Abstract
Background: Most duodenal injuries are managed with primary repair, but the degree of duodenal-wall injury may threaten the integrity of the primary repair. Therefore, we evaluated whether the primary repair site could be reinforced with histoacryl glue (HAG) or HAG with an expanded polytetrafluoroethylene (ePTFE) mesh.
Methods: Grade 3 duodenal injury in the second portion of the rat duodenum was chosen as a standard trauma model. Thirty-three male rats were divided into sham (n = 3), 2-layer primary repair (n = 10), 1-layer primary repair plus HAG application (n = 10) and ePTFE attached with HAG over the 1-layer primary repair site (n = 10) groups. Ten-day survival, adhesion grades and histological assessment were taken as outcome measures.
Results: A significant survival advantage was identified in the group that had an ePTFE graft attached with HAG over a 1-layer repair when compared with the group that had a 2-layer primary repair. Adhesion grades were found to be particularly increased in the group that had an ePTFE graft attached with HAG over the primary repair site, moderately increased in the primary repair plus HAG application group and lower in the 2-layer primary repair group. ePTFE graft application was found to be beneficial to coverage of the HAG-dependent empty spaces in the serosal layer.
Conclusions: A primary repair site after duodenal trauma or a difficult duodenal stump can be reinforced with the application of HAG or ePTFE graft implantation with HAG.
Duodenal trauma presents a special challenge to surgeons. The incidence of duodenal injuries is reported to be 3.7%–5%.1,2 Penetrating trauma is the most common cause of duodenal injuries (77.7%).1,3 The vast majority of duodenal injuries are managed by simple procedures such as débridement and primary repair or resection and anastomosis.4–7 Early operative treatment is the rule rather than the exception in penetrating duodenal trauma.4,7,8 For up to grade 3 injuries (early recognition, no associated pancreatic or biliary injuries, and disruption of 50%–75% of the circumference), primary closure of the duodenal defect is a simple method for surgeons.9–11 In duodenal stab wounds, closure of the defect may be problematic in the presence of duodenal-wall edema.
Since 1959, the application of cyanoacrylates in various surgical settings has been well documented.12–14 Study of the utility of cyanoacrylate derivatives when applied to the gastrointestinal tract has been limited to the repair of various types of fistulas.15–17 Many studies suggest that expanded polytetrafluoroethylene (ePTFE) graft may well be an acceptable membrane with which to repair hollow viscus defects.18,19 In this study, our aim was reinforcement of the suture line with cyanoacrylate derivatives in grade 3 duodenal injury. For this purpose, histoacryl glue (HAG) or HAG with an ePTFE graft (Gore-Tex, W.L. Gore & Associates, Flagstaff, Ariz.) were applied over the single-layer duodenal suture line and their effects on tissue healing processes evaluated.
Methods
Thirty-three adult, male Sprague–Dawley rats weighing 200–250 g were used. All the studies were carried out under the guidelines of Selçuk University Institutional Animal Ethics Committee. The rats were kept at room temperature and provided with free access to standard chow and tap water.
The rats were divided into 4 groups as follows: sham operation group (n = 3), 2-layer primary repair group (group A, n = 10), primary suture plus HAG application group (group B, n = 10) and primary suture and ePTFE attached with HAG group (group C, n = 10).
For 12 hours before the experiments, the animals were deprived of food but not water. All surgical procedures were performed under ketamine (Ketalar, Parke–Davis Inc., Morris Plains, NJ), 40 mg/kg, anesthesia. A midline abdominal incision was used to expose the second part of the duodenum. All animals underwent grade 3 duodenal injury, which was performed with a cold blade in a star-shape manner, extending up to 75% of the circumference of the duodenum. In addition, wound edges were crushed with pick-up forceps after opening the duodenal lumen. To achieve sufficient bile contact with the wound edges, we waited for 5 minutes. During this period, other sites of the peritoneum were kept from bile contact.
After creating a grade 3 duodenal injury, the defects were repaired. The type of duodenal injury was developed in our experimental pilot study, in which the animals had a 60% mortality rate after 1-layer primary repair (unpublished data). Both the induction of injury and 1-layer repair of the defects were randomly performed by the same surgeons. In the 2-layer primary repair group, defects were closed with continuous 4/0 Vicryl sutures in the inner layer and interrupted 4/0 Vicryl sutures in the outer layer. HAG was applied over the 1-layer continuous suture line in group B. In group C, a 1 × 0.5-cm ePTFE graft was laid over the 1-layer continuous suture line with HAG. Grade 3 injury to the second portion of the duodenum without repair was performed in the sham group. Abdomens were closed with interrupted 3/0 silk sutures.
The animals were returned to their cages after the operation and were kept at an ambient temperature of 22°C. Cefazolin, 40 mg/kg per day intramuscularly, was administered to the animals for 5 days after the operation. They were fed with a standard rat diet for a period of 10 days. On the eleventh day, the rats were anesthetized with ketamine, 40 mg/kg, again. The abdomen was opened with a midline incision, and the abdominal cavity was examined with respect to the presence of leakage, abscesses and adhesions. Adhesions were staged using the method described by Mazuji and colleagues.20 Repaired segments of the duodenum were excised and fixed in 10% formalin. After fixation they were cut in a transverse plane at multiple levels, embedded in paraffin and processed for light microscopy. All sections were stained with hematoxylin–eosin dye.
The 10-day survival rates of each group were compared with others using a Fisher–Freeman–Halton test, which is a generalization of Fisher’s exact test for a 2 × 2 contingency table to an r × c contingency table. We considered p values less than 0.05 to be significant.
Results
Patency of the anastomosis
In the sham group, all the rats died of abdominal sepsis at postoperative days 2, 2 and 3 (Table 1). As expected, the primary repair group had a better 10-day survival rate than the sham group, though this was not statistically significant (p = 0.23). In group A, anastomotic leakage was detected in 5 animals, which died at postoperative days 3, 4, 4, 5 and 6. The rats in groups B and C had significantly better survival rates than the sham group (p = 0.03 and p = 0.003, respectively). In group B, anastomotic leakage was detected in 2 animals, which died at postoperative days 4 and 5. No leakage or abscess was identified in the animals belonging to group C. Ten-day survival rates in the primary repair group were found to be lower than those in group B, but the difference was not statistically significant (p = 0.35), and were found to be significantly lower than those in group C (p = 0.03). Patency of the anastomosis was excellent except for those animals in groups A and B that died. There was no leakage, abscess or deep surgical site infection seen in the peritoneum. In group C, the ePTFE grafts were found to be in place with strong integrity of the graft into the intestinal serosa on the tenth postoperative day.
Ten-day survival rates found within groups
Adhesions
In general, adhesions were identified particularly on the right upper quadrant of the abdomen on the tenth day. Dense adhesions were identified in group B. The liver surfaces closest to the graft-applied site could not be separated easily from the duodenum in group C rats. In the primary repair group, adhesion grades were lower than in the other groups (Table 2).
Adhesion grades of the animals
Histopathological examination
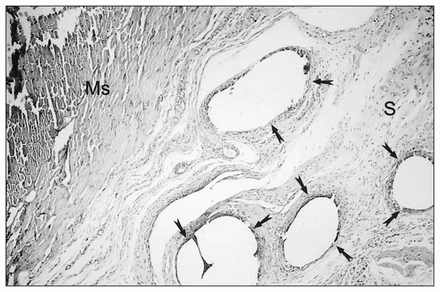
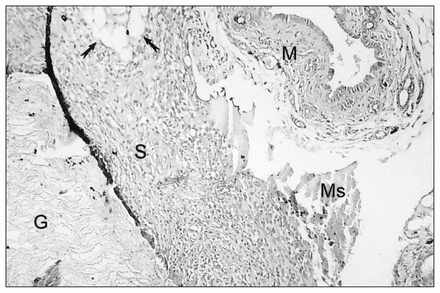
On examination of the transverse sections of the duodenum with light microscopy, repair sites in group A showed minimal inflammation and coagulative necrosis. There was normal mucosal and serosal continuity. In group B, repair sites showed moderate foreign body giant cell–type granulomatous inflammation in the serosa. Inflammation intensity was decreased in the muscularis layer and mucosa. Glue appeared as empty spaces in the serosal layer and was observed mainly around the suture line due to partial dissolution during processing (Fig. 1). In group C, empty spaces in the serosa were identified under the ePTFE patch (Fig. 2). Mucosa and muscularis layers were intact and showed minimal inflammation.
Photomicrograph on the 10th day of a closure site to which histoacryl glue had been applied. Most of the glue has dissolved during tissue processing, leaving empty spaces (marked with tailed arrows) surrounded by foreign body giant cell–type granulomatous inflammation in the serosa (S). The muscularis layer of the duodenum (Ms) was intact (hematoxylin–eosin stain, original magnification ×100).
Photomicrograph on the 10th day of a closure site to which an expanded polytetrafluoroethylene graft had been attached with histoacryl glue. Empty spaces in the serosa (S) due to histoacryl application are marked with tailed arrows. The serosa layer was completely covered with graft tissue (G). Foreign body giant cell–type granulomatous inflammation was found in the serosa layer. Mucosal (M) and muscularis (Ms) layers of the duodenum were observed in their natural continuity (hematoxylin–eosin stain, original magnification ×32).
Discussion
In recent case series, most parts of the duodenal wounds are grades 2 and 3. Primary repair or duodenorrhaphy has been used successfully in 70%–85% of duodenal wounds.2,4,5 Duodenal fistula constitutes a major cause of morbidity and mortality in patients treated with primary repair. The overall incidence of fistula formation is reported to be 2%–16%.3,4,8,21 In a recent series reported by Timaran and colleagues,22 duodenal fistula formation was observed in 3.9% of patients treated with primary closure. The mortality rate in the primary repair group was reported to be 5.3% in this study. In addition, duodenal fistulas were responsible for the deaths of 3 of 7 patients in the primary closure group.22 Overall mortality rates for duodenal injuries are reported to be between 16% and 20%, and associated injuries are responsible for most of the deaths.8,10,22–24 However, it can be clearly said that some problems cannot be resolved in cases treated with primary closure.
For reinforcement of the duodenal suture line, a 2-layer closure technique is accepted generally by most surgeons.3,10,21,22,25 The severity of chemical peritonitis and the type of injury (shotgun, stabbing, etc.) affect the fragility of the duodenum and the safety of the closure patency.4 We conducted a study to examine the effectiveness of various methods of duodenal closure after we found an injury model that reached 60% mortality after 1-layer primary repair in a previous study. When compared with data from our pilot study, 2-layer repair of the severely damaged duodenum did not decrease the leakage rate from the repair site. Enlargement of the defect or suture line dehiscence follows insufficient 2-layer repair. In the rat duodenal injury model, 2-layer closure of the defect may compromise the lumen in addition to increasing edema and ischemia of the duodenal wound. Thus, we would like to emphasize the greater possibility of leakage in the 2-layer repair group.
Several procedures have been described to avoid or minimize duodenal suture-line dehiscence. Some authors have advocated the use of a serosal patch of the jejunum to cover the duodenal suture line.25–28 However, McInnis and colleagues29 did not find any significant difference between outcomes for patients with primary closure and patients with serosal patch. On the other hand, some authors have examined the effects of decreased intraluminal pressure on closure healing. Stone and Fabian30 recommended routine duodenal decompression with triple ostomies (gastrostomy, tube duodenostomy and tube jejunostomy) after primary closure. However, other investigators could not validate these observations.7,31 Currently, the utility of duodenal decompression techniques is viewed as more restricted. Ozlem and colleagues32 attempted to resolve the problem of duodenal suture-line dehiscence with ePTFE graft implantation over the large duodenal defect in an experimental model. Neither leak, nor obstruction was observed in the graft-secured group. In another study by Astarcioglu and coworkers,19 primary repair, jejunal serosal patch, Rouxen-Y duodenojejunostomy and ePTFE patch repair techniques were compared with others in the model of rat duodenal injury. These repair modalities were associated with better survival rates than no treatment or primary repair techniques. Complete coverage of the ePTFE grafts by neomucosa was observed at about 16 weeks after surgery. Investigators demonstrated that ePTFE graft implantation might be introduced without primary closure in cases with large duodenal defects. However, surgeons are rarely faced with largetissue defects of the duodenum in clinical settings. In addition, the integrity of the primary duodenal closure is a more common demand.
Cyanoacrylate derivatives have been used for approximating of tissue edges in many medical disciplines for a long time.12–16,33–35 Clinical applications started with sutureless skin closure.13,14 Afterwards their usage in vascular surgery, ophthalmology, interventional radiology, neurosurgery, otolaryngology and plastic surgery emerged.16,33–37 Histotoxicity of the cyanoacrylate derivatives is characterized by inflammation, tissue necrosis, granuloma formation and wound breakdown.17 The rate of degradation of the compound is affected by the severity of the damage: the slower the degradation rate, the less the histotoxicity.38 For this reason, slowly degrading compounds such as butyl-2-cyanoacrylate (histoacryl) are more commonly preferred. Clinical application sites of cyanoacrylate derivatives on the gastrointestinal tract have been studied since the early seventies. In an experimental study, Orda and colleagues39 adhered autoplastic peritoneal patches to hepatic and splenic wounds using histoacryl for hemostasis. Histological examinations revealed extensive granulocyte infiltration on the second day. On the seventh day of experiment, no signs of acute reaction in the epithelial and mesenchymal tissues were observed, and complete recapsulation of the liver parenchyma was noted in this study.39 Cekirge and colleagues16 hindered bile leakage and bleeding after transparenchymal endoprosthesis placement successfully with application of HAG. In addition, recent reports support HAG application for enterocutaneous fistulas.15,40 Despite its well-documented beneficial effects in microvascular anastomosis, HAG was not used for reinforcement of the suture line after intestinal anastomosis until recently.33,34 Lemaire and colleagues33 found this sutureless microvascular anastomosis technique to be useful. Both early and late patency rates of the anastomoses were high. However, minimal histotoxicity of HAG was observed. In particular, vascular anastomoses to which HAG had been applied showed foreign body giant cell–type granulomatous inflammation and large acellular spaces resulting from dissolution of glue in the adventitia at the first week after anastomosis. In addition, in cases with extensive contact of HAG with the vessel wall, coagulative necrosis was observed in the media layer.33 Middleton and coworkers41 observed an inflammatory reaction in vascular anastomosis performed with HAG that was comparable to suture-only anastomoses in the adventitia. Besides this progress observed in vascular surgery, De La Fuente and colleagues35 repaired a large left ventricular defect with a Teflon patch attached in place with HAG. In spite of the violent pressure on the replaced graft in the ventricle, the patient was discharged in good condition without any complication. Similar microscopic findings were clearly observed in our experiments after HAG application to those seen on vascular anastomosis. Some questions arise about its utility because of intensive adhesion formation found around the application site in the early postoperative period.
In addition to the HAG application, an ePTFE graft was used in the current study to strengthen the duodenal suture line. Instead of the previous experimental studies in which an ePTFE graft was stitched to the duodenum with polypropylene sutures, the graft was attached over the duodenal suture line with HAG. With this technique, the traumatic effects of suturing on the duodenal wall were minimized. In addition, acellular spaces in the serosa derived from HAG degradation were covered with inert graft. Our results show that the ePTFE graft reinforced the duodenal suture line as a barrier against leakage. Infection secondary to graft tissue is another concern. The stimulation of fibroblast growth is essential for normal healing and tissue integration of biomaterials. The local elevation of proinflammatory mediators dependent on bacterial products has been shown to inhibit fibroblast growth.42 Both experimental and clinical studies have considered the potential tendency of vascular ePTFE grafts to infection. 43–45 In the clinical study by Deneuville,46 the onset of secondary graft infection and of postoperative graft infection were delayed 7.8 months and 1.8 months, respectively. However, colonization of the vascular ePTFE grafts was demonstrated as early as a week after induced bacteremia.43 In contrast to experiences in vascular surgery, application of ePTFE grafts into the alimentary tract wall seems successful on an experimental basis.19,32,47 There were no reported infections secondary to ePTFE grafts. We also did not observe surgical site infection in graft-applied animals. Therefore, excised grafts were not put into culture. But, colonization on the graft secondary to contamination with gastrointestinal flora is a major concern and should be investigated in future studies.
In conclusion, dense adhesion formation on the operative field after HAG application is a major limiting factor of the technique. However, HAG or ePTFE attachment for the reinforcement of the suture line may be used without reducing the calibre of the lumen after grade 3 duodenal injury. Both techniques are preferable for the difficult duodenal stump closure as well. Further studies of this subject are needed for clinical acceptance of this technique.
Acknowledgements
Statistical analyses were done by Dr. Erdem Karabulut of the Department of Biostatistics, Hacettepe University, School of Medicine Sıhhiye, Ankara, Turkey.
Footnotes
Part of this study was presented at the 5th European Congress of Trauma and Emergency Surgery, Oct. 1–5, 2002, Istanbul, Turkey.
Competing interests: None declared.
- Accepted January 31, 2005.