Abstract
Objective: To assess the influence of osteochondral graft preservation techniques on post-transplant biomechanics of graft and host subchondral bone in the knee joint.
Design: An experimental animal model (sheep), specifically the weight-bearing articular surface of the medial femoral condyle of the knee joints.
Intervention: Each sheep received, in the ipsilateral knee, an allograft that was (a) frozen without dimethyl sulfoxide (DMSO), (b) snap-frozen in liquid nitrogen or (c) frozen with DMSO. The contralateral knee received an autograft that was (a) snap-frozen, (b) treated with DMSO or (c) left untreated (fresh).
Main outcome measures: Mechanical and material properties of bone, including maximal compression stress, modulus of elasticity and bone mineral ash content of subchondral bone cores (from the graft centre and surrounding host bone).
Results: No significant differences were found in the mechanical properties of the subchondral bone under the graft, but there were significant changes in surrounding bone. Bone surrounding the grafts that were snap-frozen or frozen without DMSO was significantly stronger than the normal control bone. However, bone surrounding fresh autografts and cryoprotected allografts was not significantly different from normal control bone.
Conclusions: The changes in the mechanical behaviour of the host bone may be associated with graft cell viability. The greater stiffness of the subchondral host bone may have consequences for long-term graft integrity and for the development of degenerative osteoarthritis.
Surgical intervention is increasingly common for the treatment of articular joint injuries and diseases such as metaphyseal bone tumours, osteochondritis dissecans, osteonecrosis and osteoarthritis. In the United States, approximately 500 000 arthroplasties are performed annually at a cost of over US$300 million.1 Mechanical failure and loosening associated with artificial joint replacements lead to a significant number of revision procedures. 2 emphasizing the need for biologic, joint reconstruction, especially in the joints of young, active people.
For biologic reconstruction to be practical, however, osteochondral tissue must be preserved and stored for long periods. Some freezing protocols can damage cells and degrade graft tissues after transplantation. Also, the transplanted osteochrondral grafts can produce changes in the surrounding host tissues of the joint, exacerbating joint degeneration. The ability of osteochondral grafts to remain viable and mechanically functional depends partly on the integrity of both the articular cartilage and its subjacent bone. Our group has focused on the mechanical behaviour3–5 and cellular viability6– 8 of the cartilage component of osteochondral autografts and allografts, but little information is available about the role of the osseous component of the osteochondral graft.
Radin and colleagues9–11 have suggested that altered subchondral bone stiffness may be an early precursor of joint degeneration. Thus, if the subchondral bone properties are substantially altered after osteochondral graft transplantation, then graft viability and joint function could be compromised.
Our purpose was to investigate the interactive effects of osteochondral transplantation and cryopreservation methods on graft and host subchondral bone. Our results suggest that cryo preservation alters not only the mechanical properties of graft subchondral bone but also the mechanical properties of the surrounding host subchondral bone. To better elucidate the mechanisms involved in graft incorporation, future studies should assess bone remodelling activity and mechanical properties at multiple time intervals after transplantation. Longterm joint integrity may be affected not only by graft success but also by graft–host interactions and subsequent changes in the properties of host subchondral bone. These data are important to consider in the resurfacing of damaged joint surfaces by techniques such as small shell, partial joint or osteochondral transplantation.
Method
Twenty-four, adult, female Suffolk/ Romanoff crossbred sheep were randomly assigned to a combination of 2 grafts (1 graft per knee): 1 of 3 autograft groups and 1 of 3 allograft groups (8 knees/group). The osteochondral autograft groups (Table I) consisted of fresh autografts (fr-auto), autografts exposed to dimethyl sulfoxide (DMSO-auto) and snap-frozen autografts (sn-auto). The allograft groups consisted of noncryoprotected frozen allografts (frz-allo), cryoprotected frozen allografts (cr-allo) and snap-frozen allografts (sn-allo).
Osteochondral Autograft and Allograft Treatment Groups
Fr-autos were transferred fresh from the ipsilateral to the contralateral condyle as surgical controls. A control for cryoprotectant toxicity consisted of DMSO-autos immersed in 10% DMSO (a cryoprotectant) solution at 22 °C for 30 minutes but not frozen. Negative autograft controls consisted of sn-auto samples plunged in liquid nitrogen without cryoprotection and rewarmed in a water bath at 37 °C 4 times to cause extensive freeze–thaw damage to resident cells.
Frz-allos were frozen at 1 °C/min to −70 °C without prior treatment with a cryoprotective compound. Cr-allos were placed in a 10% (w/w) DMSO solution in balanced salts at 22 °C for 30 minutes before controlled cooling at 1 °C/min to −70 °C. Sn-allos were put through the same protocol as sn-autos to function as a negative allograft control.
After we had received the approval of the University Animal Care Committee, the sheep were anesthetized and their hind limbs were secured in flexion to a customized peg board positioning plate.12 A medial stifle (knee joint) arthrotomy and resection of the intra-articular fat pad exposed the medial femoral condylar site of the osteochondral graft transplant. A gimballed aiming jig (Concept Corp., Largo, Fla.) was attached to the positioning plate to align a coring bit (11.5 mm outside diameter [od], 10.0 mm inside diameter [id]) perpendicular to the weight-bearing surface of the medial femoral condyle. The resulting (10.0 mm) core was removed with the use of a custom extraction scoop that broke the graft off flat at its base. The depth of the graft site was measured and a previously treated (Table I) 11.5-mm allograft from a donor ovine stifle joint (Canada West Foods, Innisfail, Alta.) was cut to the required length and press-fit in to the defect with firm (thumb) pressure. The 10.0-mm diameter autograft was then treated by 1 of 3 protocols (Table I) and inserted into the contralateral medial femoral condyle. A smaller coring bit (10.0 mm od and 8.5 mm id) was used to prepare the recipient site in the contralateral condyle. The 8.5-mm core, removed from the contralateral condyle, was kept as an untreated in vitro baseline control.
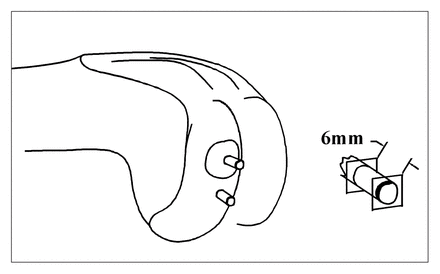
After the procedure, all 24 sheep were allowed to ambulate freely in large pens at the university farm. Twelve months later the animals were euthanized with intravenous injection of pentobarbital, and the femoral condyles were harvested. A stainless-steel coring drill bit was used to extract a cylindrical dowel of subchondral bone (3 mm diameter) from the graft centre and from the surrounding host bone (1 cm posterior to the graft; Fig. 1). Control bone samples were cored from the 8.5-mm dowels that were removed from the contralateral femoral condyles at surgery. Physiological buffer solution (pH 7.4, 21 °C) was forced through the core of the drill bit to moisten and cool the specimen during drilling. The uneven ends of the core were cut to produce a 6-mm long dowel of subchondral bone. The distal end of the dowel was then fixed to a stainless steel plate with a thin layer of polymethylmethacrylate to reduce the edge effect due to unconstrained trabeculae. 13,14 The proximal end was constrained by the remaining subchondral plate. While immersed in physiological buffer solution, each dowel of subchondral bone was compressed (Instron 1122; Instron, Canton, Mass.) to 50% of its original height at a rate of 50% strain/s.15,16 The load-time curve was digitized (IBM PC/AT; RC Computerscope ISC–16; RC Electronics, Santa Barbara, Calif.) at a sample rate of 2 kHz. A custom computer program was used to analyse the load-deformation curves. Among the variables determined for each osteochondral dowel were maximal stress (σm [strength]), modulus of elasticity (E, [stiffness]) and maximal strain energy density (SED).
Diagram showing sheep medial femoral condyle with subchondral bone cores (3-mm diameter) from graft centre and surrounding host bone (1 cm posterior to the graft). The surface and deep ends were cut to create flat ends perpendicular to the long axis of the core. The result was a 6-mm long sample of the subchondral and secondary-spongiosa bone complex.
The crushed subchondral dowels were separated lengthwise with a scalpel into 2 approximately equal parts. One part was ash dried for 24 hours at 100 °C, weighed, ashed for 24 h at 600 °C, and reweighed.17 The second part was analysed for hydroxy proline content as described by Blumenkrantz and Asboe-Hansen.18
Multivariate analysis of variance procedures (SPSS, University of Chicago, Ill.) were used to detect significant effects among the treatment groups. The dependent variables were bone ash content, hydroxyproline content, and bone material properties (σm, E and SED). When the main effects were significant, post hoc comparisons were made with Tukey multiple range tests (SPSS). Data presented are the means (and standard deviation); p < 0.05 was used for all comparisons.
Results
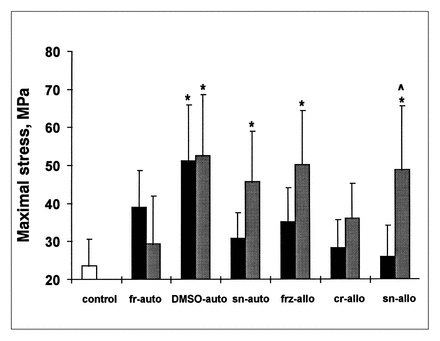
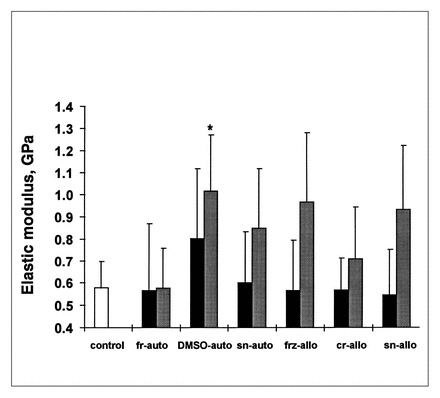
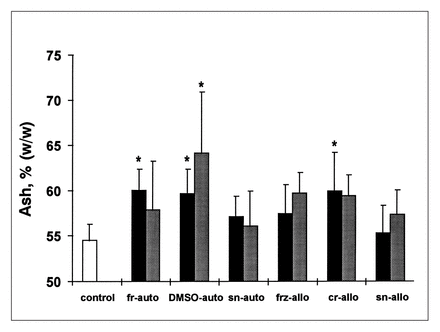
For most treatment groups, the host bone surrounding the graft had greater strength (Fig. 2) and stiffness (Fig. 3) than the bone in the graft centre. The single exception was the frauto group, in which the graft was stronger than the surrounding bone. Compared with all other treatment groups, strength and stiffness were closest to the normal control values for the subchondral bone surrounding the fr-autograft. In contrast, the DMSO-autograft and surrounding bone were the strongest and stiffest of all groups. Ash content (Fig. 4) of bone surrounding the DMSO-autografts was also significantly greater than the control and other autograft groups.
Maximal stress for subchondral bone cores from the graft centre (black bars) and surrounding host bone (shaded bars) for the 7 groups (see Table I for description of the groups). Asterisks indicate groups that were significantly stronger (p < 0.05) than the baseline control bone group (white bar); the carat indicates that the surrounding bone was significantly stronger than the graft.
Modulus of elasticity (stiffness [E]) of subchondral bone cores from graft centre (black bars) and surrounding host bone (shaded bars). The asterisk indicates a significant difference (p < 0.05) from baseline control group (white bar). Most graft E values were similar to the control, as was that of fr-auto surrounding bone.
Percent ash content of subchondral bone cores from graft centre (black bars) and surrounding host bone (shaded bars). Asterisks indicate groups that differed significantly (p < 0.05) from the baseline control group (white bar).
The nonprotected frozen groups (frz-allo, sn-auto and sn-allo) had moderately strong grafts but significantly stronger surrounding bone than the control group. The stiffness of these grafts was also similar to the control group; however, the surrounding bone showed greater stiffness than the control. Ash content did not correlate significantly with the material properties of either frz-allo or snap-frozen groups.
Of the allografts, the cr-allo material properties were most similar to the control and fr-auto. The only significant difference between the cr-allo group and the control was in subchondral bone ash content at the graft site. With the exception of the DMSO-auto group, ash content had only a low correlation with mechanical data. SED and hydroxyproline values were variable and did not correlate significantly with either σm or E.
Discussion
To study the interactive effects of osteochondral transplantation and method of cryopreservation on subchondral bone, we measured the mechanical behaviour of bone, its mineral composition and hydroxyproline content of osteochondral allografts and autografts 12 months after transplantation. This time was chosen because we were interested in the longer-term effects of cryopreservation and transplantation.
Although significant differences were found in our data, caveats and limitations should be emphasized. Researchers have noted heterogeneity of mechanical properties of cancellous bone throughout the joint.19,20 Our basal control bone originated from the anterior medial condyle removed at surgery to make room for the autograft. Because of their location, the basal control samples were suitable comparisons for the mechanical properties of the grafts because all of our graft material originated from the anterior medial condyle. Cores of surrounding bone, however, were taken 1 cm posterior to the graft, approximately midway along the medial condyle. Recent data from our group suggest that ovine subchondral bone strength in the femoral condyles increases from anterior to posterior:21 the midmedial condyle has a 25% greater σm than the anterior medial condyle. Hence, location-to-location heterogeneity would be a potential explanation for the differences in graft and surrounding bone in several of our groups (sn-auto and all allograft groups), but it is not consistent for all the groups. Indeed, the fr-auto (surgical control) and the DMSO-auto groups did not display heterogeneity at these adjacent sample sites. Furthermore, the differences among the groups (both for graft and surrounding locations) suggest that mechanisms other than the heterogeneity of subchondral bone across the joint are implicated.
A further limitation is that the core size our subchondral bone for mechanical testing was only 3 mm in diameter. Because of the size and shape of the sheep femoral condyle and because the core sites converged deeper within the condyle, larger diameter cores would have resulted in crosssample interference. Trabecular bone is not a continuous material, and our cores may not have included a sufficient number of intertrabecular lengths for continuum assumptions as suggested by Harrigan and associates. 22 Also, before the destructive compression test we did not defat the bone cores or determine apparent density. Thus, the marrow tissue in the bone cores may have contributed to the variability of the ash content data, and the apparent density may have helped to explain differences between the mechanical properties of the groups. Our interest, however, was in the comparison of mechanical behaviour between treatment groups rather than an absolute measure of the mechanical properties of trabecular bone. These limitations notwithstanding, we detected significant graft versus surrounding tissue and intergroup differences.
Our highest values of σm of subchondral bone ranged from 30 to 80 MPa for the bone surrounding the DMSO-auto group. These values were relatively high when compared with reported mechanical properties of cancellous bone, but the E values were well within the upper range of those reported by others (Table II).13,19,23 The differences in the mechanical properties may have been due to the apparent high density of subchondral bone as a result of the osteochondral transplantation. Surgical removal of the bone core at the time of transplantation was the initial insult to the subchondral bone, to which the remaining host tissue likely responded in a manner similar to that of fracture repair. Trabecular microfracture repair, like cortical repair, produces a callus of bone around the fracture zone.9,24 However, subchondral bone injury caused by the coring and graft transplantation is substantially greater than that of an individual trabecular microfracture and may have induced a much greater fracture response. Thus, bone surrounding the graft zone likely developed an extensive fracture callus.
Ranges of Maximal Stress and Elastic Modulus Values
Enneking and Mindell25 recovered massive human allografts, of which half were osteochondral grafts, and most were cryopreserved using glycerol. They reported that host osteoblasts formed a seam of “reparative” bone around graft trabeculae up to a few millimetres into the graft, and that trabeculae deeper into the graft than this remained necrotic but intact for up to several years. If the processes of incorporation were similar in our 10- to 11.5-mm diameter ovine grafts, there may have been full penetration of host bone into the graft trabecular interstices, with a layer of host bone enveloping the original graft trabeculae. The combination of host and graft trabecular bone would increase the apparent density of the graft region and would, therefore, increase graft mechanical strength and stiffness. The DMSO- and fr-auto groups were consistent with this suggestion, but the graft σm for the other groups was similar to the control values.
Another mechanism may also have influenced the mechanical properties of the subchondral bone. Data that our group reported elsewhere26 showed that the greatest cellular damage occurred in grafts that were unprotected during freezing (sn-auto, sn-allo, and frz-allo). The current data show that the greatest differences in material properties between graft and surrounding bone also occurred in these 3 unprotected groups. In these 3 groups, the surrounding bone was stronger and stiffer than either control bone or their respective grafts. The fr-auto group was the only one with greater graft strength than surrounding-bone strength, with similar elastic moduli in both regions. The intermediate strength and stiffness of the cr-allo group (between fr-auto and the 3 unprotected freezing groups) suggest a link between graft-tissue viability and the graft–surrounding bone interaction.
Stevenson, Li and Martin27 found that freezing and graft antigenicity modulated the process of graft incorporation in massive osteochondral grafts of the canine radius. The greatest amount of host bone-forming surface found on their graft trabeculae was in frozen antigen-matched allografts. However, the amount of new bone formation did not correlate with the amount of trabecular remodelling in the grafts. Freezing reduced the amount of bone remodelling in both of their antigen-matched and mismatched grafts. Thus, graft freezing can play a role in the incorporation of the graft tissue and the remodelling process, whereas cryoprotection may moderate the effects of freezing on graft incorporation.
Although antigenicity affected graft incorporation in the model of Stevenson, Li and Martin,27 in our model (at 12 months after transplantation) any effects of antigenicity were overshadowed by the effects of freezing. There were no significant differences in the mechanical properties or the ash content between the 2 snap-frozen treatment groups (auto versus allo). Also, potential differences in antigenicity between the autograft and allograft groups did not translate into differences in mechanical properties at 12 months. Graft antigenicity may not play as significant a role as freezing damage (or protection from freezing damage) in the long-term outcome of our graft model, but short-term responses will need to be explored.
Although other researchers have examined the changes that occur in graft bone after transplantation, none have examined the surrounding host tissue. The increased strength and stiffness that we found in the subchondral bone surrounding an osteochondral transplant dowel may have consequences for long-term graft viability and the likelihood that osteo arthritis will develop. The aim of surgical treatment of joint injuries and diseases is to improve and maintain joint integrity for normal daily activities. The failure of joint replacements, both artificial and biologic, and subsequent surgical revision add significant costs to the health care system. The success of allograft replacements and a reduction in surgical revisions may be improved by understanding the mechanisms of osteochondral graft incorporation. More studies will be needed to characterize the remodelling activity that occurs earlier during graft incorporation. This preliminary investigation of graft versus surrounding autograft and allograft subchondral bone highlights the need to examine bone-cell viability and bone remodelling in both the graft and surrounding regions.
Acknowledgments
This research was supported, in part, by a grant from the Medical Research Council of Canada and the Alberta Heritage Foundation for Medical Research.
Footnotes
Presented in part at the annual meeting of the Orthopaedic Research Society, New Orleans, Feb. 21 to 24,1994, and at the annual meeting of the Canadian Orthopaedic Research Society, Winnipeg, Man., June 12 to 15, 1994
- Accepted September 22, 1997.