Abstract
Background: With a growing demand for endoscopic services, the role of anesthesiologists in endoscopy units must be reassessed. The aim of this study was to compare patient outcomes in non–anesthesiologist-administered propofol (NAAP) versus anesthesiologist-administered propofol (AAP) during routine endoscopy.
Methods: We systematically searched MEDLINE, CINAHL, Embase, Web of Science, CENTRAL and the grey literature for studies comparing NAAP and AAP. Primary outcomes included endoscopy- and sedation-related complications. Secondary outcomes included measures of endoscopy quality and of patient and endoscopist satisfaction. We reported treatment effects using random-effects models.
Results: Of 602 articles identified, 5 met the inclusion criteria. Most studies included only patients with an American Society of Anesthesiologists (ASA) classification of I or II. Non–anesthesiologist-administered propofol did not result in increased rates of airway intervention (odds ratio [OR] 1.07, 95% confidence interval [CI] 0.29 to 3.95; 3443 patients) or hypotension (OR 1.47, 95% CI 0.40 to 5.41; 17 978 patients) but did result in higher rates of bradycardia (OR 3.68, 95% CI 1.65 to 8.17; 17 978 patients). Nonanesthesiologists administered lower propofol dosages than anesthesiologists (mean difference −61.79, 95% CI −114.46 to −9.12; 3443 patients), and their patients more commonly experienced awareness with recall (OR 19.99, 95% CI 7.88 to 50.76; 2090 patients). However, NAAP neither compromised patient willingness to repeat the procedure (OR 0.42, 95% CI 0.10 to 1.83; 2367 patients) nor lengthened total procedure time (mean difference −0.08, 95% CI −3.51 to 3.34; 2367 patients).
Conclusion: Endoscopists may safely administer propofol without compromising procedural quality in patients classified as ASA I or II undergoing routine endoscopy. The results of this meta-analysis are limited by a lack of available high-quality studies. Further, large-scale studies are needed for definitive conclusions.
The demand for gastrointestinal endoscopic procedures is increasing dramatically, and even with 1.6 million endoscopic procedures performed annually in Canada, demand continues to exceed supply.1,2 This growing need for endoscopic services is driving the search for more efficient and less costly procedures that minimize patient discomfort without compromising their safety.3,4 Sedation is an important aspect of endoscopic procedures as it improves the quality of the examination, patient satisfaction, and patient adherence to screening and surveillance regimens.4–6 With the vast majority of patients preferring to receive sedation, the cost that sedation adds to endoscopic procedures must also be considered.
The rate of involvement of anesthesiologists in endoscopic procedures depends on a variety of factors including patient characteristics, choice of sedative, and local institutional practices and policies. Traditionally, sedation has been provided through the combination of a narcotic and a benzodiazepine, but, more recently, there has been a shift toward the use of propofol.1,7,8 With a rapid onset (30–45 s) and short duration of action (4–8 min), propofol is the ideal agent for short outpatient procedures.9 Additional benefits compared to benzodiazepines and narcotics include faster recovery, earlier discharge, improved postanesthesia recovery, reduced postanesthesia nausea and vomiting, and, ultimately, greater patient satisfaction.7,10
Historically, propofol has been administered by anesthesiologists as per US Food and Drug Administration guidelines owing to its potential for deeper levels of sedation and lack of reversal agent.11–13 Furthermore, propofol is known to produce cardiorespiratory effects including decreased ventilatory response to hypoxia along with decreased systemic vascular resistance and arterial blood pressure.14,15 However, the push toward containment of health care costs has brought into question the sustainability of anesthesiologist-administered propofol (AAP) in healthy patients at low risk undergoing routine endoscopic procedures.12,16–18 In fact, non–anesthesiologist-administered propofol (NAAP) has already been endorsed and widely implemented in several European countries.17,19 In 2008, Singh and colleagues7 conducted a systematic review on the safety of NAAP but identified only 1 study comparing NAAP to AAP. In a 2009 study, Rex and colleagues20 searched for published and unpublished cases of endoscopist-administered propofol but did not make a comparison to control cases. Since then, several randomized and nonrandomized studies have been conducted on this topic. In a 2015 comparative meta-analysis, the authors concluded that NAAP compares favourably with AAP for patients undergoing advanced endoscopic procedures.18 However, the question remains largely unanswered for routine endoscopic procedures. Thus, the purpose of this systematic review was to evaluate whether patient safety and procedure quality are compromised when nonanesthesiologists (i.e., endoscopists) administer propofol in routine upper or lower gastrointestinal endoscopy.
Methods
Literature search strategy
This systematic review was synthesized following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline.21 We searched MEDLINE, CINAHL, Embase, Web of Science and CENTRAL for published English-language studies from the time of database conception until May 2016. We used various combinations of the following medical subject headings: “Anesthesia,” “Anesthesiology,” “Nurse Anesthetists,” “Surgeons,” “Propofol,” “Deep Sedation” and “Endoscopy.” We also searched ClinicalTrials.gov and ProQuest dissertations for unpublished and ongoing studies. We individually reviewed the references of all systematic reviews of similar topics, as well as the references of our own included studies. For studies with no available text, we requested for full access from their respective authors. The literature search strategy can be found in Appendix 1, available at canjsurg.ca/008117-a1.
We included studies conducted in any adult population (≥ 18 yr) undergoing upper or lower gastrointestinal endoscopy. The intervention of interest was administration of propofol by an endoscopist or by a nurse under the guidance of an endoscopist. The study had to have a control group in which propofol was administered by an anesthesiologist or a nurse anesthetist. As such, we searched for randomized controlled trials (RCTs), prospective and retrospective cohort studies, case–control studies and cross-sectional surveys. We excluded studies with incomplete outcome reporting only after contacting the respective authors for complete data. We also excluded studies that included advanced and specialized endoscopic procedures such as endoscopic retrograde cholangiopancreatography, upper endoscopic ultrasonography, deep small intestinal enteroscopy and endoscopic surgery. Every step of the screening process was done by 2 independent reviewers (J.D. and C.T.), with a third party (R.F.) available for resolving discrepancies.
Data extraction
We extracted onto a standardized form information such as study design, demographic and baseline characteristics, and primary and secondary outcomes. Our primary outcomes of interest were endoscopy- and sedation-related complications including airway intervention, hypotension, bradycardia, hypoxia, composite outcomes of cardiopulmonary events, gastrointestinal perforation, bleeding and death. Secondary outcomes included awareness with recall, total amount of propofol administered, total procedure time, patient and endoscopist satisfaction, time to recovery, pain relief, cecal intubation, polyp detection and cost.
Quality and risk of bias assessment
Evaluation of quality and risk of bias was carried out by 2 independent reviewers (J.D. and C.T.). For this review, the Cochrane Risk of Bias Tool22 was used to assess the quality of randomized trials. This tool employs 7 criteria for judging risk of bias. Each criterion was scored as “low risk,” “high risk” or “unclear risk,” as per published guidelines.22 We followed a previously published approach22 to formulate summary assessments of risk of bias. The Methodological Index for Non-Randomized Studies (MINORS)23 was used to assess the quality of nonrandomized studies. This tool appraises studies based on 12 different items. The items were scored as 0 if not reported, 1 if reported but inadequate, and 2 if reported and adequate. Based on this score, nonrandomized trials were categorized as low risk of bias (score of 24), moderate risk (21–24) and high risk (< 21).
Statistical analysis
We used the intention-to-treat approach. We performed meta-analyses using a random-effects model and estimated weights of included studies using the inverse variance method. We estimated pooled odds ratios (ORs) for primary and secondary dichotomous outcomes, and pooled absolute mean differences for secondary continuous outcomes, each with respective 95% confidence intervals (CIs). Heterogeneity of the data was evaluated in 2 ways. We evaluated between-study heterogeneity visually using forest plots. We assessed statistical heterogeneity using the Cochran Q test and quantified it using the I2 statistic. I2 values were categorized into 3 degrees of heterogeneity: low (< 25%), moderate (25%–75%) and high (> 75%). We performed statistical analyses using Review Manager version 5.3 (www.community.cochrane.org). For outcomes that were reported in only 1 study or had too few events to meta-analyze, we performed a narrative synthesis to summarize the results.
Results
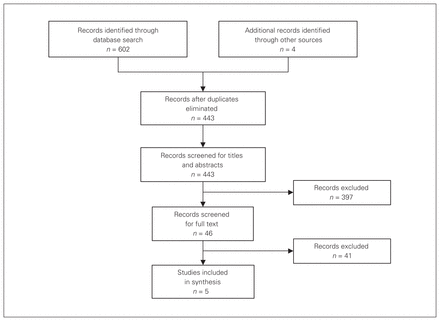
Our initial search yielded 606 articles, of which 163 were duplicates. Thus, 443 articles were screened based on titles and abstracts (Fig. 1). Following this, 46 articles were screened based on full text. This yielded 5 articles for inclusion24–28 (Table 1). The designs of the studies included RCT (2 studies26,28), prospective cohort (2 studies24,25) and retrospective cohort (1 study27). The median number of patients included in the studies was 1076 (range 90–17 611). All studies took place between 1998 and 2015. Three studies included lower gastrointestinal endoscopy only, and the other 2 included both upper and lower gastrointestinal endoscopy.
Study selection.
Characteristics of studies comparing NAAP to AAP in routine upper or lower gastrointestinal endoscopy
Baseline characteristics
In all 5 studies, the NAAP and AAP groups were comparable in age and sex (Table 2). Three studies included only patients classified as American Society of Anesthesiologists (ASA) level I or II.24,26,28 Nathan and colleagues27 included a smaller proportion of patients classified as ASA III or IV in the NAAP group than in the AAP group (9% v. 67%) and none classified as ASA I in the AAP group. Vargo and colleagues25 included patients in ASA categories I–IV and had comparable proportions in the NAAP and AAP groups. Body mass index was comparable between the 2 groups in the 2 studies in which it was reported.24,28
Baseline patient characteristics
Primary outcomes
Airway intervention
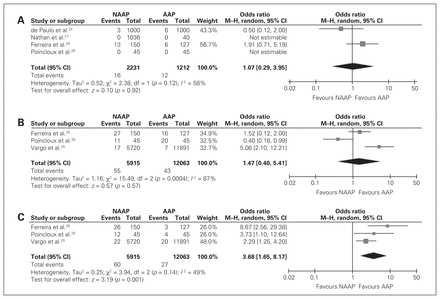
The rate of airway intervention was reported in 4 studies involving 3443 patients.24,26–28 In our pooled analysis, the odds of undergoing an airway intervention with NAAP were similar to those with AAP (OR 1.07, 95% CI 0.29 to 3.95). There was moderate heterogeneity (I2 = 58%, p = 0.1) (Fig. 2).
Endoscopy- and sedation-related complications: (A) airway intervention, (B) hypotension and (C) bradycardia. AAP = anesthesiologist-administered propofol; CI = confidence interval; M–H = Mantel–Haenszel χ2 test; NAAP = non–anesthesiologist-administered propofol.
Hypotension
The rate of hypotension was reported in 3 studies involving 17 978 patients.25,26,28 The odds of having a hypotensive episode were not significantly different between NAAP and AAP (OR 1.47, 95% CI 0.40 to 5.41). There was high heterogeneity (I2 = 87%, p < 0.001) (Fig. 2).
Bradycardia
The rate of bradycardia was reported in 3 studies involving 17 978 patients.25,26,28 The odds of experiencing bradycardia were significantly higher in patients who received NAAP than in those who received AAP (OR 3.68, 95% CI 1.65 to 8.17). There was moderate heterogeneity (I2 = 49%, p = 0.1) (Fig. 2).
Cardiopulmonary events
The rate of cardiopulmonary events was reported in 1 study involving 17 611 patients25 (Table 3). There were significantly higher rates of such events in patients who received NAAP than in those who received AAP, for both upper and lower endoscopy (upper: 1.80% v. 1.01%, p = 0.001; lower: 1.66% v. 0.86%, p < 0.001).
Rates of endoscopy- and sedation-related complications, and time to recovery
Gastrointestinal perforation and bleeding
Rates of gastrointestinal perforation and bleeding were reported in 1 study25 (Table 3). Gastrointestinal perforation occurred only in the NAAP group, for both upper and lower endoscopy (upper: 0.09%, lower: 0.06%). There was no statistically significant difference in the rate of bleeding between the NAAP and AAP groups for either upper or lower endoscopy (upper: 0.42% v. 0.16%; lower: 0.08% v. 0.16%).
Secondary outcomes
Awareness with recall
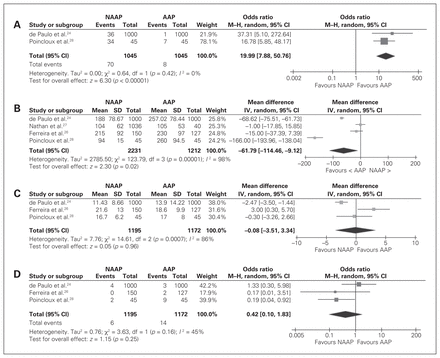
Awareness with recall was reported in 2 studies involving 2090 patients.24,28 The odds of remembering the endoscopic procedure were higher in patients who received NAAP than in those who received AAP (OR 19.99, 95% CI 7.88 to 50.76). There was low heterogeneity (I2 = 0%, p = 0.4) (Fig. 3).
Measures of endoscopy quality: (A) awareness with recall, (B) total propofol administered, (C) total procedure time and (D) patients who would not repeat the procedure. AAP = anesthesiologist-administered propofol; CI = confidence interval; IV = inverse variance; M–H = Mantel–Haenszel χ2 test; NAAP = non–anesthesiologist-administered propofol; SD = standard deviation.
Total propofol administered
The total amount of propofol administered was reported in 4 studies involving 3443 patients.24,26–28 Nonanesthesiologists administered lower dosages of propofol than did anesthesiologists (mean difference −61.79, 95% CI −114.46 to −9.12). There was high heterogeneity (I2 = 98%, p < 0.001) (Fig. 3).
Total procedure time
Total procedure time was reported in 3 studies involving 2367 patients.24,26,28 There was no significant difference in procedure length between NAAP and AAP (mean difference −0.08, 95% CI −3.51 to 3.34). There was high heterogeneity (I2 = 86%, p < 0.001) (Fig. 3).
Patient satisfaction
In 3 studies, a total of 2367 patients were surveyed about their willingness to repeat the procedure,24,26,28 which we interpreted as a measure of patient satisfaction. The odds that a patient would not be willing to repeat the procedure under the same circumstances were not different between NAAP and AAP (OR 0.42, 95% CI 0.10 to 1.83). There was moderate heterogeneity (I2 = 45%, p = 0.2) (Fig. 3).
Time to recovery
Time to recovery was reported in 1 study involving 277 patients26 (Table 3). The recovery time was significantly shorter with NAAP than with AAP (58 [SD 33] min v. 67 [SD 29] min, p = 0.03).
Pain during procedure
The rate of feeling pain during endoscopy was reported in 1 study involving 90 patients28 (Table 3). The proportion of patients who reported feeling “no pain” during the procedure was significantly lower in the NAAP group than in the AAP group (55% v. 91%, p < 0.001).
Cecal intubation and polyp detection
Rates of cecal intubation and polyp detection were reported in 1 study.28 There were no significant differences between NAAP and AAP with respect to cecal intubation (94.7% v. 96.1%, p = 0.6) or polyp detection rates (28.4% v. 23.2%, p = 0.3) (Table 3).
Other outcomes
Other outcomes of interest (cost and endoscopist satisfaction) were not reported in any of the included studies.
Risk of bias
Both RCTs26,28 had adequate random sequence generation and allocation concealment. Given the nature of the intervention, we acknowledged that it was not feasible to have blinded personnel. However, in the study by Poincloux and colleagues,28 the patients were not blinded to the intervention, and the only outcome assessor who was blinded was the investigator gathering data on patient satisfaction. In the study by Ferreira and colleagues,26 the patients were blinded to the intervention, but the outcome assessors were not. In addition, data were missing for satisfaction and pain scores, and the proportion of cases with missing data was unbalanced between the NAAP (11.3%) and AAP (22.0%) groups. Another source of bias stemmed from the selection of endoscopists in the NAAP group, which was based on experience in intensive and emergency medicine and airway management, whereas there were 9 different endoscopists in the AAP group. Both RCTs were deemed as having an overall high risk of bias (Table 4).
Quality assessment of randomized controlled trials with Cochrane Risk of Bias Tool22
All 3 observational studies24,25,27 had minimal loss to follow-up. Both de Paulo and colleagues24 and Vargo and colleagues25 had adequate control groups during similar periods. The study by Nathan and colleagues27 had selection bias in that the AAP group had a larger proportion of patients classified as ASA III than the NAAP group. There were limitations that were consistent across all 3 studies, including failure to report the use of an a priori protocol, lack of intention-to-treat analysis, and lack of blinding of participants and personnel. Vargo and colleagues25 and Nathan and colleagues27 did not report an a priori study size calculation, and, although de Paulo and colleagues24 did report this calculation, they did not specify inclusion of consecutive patients and had a vague aim statement. All 3 studies received a total score of 14, which categorized them as having a high risk of bias (Table 5).
Quality assessment of nonrandomized studies with the Methodological Index for Non-Randomized Studies (MINORS)23
Discussion
Our primary outcomes of interest were endoscopy- and sedation-related complications. We found that the overall rate of airway intervention was low among studies, ranging from 0% to 8.7% in either arm, and that the need for airway intervention was similar in the 2 groups. In 125 of the 3 studies that reported mortality rates,24,25,27 this outcome occurred in only 1 of 2166 patients. Given the infrequency of events, we provided a narrative synthesis of mortality rate across studies. In summary, there were no studies that reported a mortality benefit that favoured NAAP or AAP. The patient who died was in the NAAP group undergoing upper endoscopy.25 The patient was classified as ASA IV, and the indication for the procedure was gastrointestinal bleeding. The cause of death was unknown.
In 3 studies, rates of hypoxia and bradycardia were evaluated. The rate of hypoxia did not differ between groups, but the rate of bradycardia was significantly higher with NAAP than with AAP. Although there is no clear explanation for this, we suspect that it could be related to differences in the rate of propofol administration. There were no deaths and no need for airway intervention, which makes it difficult to conclude whether the bradycardia events were clinically significant complications that resulted in patient harm.
Other primary outcomes of interest such as cardiopulmonary events, gastrointestinal perforation and bleeding were reported inconsistently in the studies and were not amenable to meta-analysis. For instance, cardiopulmonary events were reported only by Vargo and colleagues.25 Their definition of cardiopulmonary event included chest pain, hypoxemia, transient hypoxemia, prolonged hypoxemia, bradycardia, wheezing, dysrhythmia, tachycardia, tracheal compression, hypertension, hypotension, respiratory distress, pulmonary edema and vasovagal reaction. Of note, they did not specify threshold values for transient hypoxemia, dysrhythmia, hypertension or hypotension. In addition, complications such as chest pain, wheezing, tracheal compression, respiratory distress, pulmonary edema and vasovagal reaction were reported subjectively as “occurring” or “absent” by the endoscopist or anesthesiologist. The rate of cardiopulmonary events was significantly higher with NAAP than with AAP, for both upper and lower endoscopy. Although this may reflect a true effect of who administered the sedation, it is also possible that there were subjective differences in reporting between endoscopists and anesthesiologists.
Rates of gastrointestinal perforation and bleeding were reported in only 1 study.25 Of note, the only events were in the NAAP group of both upper and lower endoscopy populations, although the frequency was very low. There were no significant differences in bleeding events between the NAAP and AAP groups. Given these outcomes, it is difficult to assess with confidence whether procedure-related complications are actually higher with NAAP. However, this speaks to one of the arguments made against NAAP, which is whether the endoscopist can be fully attentive to both performing the endoscopic procedure and administering sedation. Future studies should more consistently assess gastrointestinal perforation and bleeding as part of procedure-related adverse events.
Our secondary outcomes of interest were measures of endoscopy quality and of patient and endoscopist satisfaction. Anesthesiologists administered significantly higher dosages of propofol than did endoscopists. This may partly explain why awareness with recall was less frequent among patients who received AAP in our analysis. It may be that anesthesiologists, in general, are more comfortable attaining deeper levels of sedation. Alternatively, endoscopists may be better attuned than anesthesiologists to the amount of sedation needed relative to their progress in the procedure.18 Awareness of the procedure did not appear to negatively affect patient satisfaction, as measured by 3 patient surveys. In addition, the amount of propofol administered did not seem to affect patient satisfaction or procedure time, both of which were similar between groups.
Other secondary outcomes of interest, which could not be meta-analyzed, were time to recovery and rates of reported pain, cecal intubation and polyp detection. Time to recovery was significantly shorter with NAAP than with AAP in the study by Ferreira and colleagues.26 This could be due simply to the fact that anesthesiologists administered more propofol. Rates of cecal intubation and polyp detection were not different between groups.26 In the study by Poincloux and colleagues,28 a higher proportion of patients in the AAP group than in the NAAP group reported feeling “no pain.” However, this did not appear to necessarily correlate with patient satisfaction, as more patients in the AAP group than in the NAAP group reported that they would not repeat the procedure under the same circumstances.
It is important to highlight that most of the patients in these studies were classified as ASA I or II, and that these results may not be applicable to populations with greater comorbidity; i.e., ASA level III or IV. These patients would likely experience higher rates of cardiovascular complications from any sedation; thus, it is generally preferred that an anesthesiologist attend these cases. Aside from ASA classification, there may be other patient characteristics that can be identified before the procedure to identify those at high risk (e.g., Mallampati score and chronic narcotic use).
Goudra and colleagues18 published a meta-analysis of patient safety with NAAP versus AAP in advanced endoscopic procedures (e.g., endoscopic retrograde cholangiopancreatography, upper endoscopic ultrasonography and endoscopic surgery). Their methods differed slightly in that they included single-arm observational studies and made indirect comparisons between the 2 groups. They concluded that NAAP was safe. An argument made against NAAP is the endoscopist’s presumed relative lack of training and experience in recognizing and managing cardiopulmonary complications from propofol sedation. We conclude that there are currently no strong data to this effect. Even though bradycardia rates were higher with NAAP than with AAP, there is no indication that this resulted in patient harm. Another argument made against NAAP is that it may burden endoscopists and hinder the quality of the procedures they are performing. Goudra and colleagues18 found that endoscopist satisfaction was lower with NAAP than with AAP and hypothesized that this may have been due to endoscopists’ reluctance to target deeper sedation when they are responsible for propofol administration. Endoscopists were more likely to accept suboptimal sedation when they were in charge of propofol administration. Several sedation training programs for endoscopists and nurses have been developed in an attempt to increase comfort achieving deeper levels of sedation safely.8,29 However, endoscopists may also be hesitant to undertake the training and changes in practice needed to adopt NAAP.4 Furthermore, litigation risk continues to pose a major barrier. A survey in the United States showed that 67% of endoscopists cited medicolegal concerns as the main cause for their hesitation to adopt NAAP.12 Questions that need to be further addressed include whether endoscopists in general would be willing to assume this additional responsibility and what kind of training and supports would be required to increase endoscopists’ comfort with administering propofol. Existing programs and guidelines regarding NAAP from other jurisdictions would be logical resources for consideration.8
The benefits of implementing NAAP include cost savings and increased availability of propofol. A US study showed that there was an 18% increase in the cost of a procedure when an anesthesiologist was present.30 Future research should aim to determine what the cost savings may be with the implementation of NAAP, accepting that such savings would vary among different jurisdictions and practice models. Although obviating anesthesiologist fees is a potential advantage of NAAP, one must also take into account the costs of training endoscopists, nurses and other endoscopy suite staff in administering propofol and dealing with complications that may arise.
Limitations
This systematic review has several limitations. The main limitation is that there was substantial inconsistency in the reporting of outcomes among studies. There were few outcomes that were reported in 3 or more studies. Furthermore, for some of the outcomes that were reported, there were ambiguity and variability in their definition. For instance, bradycardia was reported in 3 studies, with 1 defining it as a heart rate less than 50 beats/min,28 another as a decrease in heart rate greater than 25% from baseline, 26 and the third not defining it at all.25 Future studies should aim to use standardized, validated tools of sedation-related complications. Another limitation was the small number of events in each study in spite of large samples. This is because routine endoscopy is a relatively safe procedure, 31 and very large data sets are required to find significant, meaningful differences between interventions. Another explanation for the limited number of events is that most of the studies in our review included mainly patients at low risk. The results of our meta-analysis must be interpreted with caution given that there is a low level of certainty in each of the conclusions. The literature on NAAP is scanty, and, thus, our systematic review was limited to 2 RCTs and 3 observational studies. Nonrandomized studies pose a high risk of selection bias, as patients at lower risk are more likely to have been offered NAAP in these studies. This would inevitably bias the results against AAP and obscure the true measures of effects. However, given the limited evidence on this topic, we were unable to restrict our analysis to RCTs or to conduct meaningful sensitivity analyses. Another major limitation of this review is the low quality of each of the included studies, which contributes a further risk of bias and weakens the robustness of our statistical conclusions. There was also significant heterogeneity among the studies for most outcomes that we reported, which may have been a direct result of the quality of included studies but also of differences in populations.
Conclusion
This meta-analysis provides evidence on the safety of NAAP. We conclude that nonanesthesiologists may safely administer propofol to patients at low risk (ASA I or II) undergoing routine upper or lower endoscopy without substantially compromising the quality of the procedure. Our results must be interpreted with caution given the high risk of bias in the included studies. The results of this systematic review should aid policy-makers seeking strategies that involve task shifting among providers and changing providers’ scope of practice in order to reduce health care costs without compromising patient safety and satisfaction standards. To influence policy, large-scale high-quality studies may be needed to further establish the safety and efficacy of NAAP. Future studies should also examine the cost savings of NAAP, taking into consideration the costs related to implementing this health care arrangement, and potential barriers to its implementation.
Footnotes
Competing interests: None declared.
Contributors: J. Daza, C. Tan, R. Fielding, A. Brown and I. Yang designed the study. J. Daza and C. Tan acquired and analyzed the data, which R. Fielding, F. Farrokhyar and I. Yang also analyzed. All authors wrote and reviewed the article and approved the final version for publication.
- Accepted November 17, 2017.