Abstract
Fat embolism syndrome, an important contributor to the development of acute respiratory distress syndrome, has been associated with both traumatic and nontraumatic disorders. Fat embolization after long bone trauma is probably common as a subclinical event. Fat emboli can deform and pass through the lungs, resulting in systemic embolization, most commonly to the brain and kidneys. The diagnosis of fat embolism syndrome is based on the patient’s history, supported by clinical signs of pulmonary, cerebral and cutaneous dysfunction and confirmed by the demonstration of arterial hypoxemia in the absence of other disorders. Treatment of fat embolism syndrome consists of general supportive measures, including splinting, maintenance of fluid and electrolyte balance and the administration of oxygen. Endotracheal intubation and mechanical ventilatory assistance can be indicated. The role of corticosteroids remains controversial. Early stabilization of long bone fractures has been shown to decrease the incidence of pulmonary complications. Clinical and experimental studies suggest that the exact method of fracture fixation plays a minor role in the development of pulmonary dysfunction. As more is learned about the specifics of the various triggers for the development of fat embolism syndrome, it is hoped that the prospect of more specific therapy for the prevention and treatment of this disorder will become a reality.
The fat embolism syndrome was initially recognized in the 1860s and was first described clinically in 1873. Since then, the syndrome has been observed in association with both traumatic disorders, such as long bone trauma and reaming of the medullary canals of the long bones, and a variety of nontraumatic disorders, including hemoglobinopathy, collagen disease, diabetes, burns, severe infection, neoplasms, osteomyelitis, blood transfusion, cardiopulmonary bypass, decompression from altitude, suction lipectomy and renal transplantation.1 Fat embolism can be an important contributing factor in the development of the acute respiratory distress syndrome.
Prevalence of fat embolism
The exact prevalence of fat embolism is uncertain. An autopsy study of 60 people who died of battle wounds in the Second World War revealed a 65% incidence of fat embolism. 1 Fat emboli were found in 855 of 6250 civilian accident victims.1 Fat embolism syndrome is most frequently seen clinically in association with multiple long bone fractures. The clinical prevalence of fat embolism syndrome in patients who have fractures has been reported to range from 0.25% (of 4530 patients) to 1.25% (of 7701 patients).2 The syndrome was found in nearly 10% of patients with multiple skeletal fractures associated with unstable pelvic injuries. 1 It must be emphasized that the phenomenon of fat embolization after long bone trauma is probably very common as a subclinical event. However, fat embolism is less likely in children, presumably because of the relative preponderance of hematopoietic elements in their bone marrow. In recent years fat embolism has been reported with increasing frequency after hip and knee replacement,3–5 and Russell, Kirk and Biddinger6 reported a case after an isolated humeral fracture.
Pathogenesis
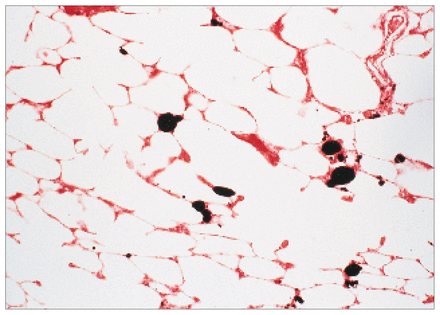
Most investigators believe that the source of embolic fat after trauma is the medullary contents of the long bones (Fig. 1).1 Bone marrow elements in the lung associated with fat deposition have been observed.1 Continued motion at the site of the fracture after the initial injury is thought to cause further intravasation of marrow fat.1 Alterations in lipid stability may also occur in the presence or absence of trauma and contribute to the development of emboli.1 Plasma-derived agglutination of chylomicrons is the likely cause of fat embolism in nontraumatic inflammatory conditions such as pancreatitis.7 The pulmonary manifestations of the syndrome have been theorized to coincide with degradation of fat by lipoprotein lipase to toxic free fatty acids.7
Photomicrograph of a cross-section of the lung, demonstrating multiple fat emboli in the pulmonary arterial vasculature (osmium tetroxide stain, original magnification × 100).
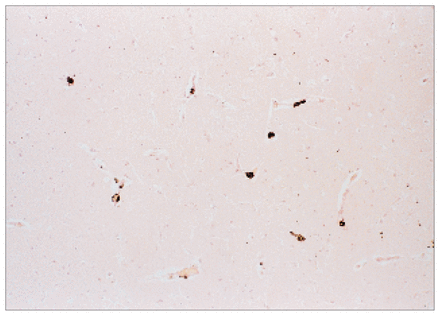
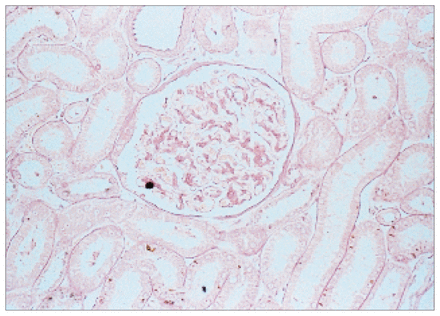
The distribution of fat emboli as they travel through the lungs and into the systemic circulation depends on the distribution of cardiac output, making the brain and kidneys more likely to receive emboli (Figs. 2 and 3). Fat emboli can form and pass through the lungs over time. Byrick and associates used a bilateral cemented arthroplasty model in anesthetized mongrel dogs to study transpulmonary systemic fat embolism. 8 By quantitative morphometry they demonstrated a decrease in the size of the pulmonary vessels occluded by fat from 12.8 μm 1 minute after embolic showering to 4.9 μm 2 hours after embolic showering. They found intravascular fat in all brain, heart and kidney specimens examined 3 hours after embolic showering. Radiolabelled microspheres 15 μm in diameter did not reach the systemic circulation. Byrick and associates concluded that intravascular fat was probably deformable, allowing it to traverse the pulmonary vasculature and that this may have accounted for the difference in distribution between the intravascular fat and the microspheres. The pathophysiologic effects of fat emboli are due to the hemodynamic changes they produce (in particular, elevated pulmonary artery pressure), impairment of pulmonary gas exchange and the effect of systemic fat embolization on the brain and other tissues (Fig. 4).
Photomicrograph of a cross-section of the cerebral cortex, demonstrating microarteriolar fat emboli (osmium tetroxide stain, original magnification × 200).
Photomicrograph of a cross-section of the kidney, demonstrating a fat embolus in a glomerular arteriole (osmium tetroxide stain, original magnification × 500).
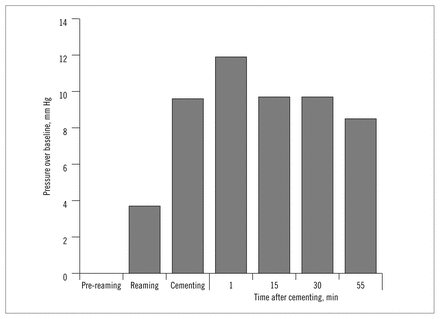
Response in a canine model (14 dogs) of pulmonary artery pressure to an embolic shower of medullary contents as a result of reaming, cementing and pressurizing the medullary canal. Subsequent experiments demonstrated that pulmonary artery pressure returns to baseline levels within 4 hours of embolic showering.
The interrelationship between fat embolism and the development of adult respiratory distress syndrome (ARDS) is currently an area of active investigation. ARDS is usually related to a direct or an indirect thoracic injury. The syndrome is recognized as resulting from infection, aspiration, multiple transfusions, intensive resuscitation with fluids, fracture, pulmonary contusion, toxic inhalation or a combination of these conditions.9 Because of inconclusive clinical evidence that intramedullary reaming potentiates the development of ARDS, this theory is being studied in animal models.
Byrick and associates found little inflammatory reaction to the presence of intravascular pulmonary fat.8 Because pulmonary fat embolization is probably very common and the prevalence of fat embolism syndrome is relatively low, there may be other factors involved in the genesis of the condition. Multiple trauma activates a systemic inflammatory response that produces elevated levels of mediators such as fibrinogen, tissue thromboplastin, prostacyclins, cytokines and elastase. Wozasek and colleagues studied the pathophysiologic effect of intramedullary reaming of sheep on their pulmonary function.10 Intramedullary nailing caused an increase in pulmonary arterial pressure but did not increase capillary permeability. Pulmonary dysfunction was only evident in the animals that had hemorrhagic shock in addition to intramedullary nailing. Ertel and associates studied the level of proinflammatory cytokines in 213 patients who had sustained mechanical trauma.11 The levels of tumour necrosis factor-α and interleukin-1β were correlated with the severity of injury and patient survival. The presence of cofactors such as the release of mediators as well as the presence of other injuries and hypotension may be more important than the presence of pulmonary fat in the development of ARDS.
Diagnosis
The diagnosis of fat embolism is difficult since the disorder may coexist with other entities such as the ARDS, pulmonary contusion, aspiration pneumonitis, head injury and drug or alcohol toxicity. Fat embolism syndrome is most frequently seen in association with a high energy long bone fracture. Symptoms may be immediate or delayed up to 3 days after trauma, although 85% of clinically apparent cases occur within 48 hours.1 Fat embolism syndrome manifests itself by a combination of pulmonary, cerebral and cutaneous symptoms. Dyspnea, tachypnea and tachycardia and pyrexia are commonly noted. Neurologic changes are also observed, including restlessness, disorientation, confusion, stupor or coma. Petechiae may be noted across the chest, in the axilla and at the root of the neck. Conjunctival petechiae can be observed by rolling back the eyelids. Fundoscopic examination will reveal retinal microinfarcts. The distribution of the petechiae is thought to be due to differential streaming of fat globules to the upper part of the aorta as an effect of gravity with the patient positioned either upright or semiupright. The rapid pulse and respiratory rates and the absence of localizing neurologic signs differentiate fat embolism syndrome from craniocerebral trauma.
Gurd and Wilson described major and minor signs to look for in diagnosing fat embolism syndrome.12 Major signs were respiratory insufficiency, cerebral involvement and petechial rash. Minor signs were fever, tachycardia, retinal and renal changes, and jaundice. They accepted the diagnosis when 1 major and 4 minor signs were present, together with macroglobulinemia. According to Lindeque and associates13 the criteria of Gurd and Wilson were too restrictive. Lindeque’s group believed that the diagnosis should be based on a Pao2 of less than 60 mm Hg, a Paco2 of more than 55 mm Hg, a respiratory rate greater than 35 respirations per minute or clinical evidence of respiratory distress. It is difficult to know how these criteria would differentiate patients with fat embolism syndrome from patients with other types of pulmonary disorders.
No pathognomonic laboratory test exists for fat embolism syndrome. Arterial hypoxemia is common, and blood gas levels should be measured in all patients suspected of having fat embolism syndrome. Thrombocytopenia may be present along with diffuse pulmonary infiltrates. Serial chest x-ray films should be obtained, although the infiltrates seen in association with fat embolism syndrome are neither specific for the disorder nor always present when the syndrome first manifests itself. Nonspecific T-wave changes and right bundle-branch block may be seen on the cardiogram. Fat globules can be found in the blood, sputum, urine and cerebrospinal fluid. Since the presence of fat in the blood may be transient and since therapy is not directed at dissolution of the fat, laboratory demonstration of the fat is not required to make the diagnosis. Biopsy of petechial cutaneous lesions or the pulmonary or renal parenchyma may reveal the presence of intravascular fat, although biopsy is neither recommended nor required for the diagnosis. Fat embolization can coexist with other pulmonary and cerebral disorders.
The diagnosis of fat embolism syndrome is based on the patient’s history, supported by clinical signs of pulmonary, cerebral and cutaneous dysfunction, confirmed by the demonstration of arterial hypoxemia in the absence of other disorders.
Treatment
The treatment of fat embolism syndrome can be divided into general supportive measures and specific measures. Above all, treatment is directed toward maintaining adequate tissue oxygenation. General supportive measures include splinting fractured extremities, maintaining fluid and electrolyte balance, minimizing unnecessary transportation and administering oxygen. Patients should receive oxygen in sufficient quantities to maintain the Pao2 at 90 mm Hg or higher. If hypoxemia is severe, endotracheal intubation and mechanical ventilatory assistance are indicated.
A variety of agents, including ethanol, heparin, hypertonic glucose and corticosteroids have been recommended to treat fat embolism syndrome. There is a paucity of controlled clinical trials, and anecdotal information abounds in the literature regarding the use of these agents. The administration of corticosteroid deserves close scrutiny and has received the most attention in the literature. Byrick and colleagues found that methylprednisolone did not alter the acute hemodynamic effect of fat embolism in a canine model.14 Similarly, the levels of inflammatory mediators were not increased, although the delayed fall in oxygen saturation the investigators usually observed after fat embolism in their model was blunted by methylprednisolone.
Rokkanen and colleagues noted a reduction in the incidence of fat embolism syndrome from 6 of 15 patients in a control group to 1 of 14 patients in a group of patients receiving methyl-prednisolone 10 mg/kg body weight every 8 hours, starting in the emergency room.15 Schonfeld and colleagues did not diagnose fat embolism syndrome in any of 21 patients receiving methylprednisolone but made the diagnosis in 9 of 41 patients receiving a placebo.16 Lindeque and associates demonstrated improved arterial oxygenation in a group of patients receiving methylprednisolone.13 Corticosteroids are thought to exert their action by protecting the capillary endothelium, stabilizing granulocyte membranes, reducing complement system activation and retarding both platelet aggregation and the release of serotonin. The safety and relative efficacy of methylprednisolone given prophylactically in specific subsets of patients remains an area of active investigation. At present, the mainstay of treatment for fat embolism syndrome is the adminstration of oxygen and mechanical ventilation to maintain tissue oxygenation. Some clinicians recommend the administration of corticosteroids for their general anti-inflammatory action, accepting the complications that can occur from steroid use. I do not routinely use corticosteroids in the treatment of patients with multiple long bone injuries who are at risk for the development of fat embolism syndrome.
Early fracture fixation has been found to decrease the incidence of pulmonary complications following long bone trauma.17 A number of studies have identified a lower incidence of fat embolism syndrome and other pulmonary disorders in patients whose long bone fractures were fixed early.18 In the 1985 study of Johnson, Cadambi and Seibert,17 the incidence of ARDS was increased more than five-fold in patients whose pelvic and major long bone fracture stabilization was delayed more than 24 hours after injury. Bone and associates compared early (less than 24 hours) and late (more than 48 hours) stabilization of femoral fractures in 178 patients.18 When stabilization of the fracture was delayed in the patients who had multiple injuries, the incidence of ARDS, fat embolism and pneumonia increased.
Although there is general agreement that early fixation of long bone fractures is beneficial in most patients, some investigators have reported a deleterious effect arising from the insertion of reamed intramedullary nails in patients with pulmonary compromise. 19 Intramedullary nailing causes an increase in intramedullary pressure and intravasation of fat emboli from the bone marrow. The benefit from early fixation of femoral fractures by intramedullary nailing in patients with pulmonary compromise is currently receiving close scrutiny. Pape and colleagues retrospectively studied patients who had an Injury Severity Score of more than 18 points, a thoracic injury and treatment of a femoral fracture with reamed nailing.19 Early fixation was associated with a higher incidence of ARDS and a higher death rate than delayed fixation (8 of 24 compared with 2 of 25, and 5 of 25 compared with 1 of 25 respectively). They concluded that in the presence of a thoracic injury primary intramedullary nailing with reaming of the femur causes additional pulmonary damage and may trigger the development of the ARDS.
On the other hand, Bosse and associates studied patients with and without a thoracic injury who were admitted to 2 trauma centres.20 At 1 centre 217 of 229 patients had their femoral fracture fixed with intramedullary nailing and at the other centre 206 of 224 patients received plate fixation. The overall incidence of ARDS was 10 of 453 (2%), and the rate did not differ between the 2 centres. Similarly, the frequencies of pneumonia, pulmonary embolism, multiple organ failure and death were similar whether nailing with reaming or plate fixation was used.
Schemitsch and colleagues examined the pulmonary effects of the timing and method of fracture fixation in a canine model of fat embolism.21 Fat embolism was induced by reaming of the ipsilateral femur and tibia followed by pressurization of the intramedullary canal. The experimental model produced pulmonary fat embolism, pulmonary hypertension, hypoxemia and impaired pulmonary gas exchange. A femoral fracture was created and fixation performed with a plate, an unreamed intramedullary nail or a reamed intramedullary nail, 4 hours after embolic showering in 15 animals and 24 hours after embolic showering in 18 animals. Fracture fixation 4 hours after embolization by intramedullary nailing with and without reaming produced alveolar–arterial gradients that were higher than baseline values, whereas fixation with a plate did not change the alveolar–arterial gradient. The gradients were 4.0 and 3.5 times higher with reamed and with unreamed nails, respectively, than with plate fixation. The other parameters of pulmonary function that were measured did not change. Twenty-four hours after embolization, pulmonary arterial pressure, which had been elevated at 4 hours, had returned to normal. In the group of animals that had fixation 24 hours after embolization none of the methods for fixation affected the alveolar–arterial gradient. The amount of embolic intravascular fat persisting in the lungs, kidneys and brain was not affected by the method of fixation of the fracture. Schemitsch’s study suggested that the exact method used for fixation of a fracture plays a minor role in the development of pulmonary dysfunction and cannot be related to the development of ARDS.
Fat embolism syndrome continues to be an important disorder because of its significant morbidity and potential mortality. An understanding of the syndrome has become even more important with the recognition that fat embolism syndrome is associated with the implantation of a variety of cemented intramedullary prostheses. The pathogenesis of the syndrome is becoming better understood and it is hoped that, with time, a better understanding of its pathophysiology and its relation to ARDS will be gained. As more is learned about the various triggers for the development of fat embolism syndrome the prospect of more specific therapy for the prevention and treatment of the disorder will become a reality.