bleeding disorders*
Management
Management strategies for IBDs must consider both episodic and preventive approaches for patient care. Compared to the general population, patients with IBDs are at 1.5 and 5 times greater risk for hemorrhage and transfusion, respectively.64–67 Short-term prophylaxis is warranted mainly to provide effective hemostatic coverage to patients undergoing surgery or invasive procedures, at the time of labour and delivery, or with recurrent excessive menstrual bleeding. Long-term prophylaxis is used to prevent bleeding in patients at increased risk for frequent and spontaneous bleeding in the joints, nose or gastrointestinal tract (Table 3).2,59
Menorrhagia
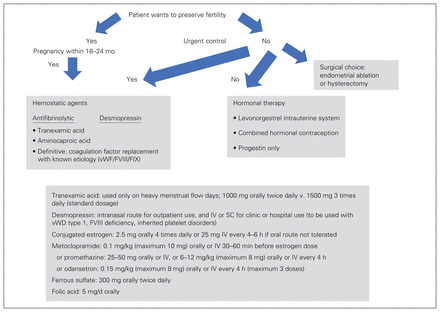
The initial clinical presentation of an IBD in female adolescents and adults is commonly associated with menorrhagia. The primary management pathway and drug or replacement products that may be required before diagnosis of an IBD etiology are summarized in Figure 234 and Table 3.2,59 Educational counselling is required for the informed-consent process. With an IBD diagnosis, discussion of treatment and choice needs to include the patient’s long-term fertility choice, contraception needs, IBD etiology, bleeding severity, and social or religious factors.
Decision guide for acute heavy menstrual bleeding (defined as any menstrual bleeding that interferes with daily activity [work, housework, leisure or sport, social activity] during most menstrual periods; bleeding is significant for any 1 of these scenarios: changing pads within 2 h, bleeding lasting ≥ 7 d, clot size > 1 cm with “flooding,” pictorial blood assessment chart score > 100).34 F = factor; IV = intravenously; SC = subcutaneously; vWD = von Willebrand disease; vWF = von Willebrand factor.
Hormonal therapy is indicated for women who want to preserve their fertility options but are not planning an immediate pregnancy (within 12–18 mo). Evidence-based assessment of hormonal therapy in patients with IBDs is limited (Table 5).68 Additional risk counselling may be required, as some women with IBDs have an increased risk of thromboembolic events (afibrinogenemia), and factor replacement may be required for women with a severe bleeding phenotype if they do not respond to medical therapy.
Evidence-based assessment of hormonal therapy in patients with inherited bleeding disorders68
von Willebrand disease
Surgical prophylaxis is required for major and minor surgical procedures in patients with vWD.44 The American Society of Hematology, the International Society on Thrombosis and Haemostasis, the National Hemophilia Foundation and the World Federation of Hemophilia formed a multidisciplinary guideline panel to establish evidence-based guidelines intended to support patients, clinicians and health care professionals in their decisions about management of vWD. Following are their clinical consensus-based recommendations for vWD management.69
Prophylaxis therapy
Recommendation 1: in patients with vWD with a history of severe and frequent bleeds, the panel suggests the use of long-term prophylaxis rather than no prophylaxis.
Desmopressin challenge (trial and administration):
Recommendation 2A: desmopressin is a valid treatment option for patients with vWD type 1 with a baseline vWF level less than 0.30 IU/mL. The panel suggests a trial of desmopressin and treating with desmopressin based on the results over not performing a trial and treating with tranexamic acid or coagulation factor concentrate.
Recommendation 2B: for these patients, the panel suggests against treating with desmopressin in the absence of desmopressin trial results.
Antithrombotic therapy (as required)
Recommendation 3: in patients with vWD and cardiovascular disease who require treatment with antiplatelet agents or anticoagulant therapy, the panel suggests giving the necessary therapy over no treatment.
Recommendation 6A: the panel suggests using either hormonal therapy (combined hormonal contraception) or levonorgestrel-releasing intrauterine system), or tranexamic acid over desmopressin to treat women with heavy menstrual bleeding who do not wish to conceive.
Recommendation 6B: the panel suggests using tranexamic acid over desmopressin to treat women with heavy menstrual bleeding who wish to conceive.
Major surgery
Recommendation 4A: the panel suggests targeting both FVIII and vWF activity levels of 0.50 IU/mL or greater for at least 3 days after surgery.
Recommendation 4B: the panel suggests against using only FVIII level of 0.50 IU/mL or greater as a target level for at least 3 days after surgery.
General surgical options are appropriate for women with IBD who do not want to preserve their fertility options. Multidisciplinary counselling should be used for informed consent and choice. Endometrial ablation is a minimally invasive procedure that is appropriate for women with no uterine or pelvic disorders, as it reduces blood loss substantially in women with IBDs. Hysterectomy provides a definitive surgical treatment for women with IBDs and heavy menstrual bleeding, or uterine or pelvic disorders. There is an operative and anesthesia risk, estimated at 1%–3% for hysterectomy. Women with IBDs are at increased risk for perioperative bleeding, subsequent red cell transfusion and potential interventional radiology. This hemorrhagic complication risk is important to consider as part of the informed-choice process (Figure 2).34
Minor surgery (invasive procedures)
Recommendation 5A: in patients undergoing minor surgery or minor invasive procedures, the panel suggests increasing vWF activity levels to 0.50 IU/mL or greater with desmopressin or factor concentrate with the addition of tranexamic acid over raising vWF levels to 0.50 IU/mL or greater with desmopressin or factor concentrate alone.
Recommendation 5B: in patients with vWD type 1 with baseline vWF activity levels greater than 0.30 IU/mL and a mild bleeding phenotype who are undergoing minor dental mucosal procedures, the panel suggests giving tranexamic acid alone over increasing vWF activity levels to 0.50 IU/mL or greater with any intervention.
X-linked hemophilia A and B carrier status
Many female hemophilia A carriers are at risk for abnormal bleeding, but these carriers are underrecognized by health care providers, and their bleeding symptoms are underreported.70 Low FVIII levels are consistently associated with clinically significant bleeding and correlate with female skewed X chromosome inactivation. Bleeding can occur in some hemophilia A carriers with normal FVIII levels.70 Chaudhury and colleagues45 reported that females with mild hemophilia (FVIII/FIX) and a bleeding phenotype (traumatic/joint/clinical bleeding) were managed with factor concentrates or antifibrinolytics, but heavy menstrual bleeding was more difficult to control.
Nongynecologic bleeding occurs in female hemophilia carriers owing to the genetic mechanisms described, and their bleeding management may be similar to that for males with hemophilia. Prophylactic management for affected female carriers minimizes bleeding risk and morbidity in joints and the central nervous system (increased with age > 60 yr and hypertension). Prophylactic management should consist of replacement of the appropriate coagulation factor after the diagnosis has been identified and an individualized prophylaxis regimen has been determined based on the patient’s peak–trough FVIII levels. An accessible app (WAPPS-hemo) is available to assist in managing the patient’s regimen.54 The prophylactic use of noncoagulation factor therapy (emicizumab) can be considered as more evidence accrues.52
Inherited platelet disorders
Potential curative treatments for inherited platelet disorders include allogenic hematopoietic stem cell transplantation and gene therapy.
Technologic diagnostic improvements have not been translated into clinical management. Therapies to increase platelet count, mitigate platelet dysfunction and definitively correct inherited thrombocytopenia continue to be used.30,31,55
Outpatient management29,71
As a first-line option, initial control will be obtained with desmopressin in 40% of cases and with an oral contraceptive pill in 30%.71 At 2 years, control will be achieved with an oral contraceptive pill or desmopressin in 50% of cases.71 Additional options for the 50% of cases not controlled by either of these medications include
Nonhormonal therapy: specific factor replacement, antifibrinolytic agent (tranexamic acid or aminocaproic acid) or desmopressin
Hormonal therapy: combined estrogen–progestin product or progestin only
Combination depends on the patient’s contraception needs, whether an estrogen product is contraindicated, the patient’s ability to tolerate an estrogen product, and major adverse effects or sensitivities (there is no real risk in using an antifibrinolytic with estrogen).
Directed procedural/surgical management via bleeding risk assessment31
Prevention/prophylaxis of mild to moderate inherited platelet disorder bleeding
♦ Topical measure: compression with gelatin sponges for wounds, epistaxis and bleeding after dental surgery
♦ Antifibrinolytic drugs: treatment before procedure and for 3–5 days afterward: 1) tranexamic acid, 15–25 mg/kg orally every 8 hours; 2) desmopressin, 150 μg/dose intranasally, 0.3 μg/kg subcutaneously or 0.2–0.3 μg/kg in saline (4 μg/mL) intravenously over 30 minutes starting 1 hour before procedure.
Prevention of moderate to severe inherited platelet disorder bleeding
♦ Antifibrinolytic drugs: severe bleeding, tranexamic acid, 10 mg/kg intravenously every 8 hours
♦ Platelet transfusion: essential; leukocyte-depleted platelet concentrate is preferable; recommended preprocedure platelet count is greater than 30 × 109/L for dental or minor surgery, greater than 50–80 × 109/L for major surgery or vaginal or cesarean delivery, and greater than 100 × 109/L for eye or central nervous system surgery
♦ Recombinant active factor VII: approved for Glanzmann thrombasthenia; off label for Bernard–Soulier syndrome; potentially useful in combination with antifibrinolytics for control of bleeding in childbirth in women with severe platelet dysfunction; 90–120 μg/kg intravenously before the procedure and then every 90–120 minutes until hemostatic control is attained.
Pregnancy
Obstetric management for women with vWD, hemophilia carriers and those with inherited platelet disorders requires counselling (preferably before conception), choice, informed consent and multidisciplinary care providers (Table 3,2,59 Table 6,2,40,59,72–81 Table 7,2,40,59,72–81 Table 842,82–90). There are substantial hemostatic changes in a normal pregnancy whose role is to preserve placental function and prevent excessive bleeding at delivery.63
Preconception or intrapartum counselling and informed consent for female hemophilia A and B carriers
An individualized protocol for the management of labour and delivery care in women with IBDs is required based on the identified individualized maternal and fetal risk (Table 3, Tables 6–⇑8). Factor VIII levels are not a good predictor of bleeding risk in hemophilia A carriers, as there are no significant differences in FVIII levels for heterozygous carriers of mutations classified as mild, moderate and severe for hemophilia A.77,78
Prenatal diagnosis techniques may be required for fetal assessment in women with IBDs after routine serum-based aneuploidy screening. No evidence-based series to determine invasive procedure risks for pregnant people with IBDs were identified. Prenatal diagnosis procedures have been used for maternal management decisions in 50% of female hemophilia carriers.72,91 In patients with vWD, early genetic testing of the fetus is not routinely recommended owing to gene size and molecular testing, as well as the lack of clinical implication (other than for delivery and neonatal risk), except for couples with a child with autosomal recessive vWD type 3.72 These couples should have preconception or early pregnancy counselling regarding prenatal testing. Pregnant people with severe autosomal dominant vWD have a 50% chance of having an affected child. For patients with vWD type 1–3, amniocentesis testing at 30–32 weeks can be considered to identify significant fetal risk for labour and delivery management.72
Preconception counselling of female hemophilia carriers (obligate or highly possible) is recommended, if possible, to identify the causative mutation, measure FVIII and FIX levels, provide information for the management of pregnancy and delivery, and discuss the pregnant person’s options and choice in the case of an affected male fetus. Prenatal diagnosis techniques (chorionic villus sampling, amniocentesis, plasma cell-free placental DNA screening [noninvasive prenatal screening]) and the availability of assisted reproductive technology are options to inform hemophilia carriers’ reproductive choice.92 Plasma cell-free placental DNA screening for a Y chromosome via Y-specific polymerase chain reaction is an early prenatal screening option. If a male fetus is identified, chorionic villus sampling to test for the hemophilia mutation can be done. This procedure is normally done at 10–14 weeks’ gestation, via transcervical or transabdominal techniques under ultrasonography guidance. Vaginal bleeding or fetal loss may occur (rate 0.5%–3%). Placental tissue has a 1%–2% biologic risk of confined placental mosaicism.
Second-trimester screening with ultrasonography will identify the gender of the fetus; if male, amniocentesis under ultrasonography guidance to test for the hemophilia mutation can be done.12,60,72 This procedure is normally performed at 15–18 weeks but can be done into the third trimester. Amniotic fluid, with amniocyte cells, is used for testing. Procedural risks include minimal bleeding risk, fetal loss (< 1%) and premature rupture of membranes (2%–3%). Direct studies are carried out with the focus on identifying the responsible mutation (intron 22 inversion) in the carrier’s FVIII gene through full sequencing of the gene. The intron 22 inversion mutation of FVIII accounts for 45% of mutations associated with severe hemophilia, and large deletions and duplications are present in 6% of cases; complex rearrangements involving both inversion and deletion/duplication and mosaicism for Inv22 are rare. If the hemophilia mutation testing is not predicted to be severe, the FVIII gene can be sequenced directly. Indirect studies are important if the carrier’s gene testing does not detect a mutation. The FVIII gene is examined for normal variants or polymorphisms within the family to identify which X chromosome is associated with the family hemophilia. Indirect studies always complement direct studies.
In noninvasive prenatal screening, placental cell-free DNA present in the maternal plasma is used for fetal testing (Y chromosome sex; X chromosome analysis). If assisted reproductive technology is used, preimplantation genetic diagnosis can be performed (blastomere or trophectoderm biopsy, with fluorescence in situ hybridization, polymerase chain reaction or next-generation sequencing).92 Pregnancy outcomes are facility specific, but generic results indicate delivery in 18% of oocyte retrieval procedures and 25% per embryo transfer procedure.93
Directed prelabour anesthesia consultations are recommended for management of regional anesthesia block in labour, with individualized risk assessment and planning. Treatment options of desmopressin acetate or recombinant vWF/FVIII Haemate P (intermediate-purity vWF/FVIII concentrate) (CSL Behring) should normalize coagulation factors and decrease maternal bleeding risk in patients with vWD.94–99
Regarding neuraxial anesthesia, the panel recommends the following:69
Recommendation 7: in women with vWD for whom neuraxial anesthesia during labour is deemed suitable, the panel suggests targeting a vWF activity level of 0.50–1.50 IU/mL over targeting an activity level of more than 1.50 IU/mL to allow neuraxial anesthesia.
Directed obstetrical delivery management for fetal vWD type 1–3 and hemophilia A and B risk (using a stratification process for estimated obstetric bleeding risk levels of mild, moderate and high [Table 3, Tables 6–⇑8]) suggests that, in pregnant people with associated mild or unlikely risk, a recommendation for avoidance of ventouse use and external cephalic version, and selected use of rotational forceps, fetal scalp blood sampling and fetal scalp electrodes should be considered.2,40,59,72–81 In those with associated moderate risk, care providers should consider a recommendation for avoidance of use of midcavity or rotational forceps, ventouse use procedure and external cephalic version, and selected use of fetal scalp blood sampling and fetal scalp electrodes. In those with associated high risk, a recommendation for avoidance of use of all forceps, external cephalic version, ventouse procedure, fetal scalp blood sampling and fetal scalp electrodes should be considered.
Regarding the postpartum period, the panel recommends the following:69
Recommendation 8: the panel suggests the use of tranexamic acid over not using tranexamic acid in women with vWD type 1 or low vWF levels (which may apply to vWD type 2 and 3) during the postpartum period.
Neonatal management has identified that the vWF level is raised at birth, but neonates with severe vWD type 1 and vWD types 2 and 3 have continued low vWF and are at risk for bleeding complications.72 Prenatal risk assessment with amniocentesis at 30–32 weeks can be considered for optimal neonatal planning.72 Cord blood testing for FVIII and vWF antigen and activity is recommended. Cranial imaging and short-term prophylaxis with factor concentrate should be considered neonates with vWD type 3, especially with a history of traumatic delivery. Oral vitamin K administration is recommended for neonates with vWD type 2 or 3. The smallest-gauge needles should be used for later newborn vaccination dosing. The risks and issues for the neonate and during the first year of life should be reviewed with the parents before discharge.2,59,72
Female hemophilia B carriers do not exhibit the hemostatic acute phase reactant elevations with FVIII, and this carrier state may be associated with increased risk of postpartum hemorrhage.25 Postpartum hemorrhage rates have been found to be higher for carriers with a severe hemophilia mutation (54%) compared to those with a mild mutation (29%).78–81
Best-practice considerations
Best-practice considerations for screening, diagnosis and management of IBDs in female adolescents and adults are presented in Table 9.
Best-practice considerations for screening, diagnosis and management of inherited bleeding disorders in female adolescents and adults
New management considerations
With the management goal of treating the primary deficiency in vWD — vWF — the novel approach of replacing only vWF was introduced about a decade ago. After the manufacture of a concentrate fractionated from human plasma and another obtained through recombinant DNA technology, clinical trials showed that vWF products correct not only the primary vWF deficiency, but also the secondary FVIII coagulant activity deficiency safely and effectively in surgical cohorts.100 Other clinical areas, such as pediatrics, long-term prophylaxis and recurrent gastrointestinal bleeding due to angiodysplasia, have not been as well researched.
There have been major advancements in management and treatment options for X-linked hemophilia, with prolonged half-life FVIII and FIX concentrates, nonfactor therapies (emicizumab, anti-tissue factor pathway inhibitor antibodies, siRNA targeting of antithrombin) and gene therapy. Fassel and McGuinn101 reported that these new molecules significantly reduced the burden of hemophilia and improved the quality of life for patients with severe disease. Prophylaxis with FVIII replacement remains the standard of care for male hemophilia A, as nonreplacement products such as prophylactic emicizumab, given subcutaneously every 4 weeks, are not suitable for acute bleeding episodes or management in major surgery.80,81,102
Additional treatments to increase platelet count stability, transiently before surgery or via invasive interventions, are57
Splenectomy: for use in Wiskott–Aldrich syndrome or X-linked thrombocytopenia only; may reduce bleeding but increase immunodeficiency and rate of severe infections
Eltrombopag/romiplostim: recommended for use in Wiskott–Aldrich syndrome, MYH9-related disease and thrombocytopenia related to the ankyrin repeat domain 26 gene only.
Limitations
Limitations of this scoping review are the quality and quantity of the subject-based publications sorted for review, as the selection and elimination of the publications were done by a single author.
Conclusion
Health care providers need to increase their recognition of and support for female adolescents and adults with IBDs. Improved access to counselling, screening, testing and hemostatic management is also required. Patients should be educated and encouraged to report abnormal bleeding symptoms to their health care provider when they have a concern. It is hoped that this review of preoperative IBD diagnosis and management will enhance access to women-centred care to increase patients’ understanding of IBDs and decrease their risk of IBD-related morbidity and mortality.
Footnotes
Competing interests: The author reports travel support to attend Society of Obstetricians and Gynaecologists of Canada (SOGC) meetings. He is the 2022–2023 SOGC president. No other competing interests were declared.
- Accepted January 18, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- 1.↵
- 2.↵
- 3.↵
- 4.↵
- 5.
- 6.
- 7.
- 8.
- 9.
- 10.
- 11.
- 12.↵
- 13.
- 14.
- 15.
- 16.
- 17.↵
- 18.↵
- 19.↵
- 20.↵
- 21.↵
- 22.↵
- 23.↵
- 24.
- 25.↵
- 26.↵
- 27.
- 28.
- 29.↵
- 30.↵
- 31.↵
- 32.↵
- 33.↵
- 34.↵
- 35.↵
- 36.↵
- 37.↵
- 38.↵
- 39.↵
- 40.↵
- 41.↵
- 42.↵
- 43.↵
- 44.↵
- 45.↵
- 46.↵
- 47.↵
- 48.↵
- 49.
- 50.
- 51.↵
- 52.↵
- 53.↵
- 54.↵
- 55.↵
- 56.
- 57.↵
- 58.↵
- 59.↵
- 60.↵
- 61.↵
- 62.↵
- 63.↵
- 64.↵
- 65.
- 66.
- 67.↵
- 68.↵
- 69.↵
- 70.↵
- 71.↵
- 72.↵
- 73.
- 74.
- 75.
- 76.
- 77.↵
- 78.↵
- 79.
- 80.↵
- 81.↵
- 82.↵
- 83.
- 84.
- 85.
- 86.
- 87.
- 88.
- 89.
- 90.↵
- 91.↵
- 92.↵
- 93.↵
- 94.↵
- 95.
- 96.
- 96.
- 98.
- 99.↵
- 100.↵
- 101.↵
- 102.↵