Neuropathic shoulder secondary to syringomyelia
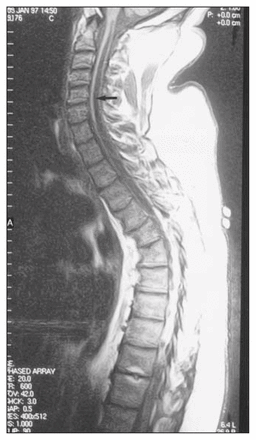
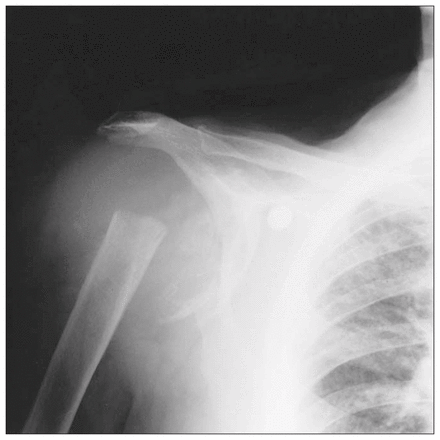
The radiographic findings in this case were suggestive of a destructive arthropathy, although a number of etiologies could account for these appearances. Pyogenic arthritis, crystal deposition, rheumatoid and seronegative arthritides could all give similar radiographic findings. However, the clinical findings and the absence of other joint involvement pointed to a neurologic cause. The location and history of previous cervical trauma, although minor, should prompt a search for a cervical syrinx. T1-weighted sagittal magnetic resonance images confirmed syringomyelia extending from C2 to the midthoracic spine (Fig. 2 [see page 300] and Fig. 3 [arrow]). It is important to remember that syringomyelia may involve more of the spinal cord than is clinically evident, and it is therefore important to image the entire cord to define the true extent of the condition.
Neuropathic (Charcot) joints represent the chronic form of a degenerative arthropathy that is associated with decreased sensory innervation of the involved joints. Decreased pain sensation leads to repetitive trauma, and sympathetic dysfunction results in local hyperemia and bone resorption, although up to one-third of patients have no demonstrable neurologic deficit.1 This characteristic destructive arthropathy is frequently associated with loss of proprioception and can be considered as an accelerated osteoarthritis that is precipitated by trauma to the joint not protected by its proprioceptive or nociceptive reflexes. This process continues until destruction of the joint occurs and is characterized by painless but unstable articulation.2
Causes of neuropathic arthropathy can be congenital or acquired (Table 1). Acquired causes include central neuropathies due to spinal cord injury or tumour and syringomyelia, in which the shoulder is usually affected3,4 as in this case. Acquired peripheral neuropathies such as diabetes or leprosy tend to affect the lower limb.1 Rarer causes include collagen vascular diseases, rheumatoid and seronegative arthritides, amyloidosis, tabes dorsalis and congenital indifference to pain.
Causes of Neuropathic Arthropathy
The etiology of this entity has varied throughout history as different diseases have become less or more prominent. It is now most frequently seen with syringomyelia and may be the presenting symptom, as in our case, or the symptom leading to the diagnosis. The diagnosis should be considered whenever a patient has what appears to be a severe osteoarthritis after minor trauma.1
Radiologically, Charcot joints are typically characterized by the 5 Ds: dense subchondral bone, destruction of articular cortex, deformity, debris and dislocation.3,5 A persistent joint effusion may also be present, especially in the early stages.1,2 Speckled calcification and shards of bone in the periarticular soft tissues will be seen with disease progression.1 When the process is severe, the radiographic changes are pathognomonic with joint disorganization more profound than any other arthropathy, although infection and deposition of calcium pyrophosphate dihydrate crystals may be difficult to exclude.1 An atrophic form with resorption of the proximal humerus has been described in syringomyelia, diabetes and syphilis1 (Fig. 4). The joint fluid may be inflammatory, and infection may be a complication.
Syringomyelia is defined as a longitudinally orientated cerebrospinal fluid-filled cavity with gliosis of the spinal cord frequently involving both the parenchyma and the central canal.3 The predominant location of syringomyelia is the lower end of the cervical cord.3 If the condition is acquired, it is usually post-traumatic or postinflammatory. The resulting loss of pain and temperature sensation typically leads to trophic changes and neuropathic (Charcot) joint changes.3 Muscle weakness, spasticity and hyperreflexia may also be seen. Radiologically, magnetic resonance imaging is required for diagnosis, although decreased attenuation within the spinal cord can be seen on computed tomography, and the cord itself may be swollen, normal-sized or atrophic.3 On magnetic resonance imaging, a cystic area of low signal intensity on T1- and high signal intensity on T2-weighted images is typical. 3,6 A cerebrospinal fluid flow void (low signal on T2) within the cavity from cerebrospinal fluid pulsations may be seen, and the cavity is often beaded due to multiple incomplete septations.3 No enhancement will be seen after intravenous injection of contrast medium, and this can help in differentiation from scar or tumour. Occasionally, a change in shape and size of the cord may be seen with a change in position.3
The treatment of neuropathic joints includes anti-inflammatory therapy and splinting, with few indications for surgery.1 Management is aimed at maintaining a functional arc of motion through physical therapy aimed at regaining muscle strength and the use of orthoses for support.2 Operative treatment can be performed for nerve compression, and fractures may need internal fixation2 or arthrodesis.5 Magnetic resonance imaging has an important evaluative role for potential surgery. The treatment of post-traumatic syringomyelia is surgical in many cases and involves insertion of a syringosubarachnoid shunt with cord untethering and duraplasty if necessary. Magnetic resonance imaging is essential to determine extent of the syrinx and define any septations preoperatively.