Abstract
Introduction: The benefit of administering β-adrenergic blocking agents perioperatively to surgical patients at high risk for myocardial ischemia has been demonstrated in several well-designed randomized controlled trials. These benefits have included a reduction in the incidence of myocardial complications and an improvement in overall survival for patients with evidence of or at risk for coronary artery disease (CAD). We designed a retrospective study at the Ottawa Civic Hospital to investigate the use of β-blockers in the perioperative period for high-risk general surgery patients who underwent laparotomy and to explore the reasons for failure to prescribe or administer β-blockers when indicated.
Methods: All 236 general surgery patients over the age of 50 years who underwent laparotomy for major gastrointestinal surgery between Jan. 1, 2001, and Dec. 31, 2001, were assigned a cardiac risk classification using the risk stratification described by Mangano and colleagues. The perioperative prescription and administration of β-blockers were noted as were the patient’s heart rate and blood pressure parameters for the first postoperative week, in-hospital adverse cardiac events and death.
Results: Of the 143 patients classified as being at risk for CAD or having definite evidence of CAD, 87 (60.8%) did not receive β-blockers perioperatively. Of those who did, 43 were previously on β-blockers and 13 had them ordered preoperatively. Patients with definite CAD were significantly more likely than others to receive β-blockers perioperatively (p < 0.001), as were patients seen by an anesthesiologist or an internist preoperatively (p < 0.001). Twenty (33%) of the 61 patients who were already taking β-blockers preoperatively had them inappropriately discontinued postoperatively. Once prescribed by the physician, β-blockers were administered by the nurses irrespective of nil par os status. The mean heart rate and blood pressure parameters for patients receiving β-blockers postoperatively was 82 beats/min and 110 mm Hg, respectively, and these values were not significantly different from the mean heart rate of patients not receiving β-blockers. The number of postoperative cardiac events was significantly higher in patients with definite evidence of CAD, and among this group, the use of β-blockers was associated with a significant reduction in postoperative cardiac events. This was not true for patients at risk for CAD or patients with no risk of CAD.
Conclusions: A significant proportion (> 60%) of general surgery patients who were identified as having definite evidence of, or being at risk for, CAD were not prescribed β-blockers preoperatively. More than 30% of patients who were on β-blockers preoperatively did not have them reordered postoperatively. These results may reflect controversy surrounding the recommendations, miscommunication between surgeons and anesthesiologists and errors in postoperative ordering.
Cardiac events complicate noncardiac surgery in up to 18% of patients with a known history of coronary artery disease (CAD) or at risk for CAD.1 The most significant risk factor for death and cardiovascular morbidity is postoperative myocardial ischemia, which is related to a persistently elevated heart rate in the first postoperative week.2 Results from several well-designed randomized controlled trials1,3– 6 suggest that appropriately administered β-adrenergic blocking agents reduce perioperative ischemia and may reduce the risk of myocardial infarction and death in high-risk patients. The calculated “numbers needed to treat” in these studies ranged from 2.5 to 8.3, indicating a clinically and statistically significant improvement in adverse cardiac outcomes.
The study of Mangano and colleagues1 is the largest and most frequently cited of the randomized control trials. It involved 200 high-risk patients who underwent noncardiac surgery and were randomized to receive either atenolol or placebo in the perioperative period. A significant reduction in perioperative myocardial ischemia3 and cardiac death1 was demonstrated, and these results have formed the basis for the class IIa recommendation from the American College of Cardiology and the American Heart Association that β-blockers be administered perioperatively to patients with known CAD or with major risk factors for CAD7 and for the American College of Physicians Clinical Guidelines for the management of perioperative risk from CAD associated with noncardiac surgery.8
This study was designed to determine if patients with a history of CAD or at risk for CAD were being appropriately identified and treated perioperatively with β-blockers among a group of general surgery patients who underwent laparotomy at a university-affiliated tertiary care hospital.
Patients and methods
The medical records computer database was used to identify all patients over the age of 50 years who underwent a laparotomy for major gastrointestinal surgery and were discharged from our institution between Jan. 1, 2001, and Dec. 31, 2001. This search yielded 261 patient charts; 9 patient charts were excluded because of miscoding, and 16 patient charts could not be located, leaving 236 patient charts for evaluation.
We searched the patient charts for information regarding patient and surgery demographics such as age, sex, diagnosis, type of anesthesia used, type of procedure and length of hospital stay. In addition, the medical history was used to determine if patients had definite evidence of CAD or were at risk for CAD. This definition was based on the criteria presented by Mangano and colleagues.1 Definite CAD was defined as a previous myocardial infarction, coronary artery bypass grafting (CABG), percutaneous transluminal angioplasty (PTA), a stress test that revealed abnormalities or documented typical angina. Patients at risk for CAD were defined as those having 2 or more of the following risk factors: age 65 years or older, current smoking, hypertension, hypercholesterolemia or diabetes mellitus. Patients were then stratified with respect to risk such that patients with no risk of CAD were labelled Mangano class I, those at risk for CAD were labelled Mangano class II and those with definite evidence of CAD were labelled Mangano class III.
Information was obtained on the type of preoperative consultation (i.e., preadmission unit, anesthesia or internal medicine/cardiology) and on the preoperative prescription of β-blockers. Data were also collected on postoperative use of β-blockers, including the reason for prescription (a reorder, tachycardia, or the treatment of an adverse cardiac event), the route ordered and the reliability of administration by the nursing staff. Patients were then subdivided into those who received a β-blocker both pre- and postoperatively, those who did not have their β-blocker reordered postoperatively and those who did not receive β-blockers prophylactically at any time in the perioperative period.
The outcome measures included hemodynamic parameters and adverse cardiac events. Data were collected on the mean systolic, diastolic and mean blood pressure and maximum, minimum and mean heart rate for each day of the first postoperative week. Cardiac events included myocardial infarction (defined as any 2 of the following: electrocardiographic [ECG] evidence, a positive troponin test [TnT] or unrelenting chest pain), a TnT elevation or ECG change not meeting the criteria for myocardial infarction, a new dysrhythmia, decompensated congestive heart failure or cardiac death.
All data were collated using Microsoft Excel 2000 into spreadsheet format. The data were analyzed using Systat 7.0 for Windows 1997. Categorical variables were compared with the χ2 test, and continuous variables were compared using the t-test.
Results
Patient and surgery demographics for the 236 study patients are presented in Table 1, and the distribution of patients within each risk stratification along with the inclusion criteria in Table 2.2 Ninety-three patients (39.4%) had no risk or evidence of CAD (class I), 81 (34.3%) patients were at risk for CAD (class II) and 62 (26.3%) patients had definite evidence of CAD (class III). Of the 143 patients who were defined as having evidence of or being at increased risk for CAD, 43 (30.1%) were previously on β-blockers and 13 (9.1%) were taking β-blockers prescribed for prophylaxis in the preadmission unit or during a preoperative internal medicine or cardiology consultation. The remaining 87 (60.8%) patients received no β-blockers preoperatively.
Demographic Features of the 236 Study Patients
Risk Stratification for Coronary Artery Disease (CAD) of Study Patients According to Mangano and Colleagues2
When subdivided by Mangano class and by the type of preoperative consultation, patients in class III were significantly more likely to receive β-blockers preoperatively than patients in classes I and II (p < 0.001). Preoperative consultation either in the preadmission unit or by an internist or cardiologist was significantly associated with the preoperative use of β-blockers prophylactically (p < 0.001) (Table 3).
Preoperative β-Blockade as a Function of Cardiac Risk and Preoperative Consultation
A total of 61 patients in all 3 classes received β-blockers preoperatively; 47 patients had been taking β-blockers previously and 14 patients had β-blockers prescribed perioperatively. Among the 47 patients who were previously on β-blockers, 34 had their β-blockers reordered within the first 2 postoperative days by intensive care unit (ICU) or general surgery staff, 6 patients had them reordered after postoperative day 2 once reminded by the pharmacist or nursing staff to do so; 7 patients did not have their β-blockers reordered or had them reordered only after sustaining an adverse cardiac event. Of the 14 patients who were prescribed β-blockers perioperatively, 7 had their β-blockers reordered within the first 2 postoperative days by ICU or general surgery staff and 7 did not or had them reordered only after sustaining an adverse cardiac event. Among the 175 patients who were not receiving β-blockers preoperatively, 25 patients received β-blockers postoperatively; 18 patients for treatment of postoperative tachycardia and 7 patients for the treatment of a postoperative adverse cardiac event. The use of β-blockers for the treatment of postoperative tachycardia was initiated by the Post Anesthetic Care Unit (PACU) or the ICU in 14 patients and by the general surgery team in 4 patients. Patients who had β-blockers ordered preoperatively were significantly more likely to receive them postoperatively (p < 0.001) (Table 4).
Postoperative β-Blockade as a Function of Preoperative β-Blockade
The β-blockers were ordered to be administered by the enteral route on the ward and were given by the nurses irrespective of nil par os status. Nine doses of β-blockers were missed out of a total of more than 800 doses. β-blockers were ordered primarily by the intravenous route in the PACU or the ICU and were frequently titrated to a heart rate of less than 90 beats/min. Nine of the 14 patients who received β-blockers intravenously in the monitored setting were not ordered oral β-blockers on transfer to the ward despite documented tachycardia.
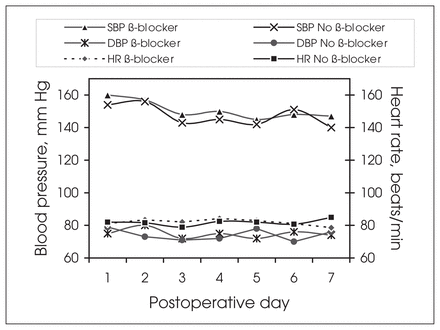
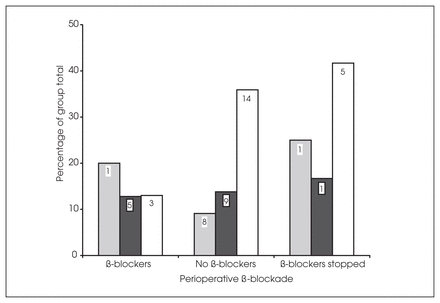
The average of the mean heart rate, systolic and diastolic blood pressures on the first 7 postoperative days for patients receiving β-blockers perioperatively was not significantly different from patients not receiving perioperative β-blockers (p = 0.6) (Fig. 1). Cardiac events were documented in 10 patients with no CAD (11%), 15 patients in class II at risk for CAD (18%) and 22 (36%) patients in class III having definite CAD (Table 52). Patients in class III were significantly more likely to experience a postoperative cardiac event (p < 0.001) when compared with patients in classes II and I. Within class III, β-blockers were associated with a significant decrease in the number of documented cardiac events from 49% to 13% (p < 0.005) (Fig. 2).
Comparison of mean heart rate (HR), mean systolic blood pressure (SBP) and mean diastolic blood pressure (DBP) for patients receiving and not receiving β-blocking agents perioperatively.
Postoperative adverse cardiac events as a function of risk stratification for coronary artery disease (CAD) and perioperative β-blockade. Shaded columns = no risk of CAD (class I), black columns = at risk for CAD (class II), white columns = definite CAD. Numbers in columns represent numbers of patients in each subgroup.
Comparison of Postoperative Adverse Cardiac Events by Risk Stratification and Perioperative β-Blockade
Discussion
Despite clear recommendations in the literature, more than 60% of patients with evidence of CAD or at high risk for CAD did not receive β-blockers perioperatively at our institution. Patients with definite evidence of CAD were significantly more likely to receive β-blockers preoperatively than patients with risk factors for CAD. This may represent controversy among anesthesiologists and internists surrounding the evidence supporting these recommendations and their ability to be generalized. Those skeptical of the recommendations argue that the study of Mangano and colleagues2 is too small and has too many limitations to warrant the proposed widespread use of β-blockers.9,10 Specifically, Mangano and colleagues randomized patients to the placebo group who had previously been on long-term β-blockers, resulting in their inappropriate discontinuation postoperatively. Moreover, the study failed to demonstrate a reduction in cardiac morbidity or mortality in the first postoperative week, the period of atenolol therapy (4 deaths in the atenolol group and 2 deaths in the placebo group). A reduction in cardiac mortality was only observed at follow-up 6 months after discharge, but if the deaths that occurred in the first postoperative week are included in this analysis, the findings are no longer statistically significant (13% mortality in the atenolol group v. 23% mortality in the placebo group, p = 0.07). In contrast, the smaller study by Poldermans and colleagues,4 evaluating the efficacy of perioperative bisoprolol, demonstrated a striking 91% relative risk reduction for the combined outcomes of cardiac death and myocardial infarction. This study, however, included a highly selected group of vascular surgery patients with a positive dobutamine stress echocardiogram, making these results difficult to generalize. A meta-analysis of 10 randomized controlled trials1,3–6,11–15 evaluating the effects of β-blockers administered perioperatively in patients who underwent noncardiac surgery includes a total of 855 patients with only 15 cardiac deaths and 18 nonfatal myocardial infarctions. This overall analysis demonstrated only a marginally significant difference in cardiac mortality (3 cardiac deaths in the combined β-blocker arms and 12 in the placebo arms, p = 0.052) and none in nonfatal myocardial infarctions (2 in the β-blocker arm and 16 in the placebo arm, p = 0.43). Most importantly, there are still many unanswered questions with respect to who should receive β-blockers perioperatively, how much to give and for how long. These concerns have led to the design of the Perioperative Ischemic Evaluation Study, a large, Canadian, multicentre, randomized controlled trial of metoprolol versus placebo, with inclusion criteria based on the Revised Cardiac Risk Index described by Lee and associates.16 The results of this study should help to definitively establish or refute a statistically significant benefit for β-blockers given perioperatively and help to define the target population.
Despite this controversy, it appears from our results that surgeons, in the absence of a preoperative assessment, are not independently prescribing β-blockers prophylactically. An informal survey of surgeons and surgical residents at our institution suggests that this reflects a lack of familiarity with the recommendations and the indications for perioperative β-blockade as opposed to skepticism about the evidence behind these recommendations.
Of greater concern is the significant number of patients (20 of 61 [33%]) for whom long-term therapy with β-blockers was not reordered within the first 2 days postoperatively. Discontinuation of long-term β-blockers is associated with adverse outcomes and decreased patient survival. 17,18 The most likely explanation for the discontinuation is oversight as patients were often reordered their β-blocker after a reminder from the pharmacist or nursing staff or after an adverse cardiac event. Seven (15%) of the 47 patients who were previously on β-blockers had them inappropriately discontinued compared with 7 (50%) of the 14 patients who had β-blockers prescribed at the preoperative consultation. This difference can be explained by considering that when preoperative medications are reordered postoperatively, referral is made to the list of medications recorded by a nurse at the preoperative consultation. In order to be aware of the addition of a β-blocker to the preoperative medications, the surgeon or surgical resident writing the postoperative orders has to recall the preoperative consultation letter. This represents inadequate communication between the anesthesia and surgical teams. A second example of inadequate communication is demonstrated by the finding that 9 (64%) of the 14 patients who received β-blockers in either the PACU or the ICU did not have them continued enterally on the ward despite a documented need. In our opinion, this type of medical error and miscommunication is unacceptable and completely avoidable. Preprinted orders are simple and cost-effective and have been shown to increase safety and reduce medication error for cancer chemotherapy.19 Standard preprinted postoperative order sheets, where medications and recommendations for the individual patient can be added at the preoperative assessment, are currently being considered at our institution to address this problem.
Even among patients who did receive appropriate β-blockade perioperatively, the mean heart rate in the first postoperative week was between 75 and 85 beats/min and was not significantly different from patients who were not receiving a β-blockade. Although a specific target heart rate has not been demonstrated in the literature, there is reasonable evidence to suggest that postoperative tachycardia is the most consistent hemodynamic abnormality associated with myocardial ischemia and adverse cardiac outcome. 2,20 In a randomized controlled trial of β-blockers versus placebo, Raby and colleagues5 demonstrated that, although the β-blocker group experienced less postoperative ischemia, it was strict heart rate control that independently avoided ischemia. The mean heart rate recorded for the group receiving β-blockers in our study may reflect inadequate dosing and lack of titration to a target heart rate. With the exception of patients receiving β-blockers intravenously in a monitored setting, the dose of β-blocker ordered on the ward was not increased, even in the setting of persistent tachycardia. Inadequate dosing may be compounded by malabsorption of the medication in postoperative ileus and in the absence of oral intake. There are currently no pharmacokinetic studies investigating the absorption of β-blockers after gastrointestinal surgery or altered intestinal motility. This information would be valuable because many hospitals do not currently support the use of β-blockers given intravenously on the ward. If β-blockers taken orally are not well absorbed after major gastrointestinal surgery it would have important implications for the postoperative management of high-risk general surgery patients. The pharmacokinetics of a metoprolol suppository are currently under investigation at our institution and may provide a reasonable alternative for the postoperative administration of β-blockade for general surgery patients.
The significant reduction in postoperative adverse cardiac events among class III patients receiving β-blockers demonstrated in our retrospective study supports the results of previously published prospective randomized controlled trials.2–6,11–15 These study results combined with a review of the literature indicate that, although there is general agreement that there exists a subset of patients who will benefit from perioperative β-blockade for the prevention of postoperative myocardial ischemia and adverse cardiac events, there is still controversy. Specifically, questions remain regarding which patients will benefit, at what, if any, therapeutic target heart rate and regarding the ideal method of administration postoperatively, particularly after major gastrointestinal surgery. Several studies are currently underway at our institution that will likely help resolve some of these issues.
Footnotes
↵† Winner of the 2002 MacLean–Mueller Prize.
Competing interests: None declared.
- Accepted February 19, 2003.