Abstract
Background: Fat embolism syndrome (FES) is a potentially lethal condition most commonly seen in polytrauma patients with multiple long-bone fractures. Treatment has centred around supportive care and early fracture fixation. Several small clinical trials have suggested corticosteroids benefit patients with FES, but this treatment remains controversial. Our objective was to determine the effect of corticosteroids in preventing FES in patients with long-bone fractures.
Methods: We conducted a meta-analysis of published studies of patients with long-bone fractures who were randomly assigned to groups receiving corticosteroids or standard treatment for the prevention of FES (1966–2006). Data were extracted on quality, population, intervention and outcomes. Our primary outcome was the development of FES. We used random-effects models to pool results across studies, assessing for study heterogeneity.
Results: Of the 104 studies identified, 7 met our eligibility criteria. Overall, the quality of the trials was poor. Our pooled analysis of 389 patients found that corticosteroids reduced the risk of FES by 78% (95% confidence interval [CI] 43%–92%) and that only 8 patients needed to be treated (95% CI 5–13 patients) to prevent 1 case of FES. Similarly, corticosteroids significantly reduced the risk of hypoxia. We found no differences in the rates of mortality or infection. Rates of avascular necrosis were not reported in any of these studies.
Conclusion: Evidence suggests that corticosteroids may be beneficial in preventing FES and hypoxia but not mortality in patients with long-bone fractures. The risk of infection is not increased with the use of cortisosteroids. However, methodological limitations of these trials necessitate a large confirmatory randomized trial.
Fat embolism syndrome (FES) is a potentially lethal condition, seen most commonly in polytrauma patients, particularly those with multiple long-bone fractures.1,2 It typically affects those in their second or third decade of life, which is consistent with a typical trauma population. The incidence of FES following trauma has been reported to be between 1% and 10%,3–5 and evidence of fat emboli has been seen in as many as 90% of patients with long-bone fractures.6 In addition to blunt trauma, FES has been implicated in orthopedic procedures that involve reaming of an intramedullary canal and insertion of a pedicle screw as well as many other nonorthopedic conditions.7–14 The overall mortality of FES is historically thought to be as high as 10%–20%.3,4,15
Diagnosis of FES is based on a clinical triad including respiratory, neurologic and cutaneous manifestations.16,17 Treatment of FES has centred around supportive care and early fracture fixation.18–21 Pharmacologic treatment of FES remains controversial, despite the previous use of a wide variety of agents apart from corticosteroids.22–26 Since the development of FES has been attributed to the release of free fatty acids from the lysis of triglycerides, which cause endothelial damage, vasculitis and direct toxicity to pneumocytes, corticosteroids have been proposed to limit the elevation of free fatty acid levels, thus blunting the inflammatory response.20,27 Although corticosteroids have been shown to be beneficial for prevention of FES in several small trials, concern for infection and even death has limited its use in trauma patients. Similarly, corticosteroid use in critically ill patients with acute respiratory distress syndrome (ARDS), traumatic head injury and spinal cord injury also remains controversial.28–30 Recently, the use of “low-dose” corticosteroids has shown significant benefit for patients early in the course of ARDS.31 It is therefore unclear whether the use of corticosteroids can prevent the development of FES in patients with multiple long-bone fractures. The main objective of our study was to evaluate the effect of corticosteroids versus standard treatment on the risk of FES in patients with long-bone fractures. Secondarily, we sought to examine the rate of mortality and complications following administration of corticosteroids, with regard to infection and avascular necrosis.
Methods
We conducted a meta-analysis of randomized trials to determine the effect of corticosteroids in preventing FES in patients with long-bone fractures.
Eligibility criteria
Our study population included all randomized and quasi-randomized (alternate sequence assignment) trials of patients with at least 1 long-bone fracture treated with either prophylactic corticosteroid intervention or control. We excluded all trials including patients with clinically important associated head, chest or abdominal injuries or trials in which no attempt was made to assign treatment based on chance (not randomized). Our primary outcome was the development of FES. Recognizing that this diagnosis relies on clinical interpretation that is subject to bias, we decided a priori to rely on the authors’ own clinical criteria for diagnosis (based on a combination of respiratory, neurologic and cutaneous manifestations). In the case of multiple diagnostic criteria within a single study, we chose to use the most stringent clinical criteria specified. Owing to the subjective and variable nature of clinical diagnostic criteria, we also assessed objective findings suggestive of FES, including the presence of hypoxia as confirmed by arterial blood gas measurements and the presence of the classic petechial rash. Our secondary outcomes included overall mortality, any major infective complication and the development of avascular necrosis within the follow-up period.
Study identification
To locate relevant clinical studies, we searched computerized databases (MEDLINE, EMBASE, HealthSTAR, CINAHL and Cochrane Central Register of Controlled Trials) for published studies from 1966 to September 2006. We used search criteria for corticosteroids (medical subject headings “corticosteroids,” “glucocorticoids,” “methylprednisolone,” “adrenal cortex hormones”) and fat embolism (medical subject heading “fat embolism syndrome”) and only limited our search to human studies. We included both English and non-English studies. Additionally, we performed exhaustive manual searches of indices of major orthopedic journals (Journal of Bone and Joint Surgery[Am], Journal of Bone and Joint Surgery[Br], Journal of Orthopaedic Trauma, Clinical Orthopaedics and Related Research, Journal of Trauma, Injury Infection and Critical Care) and published abstracts of major meeting proceedings (American Academy of Orthopaedic Surgeons, Orthopaedic Trauma Association and Canadian Orthopaedic Association) from 1966 to 2006. We also searched bibliographies of major orthopedic textbooks.32–34 We again searched by hand bibliographies of selected relevant articles for further relevant references.
We first applied the eligibility criteria to both titles and abstracts of all articles identified from the initial search by 2 reviewers independently (S.S.B., M.B.). We then retrieved for review those articles that were deemed potentially eligible. We prepared the methods section for 2 other independent reviewers (M.D.M., E.H.S.) with blinding of publication date, journal, author, institution and results. We then applied the eligibility criteria to the potentially eligible studies by these reviewers. Any disagreements were resolved by the 2 other reviewers (S.S.B., M.B.).
Data extraction and quality assessment
We retrieved each identified study in full but prepared studies for review with blinding of publication date, journal, author and institution. Two authors (M.D.M., E.H.S.) independently abstracted all data. We abstracted in a standardized fashion relevant data on population, interventions and clinical parameters, as well as data on primary and secondary outcomes. We also assessed each study for quality by examining the reporting of randomization methods and the blinding of outcome assessors. Additionally, we assessed quality using a 21-point assessment scale35 converted to a score out of 100, with a higher score indicating better quality.36 This widely used scale grades the reporting of studies with respect to eligibility criteria, adequacy of randomization, description of therapies, assessment of outcomes and statistical analysis. The other 2 authors (S.S.B., M.B.) then reviewed all abstracted data to ensure consistency. We also explicitly assessed whether the randomization process was concealed and whether patients, clinicians, outcomes assessors and data analysts were blinded.
Statistical methods
Studies included by eligibility criteria were assessed for agreement by the κ statistic, a measure of agreement correcting for chance. We considered an a priori agreement of greater than 0.65 to be adequate.37 We then pooled data across studies using random effects to model the variability both within a study and between studies.38 We calculated risk ratios, absolute risk differences and numbers needed to treat, with their associated 95% confidence intervals (CIs) for all outcomes. We constructed funnel plots (sample size v. treatment effect) to assess for publication bias. We performed the Breslow–Day test of heterogeneity for risk differences to determine the degree of variation between studies with a significance threshold of p < 0.1.39 We further used the I2 test as a measure of heterogeneity for each outcome analysis.40 An I2 value represents the percentage of total variation across trials that is owing to heterogeneity rather than chance, and we considered an I2 value 25% or less as low and an I2 value 75% or higher as high.
Since corticosteroids for critically ill patients have varied from high-dose (≥ 30 mg/kg) to low-dose (1 mg/kg/d, tapered over 28 d) regimens,41 and we anticipated a wide time frame during which these studies were performed with variable methodological reporting, we decided a priori to perform sensitivity analyses based on dose of methylprednisolone administered (> 20 mg/kg v. ≤ 20 mg/kg) and the quality of studies (score > 50 v. ≤ 50).
Results
Literature search
We identified 104 studies from the initial search of databases, journals, meeting proceedings, textbooks and bibliographies. After applying eligibility criteria to both titles and abstracts, we found 9 studies to be potentially eligible with adequate agreement (κ = 0.87). We masked the 9 potentially eligible studies and only 717,26,42–46 met our eligibility criteria with adequate agreement (κ = 0.73). Two studies were excluded because they were both nonrandomized comparisons (no intervention assignment based on chance).47,48 One of the studies that was included did not report on the primary outcome of interest; however, because the overall number of included studies was low, and the authors did report on mortality and the development of a petechial rash, it was, therefore, retained for analysis.26
Study characteristics
There was significant variation in the characteristics of the individual studies (Table 1). Inclusion and exclusion criteria were generally similar across studies; however, corticosteroid treatment regimens (doses and total durations) and use of controls differed significantly. Two of these studies were randomized by alternate sequence and thus considered “quasi-randomized.” All but 1 of the studies was published more than 20 years ago, and there was a wide range in quality between studies (score range 24–74) with the highest quality attributed to the only recent study, one that was randomized by alternate sequence.43
Characteristics of included studies
Most study patients were young men with similar fracture distributions between groups. Variation in the criteria for diagnosis of FES also varied across studies (Table 2). Oxygen tension cut-off values for the diagnosis of hypoxia, however, were relatively similar across studies (arterial partial oxygen pressure 60–67 mm Hg).
Diagnostic criteria of studies
Fat embolism syndrome
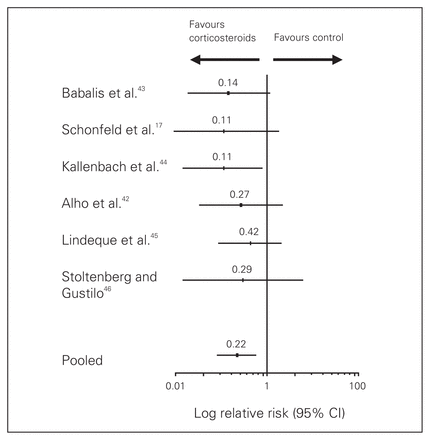
From our pooled analysis of 6 studies with 389 patients, we found that corticosteroids reduced the risk of FES by 78% (relative risk [RR] 0.22) compared with control (Fig. 1). No significant heterogeneity among the trials was identified (p = 0.67, I2 = 10%). The risk difference was found to be 13% (95% CI 8%–19%) and, furthermore, only 8 patients (no. needed to treat = 7.5) needed to be treated to prevent 1 case of FES (95% CI 5–13 patients). Funnel plots did not suggest publication bias.
Pooled results for fat embolism syndrome. CI = confidence interval.
Secondary outcomes and complications
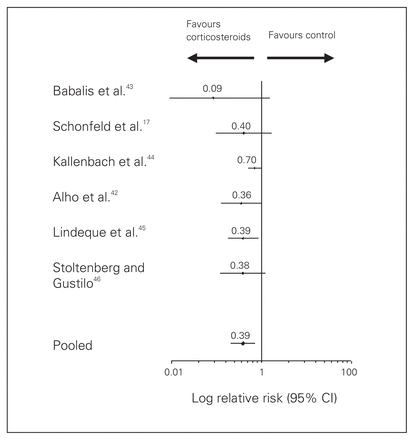
Corticosteroids reduced the risk of hypoxia by 61% (RR 0.39) compared with control (Fig. 2.). Furthermore, the absolute risk reduction for the development of hypoxia was 0.22 (95% CI 0.14–0.31) yielding a number needed to treat of only 4.5 patients (95% CI 3.2–7.1). Corticosteroid administration did not have any significant effect on the development of the petechial rash or on overall mortality. The analysis was repeated omitting the one study that did not report on primary outcome with no changes in our results. Similarly, corticosteroids were not found to affect differences in rates of infection (Table 3). Avascular necrosis was not reported as an outcome or complication in any of these studies; however, follow-up was short and the incidence of avascular necrosis may not even be detected within 1 year.49
Pooled results for hypoxia. CI = confidence interval.
Results of statistical pooling among studies
Sensitivity analysis
We performed sensitivity analyses for high- (> 20 mg/kg) and low-dose (≤ 20 mg/kg) methylprednisolone and high-(score > 50) and low-quality (score ≤ 50) studies (Table 4). Low-dose methylprednisolone (6–9 mg/kg) demonstrated an improved benefit in the prevention of FES over high-dose regimen studies (≥ 30 mg/kg) as did higher quality studies (score > 50) over poorer quality studies.
Sensitivity analysis for fat embolism syndrome among pooled studies
Based on the degree of variability in study methods, we decided a posteriori to perform sensitivity analyses for studies with and without blinding of assessors and the type of randomization (truly randomized v. quasi-randomized). All subgroups of studies maintained the beneficial effect of corticosteroids in the prevention of FES with the exception of those of poor quality and with inadequate blinding of assessors.
Discussion
With a substantial prevalence in multiple long-bone fracture patients and a significant mortality risk, FES remains a potentially lethal problem. The clinical use of corticosteroids in preventing FES is still presently controversial. In our pooled analysis of 389 patients, we found that corticosteroids significantly reduced the risk of FES in multiply injured patients. This benefit is at least as much as a 43% (but up to a 92%) reduction in the risk of FES. However, the study sample sizes were small, the quality of the primary studies was relatively low, and the number of outcome events few, suggesting caution in the interpretation of our findings. We did not identify any significant differences in mortality or infection with the administration of corticosteroids. We did not assess incidence of avascular necrosis.
The routine use of corticosteroids in other critically ill patients also remains controversial. Earlier studies of high-dose corticosteroids demonstrated no survival benefit in patients with ARDS and even raised the possibility of the development of superinfections and other complications.50–53 High-dose steroids in the treatment of patients with severe head injury and spinal cord injury have raised similar concern.28,30 More recently, several randomized trials have shown lower-dose regimens with prolonged treatment to improve lung function and reduce ventilation time for patients with ARDS.29,31,54–56
Fat embolism syndrome, although similar to ARDS, is a distinct clinical entity most commonly seen in patients with multiple long-bone fractures.57,58 Results from trials of patients with ARDS, and certainly head or spinal cord injury, may not directly extrapolate to patients with FES. Furthermore, it has been shown that owing to differences in hepatic metabolism between the sexes, female patients demonstrated faster clearance and lower plasma concentrations of methylprednisolone compared with men, thus resulting in the possibility of a differential effect of corticosteroids in patients with FES (mostly male) compared with ARDS.59
The best clinical evidence for the use of corticosteroids in preventing FES comes from these multiple small randomized trials. Meta-analysis, a method of systematic review that uses statistical methods to combine data from multiple trials, increases the “effective” study power and increases the precision of the final estimates of treatment effect. In the case of FES, our findings represent the best available clinical evidence for the use of corticosteroids.
As in any other systematic review, the quality of the results depends on the quality of the individual studies included. The included studies span a wide time frame (1977–2004) and demonstrate variable quality (score range 24–74). The range of publication dates raises concern since dramatic changes in management have occurred over this time span, thus likely lowering the overall rates of FES, which, in turn, may limit the value of corticosteroids. For example, avoidance of higher tidal volumes and airway pressures, conservative fluid management strategies and early stabilization of long-bone fractures have all improved outcomes for these patients.18,19,21,60–62 Furthermore, newer techniques in intramedullary nailing, such as the reamer–irrigator–aspirator, may further diminish the embolic load and improve outcomes.63,64 Nonetheless, our findings are strengthened by the inclusion of randomized controlled trials that compare equivalent groups at the same time treated with otherwise similar techniques, thus ensuring that outcomes measuring changes in rates, in particular relative risks, remain valid. Differences in diagnostic criteria also varied between studies, making direct comparison problematic. To best avoid the problem of subjective diagnosis of FES, we also included a more objective measure, hypoxia, a common condition seen in patients with FES. Our analysis demonstrated that the administration of corticosteroids also significantly reduced the risk of hypoxia. Dosing regimens also varied between studies from a total dose of 6 mg/kg up to 90 mg/kg. Although the beneficial effect of corticosteroids was seen in both high- and low-dose regimens, the lower-dose subgroup showed an improved benefit over higher doses, in keeping with findings from recent randomized trials in ARDS.31 To validate our findings and ensure that variability across studies was not a significant factor, heterogeneity was explored in our analyses and did not demonstrate statistically significant differences.
Furthermore, duplicate assessment of study validity and data abstraction, and a comprehensive search for eligible studies improves the quality of meta-analysis. Despite this comprehensive strategy, we may have missed studies that might have met our inclusion and exclusion criteria, or missed studies owing to publication bias, thus affecting our overall results. Although our funnel plots suggested no such bias, our sample of trials was small. A further concern is the lack of information on the incidence of avascular necrosis. There exists a clear association between corticosteroid administration and the development of avascular necrosis, and even short-term corticosteroid administration can cause avascular necrosis 2–5 years later.49 Future studies are needed to better assess the long-term effects of corticosteroid administration in this patient population.
Conclusion
In summary, the current evidence suggests that corticosteroids may prevent FES in patients with long-bone fractures. We found no significant differences in mortality or rates of infection with the use of corticosteroids. Based on our findings, which included mostly older and low-quality studies, we would not currently recommend a change in practice. Our findings do, however, provide compelling rationale for the re-evaluation of corticosteroid use in the multiple-trauma setting with modern nailing techniques, perioperative protocols, standardized definitions of FES and a low-dose protocol (e.g., 6 mg/kg over 48 h in 6 divided doses). Ultimately, a large confirmatory randomized trial will provide the necessary evidence to guide patient care.
Footnotes
Presented as a poster at the Orthopaedic Trauma Association Annual Meeting, Ottawa, Ont., Oct. 20–22, 2005, and at the 10th Conference of the International Society for Fracture Repair, Adelaide, Australia, May 21–24, 2006. Presented from the podium at the Canadian Orthopaedic Association Annual Meeting, Halifax, NS, Jun. 1–3, 2007, and at the Canadian Surgical Forum, Canadian Association of General Surgeons Annual Meeting (American College of Surgeons, Committee on Trauma Resident Paper Competition), Toronto, Ont., Sept. 6–9, 2007.
- Accepted May 15, 2008.