Abstract
Background: Many patients with end-stage kidney disease (ESKD) have valvular heart disease requiring surgery. The optimal prosthetic valve is not established in this population. We performed a systematic review and meta-analysis to assess outcomes of patients with dialysis-dependent ESKD who received mechanical or bioprosthetic valves.
Methods: We searched Cochrane Central, Medline and Embase from inception to January 2020. We performed screening, full-text assessment, risk of bias and data collection, independently and in duplicate. Data were pooled using a random-effects model.
Results: We identified 28 observational studies (n = 9857 patients, including 6680 with mechanical valves and 3717 with bioprosthetic valves) with a median follow-up of 3.45 years. Twenty-two studies were at high risk of bias and 1 was at critical risk of bias from confounding. Certainty in evidence was very low for all outcomes except bleeding. Mechanical valves were associated with reduced mortality at 30 days (relative risk [RR] 0.79, 95% confidence interval [CI] 0.65–0.97, I2 = 0, absolute effect 27 fewer deaths per 1000) and at 6 or more years (mean 9.7 yr, RR 0.83, 95% CI 0.72–0.96, I2 = 79%, absolute effect 145 fewer deaths per 1000), but increased bleeding (incidence rate ratio [IRR] 2.46, 95% CI 1.41–4.27, I2 = 59%, absolute effect 91 more events per 1000) and stroke (IRR 1.63, 95% CI 1.21–2.20, I2 = 0%, absolute effect 25 more events per 1000).
Conclusion: Mechanical valves were associated with reduced mortality, but increased rate of bleeding and stroke. Given very low certainty for evidence of mortality and stroke outcomes, patients and clinicians may choose prosthetic valves based on factors such as bleeding risk and valve longevity.
Study registration: PROSPERO no. CRD42017081863
In 2010, more than 2.6 million people worldwide were receiving dialysis for end-stage kidney disease (ESKD), a prevalence that is likely to increase to more than 5.4 million people by 2030.1 Cardiovascular disease is the leading cause of death in patients with ESKD, responsible for 39% of deaths.2 One in 3 patients with ESKD has valvular heart disease.3 In this population, valvular heart disease is diagnosed at a rate 4–5 times higher than the general population and progresses at double the rate.2–4
Left untreated, valvular heart disease leads to cardiac dysfunction, heart failure and death.5 Valve replacement can prevent these complications, but the choice of a prosthetic valve for patients with dialysis-dependent ESKD is uncertain. This uncertainty is reflected by the lack of guideline recommendations for valvular heart disease in patients with kidney failure.6–9
Mechanical valves are more durable than bioprosthetic valves but require life-long anticoagulation with vitamin K antagonists.5 Use of vitamin K antagonists in patients on dialysis is associated with a 3- to 10-fold increased risk of bleeding compared with the general population.10,11 Bioprosthetic valves do not require lifelong anticoagulation,5 but are less durable, with case reports describing dysfunction as early as 4 months after surgery.12,13
Four systematic reviews exist but are limited by language restrictions, number of databases searched and narrow search strategies.14–17 Given these limitations, we performed a systematic review and meta-analysis to address the following question: in adult patients with a history of ESKD and chronic dialysis who undergo valve replacement surgery, what is the frequency of major adverse outcomes, comparing mechanical and bioprosthetic valves?
Methods
We registered our protocol on PROSPERO, registration number CRD42017081863.18 Appendix 1, Section 1, available at www.canjsurg.ca/lookup/doi/10.1503/cjs.001121/tab-related-content), presents the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) checklist.
Search strategy
In collaboration with a medical librarian, we developed a broad search strategy (Appendix 1, Section 2). We searched the Cochrane Central Register of Controlled Trials, Medline, and Embase from inception to January 2020, for randomized controlled trials (RCTs) and observational studies. We reviewed trial registries (ISRCTN, World Health Organization International Clinical Trial Registry Platform, clinicaltrials.gov), proceedings of key conferences for the past 2 years (American Heart Association Scientific Session, European Society Cardiology Congress, Canadian Cardiovascular Conference, American Association Thoracic Surgery Annual Meeting, The Society of Thoracic Surgeons Annual Meeting), references of included studies and relevant systematic reviews for eligible studies.
Inclusion criteria
We included RCTs or observational studies that compared outcomes of mechanical or bioprosthetic valves in the aortic or mitral position for patients with dialysis-dependent ESKD. We performed screening of titles and abstracts and of full texts in duplicate and independently using Covidence online software. We recorded reasons for study exclusion after full-text review. Through discussion, reviewers resolved disagreements regarding eligibility, consulting a third reviewer when they could not reach consensus. If all criteria were met for inclusion except for 1, reviewers contacted the corresponding author for further information. If multiple references reported the same outcome from the same cohort, we included only the study with the longest follow-up.
Data collection
Reviewers conducted data extraction independently and in duplicate using Covidence software. We recorded study characteristics, demographic data, details of procedure and outcomes. We resolved disagreements through discussion and, if needed, consulted a third reviewer. For missing data, we contacted corresponding authors twice over a 2-week period, requesting additional information. If we did not receive a response, we deemed the data unavailable.
Outcomes
Outcomes of interest included mortality at 30 days, 1 year, 3 years, 5 years and 6 or more years after surgery; valve-related complications, including valve thrombosis, systemic thromboembolism and valve deterioration; reoperation; major gastrointestinal bleeding; myocardial infarction; postoperative and nongastrointestinal bleeding; stroke (composite of hemorrhagic and ischemic); and health-related quality of life, as reported by any validated instrument. We used indirect evidence to estimate the bleeding risk in patients on dialysis. Appendix 1, Section 3 presents the forest plot for outcomes not reported in results.
Assessment of risk of bias
As our search did not yield eligible RCTs, we discuss only assessment of the risk of bias for observational studies. Two independent reviewers assessed the risk of bias of each included study using the Risk Of Bias In Non-randomized Studies – Interventions (ROBINS-I) tool.19 Reviewers evaluated the risk of bias as either low, moderate, serious or critical for each outcome of interest and study. The ROBINS-I tool includes 7 domains, assessing bias from confounding, selection of participants into the study, classification of interventions, deviations from intended interventions, missing data, measurement of outcomes and selection of the reported results.
Overall risk of bias for each study was low if all domains were rated as low and was moderate if 1 domain was rated as moderate without any other domain deemed as serious or critical. If at least 1 domain was deemed serious, without another other domain deemed critical, the study’s risk of bias was serious. If at least 1 domain was deemed critical, the overall risk of bias was critical.
Assessment of confidence in pooled effect estimates
We used Grading of Recommendations Assessment, Development and Evaluation (GRADE) to evaluate the certainty in evidence.20 We rated each pooled outcome and its body of evidence as high, moderate, low or very low. Outcomes started at high-quality evidence but could be rated down based on the following 5 criteria: limitations in detailed study design and execution (i.e., risk of bias), the applicability of evidence to the patient population of interest (i.e., directness), heterogeneity between study data in the pooled estimate (i.e., inconsistency), confidence intervals of pooled estimates (i.e., imprecision) and publication bias. Quality of evidence could be rated up if there was a dose–response gradient or a large (relative risk [RR] ≥ 2) or very large (RR ≥ 5) effect estimate.
Summary measurement of treatment effect and unit of analysis
We analyzed data using Review Manager version 5.3 (RevMan5.3) and R Studio. We examined the clinical and methodological heterogeneity to ensure pooling data were appropriate. Because of variability between studies, we used a random-effects model and weighted studies using the DerSimonian and Laird inverse-variance method.21 We also used a mixed-effects Poisson regression model, using the metafor package in R Studio, to pool outcomes at latest follow-up to address variability in study duration.22,23 We included studies with outcomes of 0 in both treatment arms in the meta-analysis using the meta package in R Studio. This package uses the continuity correction to estimate individual study outcomes with confidence intervals, and to conduct meta-analysis based on the inverse-variance method. We also used this package to conduct meta-regression, examining the duration of follow-up as a predictor of mortality. All outcomes are dichotomous and presented as an RR or incidence rate ratio (IRR) with 95% confidence intervals (CIs).
Assessment of heterogeneity
We assessed heterogeneity by inspecting the point estimates and confidence intervals in a forest plot. We also used the χ2 test for homogeneity and the I2 index.
Where possible, we performed subgroup analyses to explain observed heterogeneity. These included analysis of studies from Asia versus North America, hypothesizing that studies from Asia would favour mechanical (v. bioprosthetic) valve owing to longer life expectancy on dialysis;24 studies evaluating long-term risk of death in patients receiving peritoneal dialysis versus hemodialysis, hypothesizing that patients selected for peritoneal dialysis would be earlier in the ESKD disease process, leading to longer life expectancy on average than patients receiving hemodialysis;25–28 patients receiving mitral versus aortic valve replacement, hypothesizing that mitral valve replacement would benefit more from mechanical prostheses owing to accelerated bioprosthesis deterioration in the mitral position; patients undergoing valve replacement only versus valve replacement with concomitant procedure, hypothesizing that patients undergoing concomitant procedure would benefit from the bioprosthetic valve owing to greater comorbidity; patients younger than 50 years versus those 50 years and older, hypothesizing that patients younger than 50 years would benefit from mechanical valves, owing to accelerated valve calcification and longer life expectancy; diabetic status, hypothesizing that patients without diabetes would benefit from mechanical valve owing to fewer comorbidities and complications; studies at low risk of bias versus serious risk of bias, hypothesizing that studies at serious risk of bias would report larger effect sizes.
Publication bias
We assessed outcomes pooled from 10 or more studies for publication bias using funnel plots generated by RevMan 5.3. We confirmed bias using the arcsine test. Appendix 1, Section 3, presents funnel plots.
Results
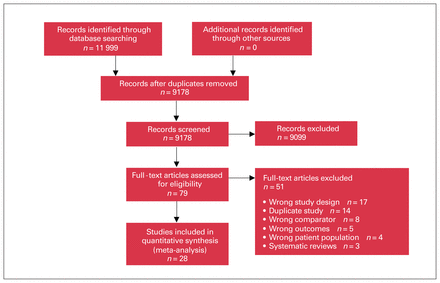
We screened 9178 references, yielding no RCT but 28 observational studies with a total of 9857 patients, including 6680 with mechanical valves and 3717 with bioprosthetic valves (Figure 1). Follow-up ranged from 30 days to 12 years (median 3.45 years).29–57 Six studies included only aortic valves.30,32,38,40,41,53 One study included patients with moderate-to-severe renal failure, some not requiring dialysis.30 We included this study as most patients (70%) received dialysis preoperation. We included 2 studies published in Japanese and 2 studies published as abstracts.41,49–51 No studies reported functional capacity or health-related quality of life. We emailed 25 authors requesting missing data or additional information. We received 3 initial replies, but none to subsequent emails and deemed the data unavailable. Table 1 presents patient characteristics of included studies and Appendix 1, Supplemental Table S1, presents other study characteristics. We pooled baseline patient characteristics (Table 1) using weighted means.
Prisma flow diagram.
Patient characteristics of included studies
Five studies were considered at moderate risk of bias owing to selection of reported results; they lacked a preregistered protocol or statistical analysis plan.29,41,52 Twenty-two studies were at serious risk of bias owing to confounding; statistical adjustment was nonexistent or insufficient. One study was at critical risk of bias as the authors did not report baseline variables, making controlling confounding infeasible.49 We could not conduct a sensitivity analysis because of the overall serious risk of bias of included studies. We conducted post hoc sensitivity analyses for key outcomes based on year of publication to explore timebias, comparing effect estimates of studies published 2010 and earlier to studies published after 2010. We detected no difference based on year of publication for mortality at 30 days, mortality at 6 or more years, stroke and reoperation. We also conducted post hoc sensitivity analyses for these key outcomes based on risk of bias (moderate v. high). We found a significant difference for mortality at 6 or more years (p for interaction = 0.01), but no difference for others. Appendix 1 presents a risk of bias table for each study, and a summary table of our post hoc sensitivity analysis (Appendix 1, Supplemental Tables S2, S6, S7). We detected asymmetry in funnel plots for all outcomes and further examined possible publication bias using the arcsine test. Results of the arcsine test are reported for each outcome.
Mortality
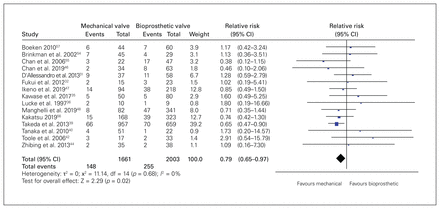
Fifteen studies (n = 3664 patients) reported 30-day mortality. Mortality was 8.9% (148/1661) in the mechanical group and was 12.7% (255/2003) in the bioprosthetic group (RR 0.79, 95% CI 0.65–0.97, p = 0.02, I2 = 0%, very low quality) (Figure 2). We rated down the quality of evidence for very serious risk of bias and imprecision. On visual inspection, we suspected publication bias, but the arcsine test did not confirm this (p = 0.06).
Forest plot for 30-day mortality. Note: CI = confidence interval.
Twenty studies (n = 8274 patients) reported 1-year mortality. Mortality in the mechanical group was 42.8% (2508/5856) and was 35.9% (868/2418) in the bioprosthetic group (RR 0.97, 95% CI 0.83–1.12, p = 0.67, I2 = 31%, very low quality). We rated down the quality of evidence for very serious risk of bias and imprecision. The arcsine test did not detect publication bias (p = 0.97).
Nineteen studies (n = 8187 patients) reported 3-year mortality, suggesting no significant difference (RR 0.97, 95% CI 0.90–1.06, p = 0.52, I2 = 17%, very low quality). We rated down the quality of evidence for risk of bias and imprecision. The arcsine test did not detect publication bias (p = 0.60).
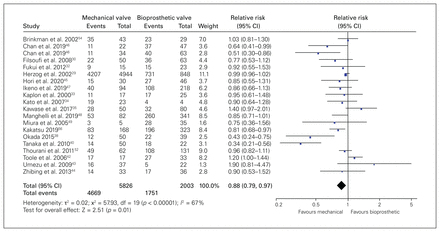
Twenty studies (n = 8254 patients) reported 5-year mortality; it was 80.1% (4669/5826) with mechanical valves and 72.1% (1751/2428) with bioprosthetic valves (RR 0.88, 95% CI 0.79–0.97, p = 0.01, I2 = 67%, very low quality) (Figure 3). We rated down the quality of evidence for risk of bias, inconsistency and imprecision. We did not detect publication bias using the arcsine test (p = 0.20).
Forest plot for 5-year mortality. Note: CI = confidence interval.
Ten studies (n = 6369 patients) reported mortality at 6 or more years (mean 9.7 yr); mortality was 92.7% (4972/5361) with mechanical valves and 85.3% (1214/1423) with bioprostheses (RR 0.83, 95% CI 0.72–0.96, p = 0.01, I2 = 79%, very low quality). We rated down the quality of evidence for very serious risk of bias and inconsistency. We suspected publication bias from the funnel plot; smaller studies favouring bioprosthetic valves were missing. We confirmed publication bias using the arcsine test (p = 0.02). We conducted a meta-regression to assess duration of follow-up as a predictor of mortality; it was not significant (β = −0.0001, p = 0.98). Appendix 1, Supplemental Figure 1 presents the plot of the meta-regression. We also conducted a post hoc sensitivity analysis based on risk of bias. Comparing studies at moderate risk of bias to those at high risk of bias, we found no significant difference between mechanical and bioprosthetic valves for mortality at 6 or more years (RR 1.01, 95% CI 0.99–1.03, p = 0.22, I2 = 0%).
Postoperative and nongastrointestinal bleeding at latest follow-up
Sixteen studies (n = 3548 patients) reported on bleeding (median follow-up 2.8 yr), occurring in 8.7% (144/1650) of patients in the mechanical group and 6.2% (118/1898) of patients in the bioprosthetic group (IRR 2.46, 95% CI 1.41–4.27, p < 0.01, I2 = 59%, very low quality). Small studies favouring mechanical valves were missing in the funnel plot, creating asymmetry. The arcsine test confirmed publication bias (p = 0.001). We rated down the quality of evidence for serious risk of bias and publication bias.
Given the very low certainty in evidence among patients with dialysis-dependent ESKD, we conducted a meta-analysis, whereby we pooled bleeding reported from 5 RCTs (n = 2786 patients) comparing warfarin to placebo in the general population (IRR 2.99, 95% CI 1.46–6.13, p < 0.01, I2 = 74%, moderate quality).58–62 We rated down the quality of evidence for serious indirectness. Appendix 1 details how we assessed the risk of bias for the 5 RCTs (Appendix 1, Supplemental Table S3).
Gastrointestinal bleeding at latest follow-up
We pooled 11 studies (n = 2475 patients) that reported gastrointestinal bleeding (median follow-up 3.2 yr). Gastrointestinal bleeding occurred in 6.4% (84/1314) of patients with mechanical valves and 6.3% (73/1161) of patients with bioprosthetic valves (IRR 1.45, 95% CI 0.79–2.64, p = 0.23, I2 = 27%, very low quality). We rated down the quality of evidence owing to risk of bias and imprecision. The arcsine test confirmed publication bias (p = 0.03).
Stroke
Seventeen studies (n = 2870 patients) reported stroke at a median of 2.6 years; it occurred more frequently in patients with mechanical valves (6.0%, 103/1718) than in those with bioprosthetic valves (3.9%, 81/2060) for an IRR of 1.63 (95% CI 1.21–2.20, p < 0.01, I2 = 0%, very low quality). We rated down the quality of evidence for serious risk of bias and publication bias (p = 0.01).
Reoperation
Sixteen studies (n = 3602 patients) reported on reoperation at a median 3.6 years of follow-up. The rate of reoperation with mechanical valves was 5.7% (93/1627) and was 4.8% (95/1975) with bioprosthetic valves (IRR 0.93, 95% CI 0.69–1.26, p = 0.65, I2 = 0%, very low quality). The arcsine test did not detect publication bias (p = 0.90). We rated down the quality of evidence for risk of bias and imprecision.
Valve-related complications
Table 2 summarizes the results for valve-related complications. We found significant difference for myocardial infarction and systemic thromboembolism in favour of the bioprosthetic valve, and no significant difference for endocarditis, valve thrombosis, valve deterioration and a composite of valve-related complications (i.e., valve thrombosis, systemic thromboembolism and valve deterioration). The quality of evidence for all outcomes was very low. Appendix 1, Section 3, includes the forest plots and funnel plots for each outcome within valve-related complications. Appendix 1, Supplemental Tables S4 and S5, include the GRADE summary of findings for all outcomes in this review.
Meta-analysis of valve-related complications (bioprosthetic valve as reference)
Discussion
In patients with dialysis-dependent ESKD who underwent valve replacement, the current evidence provides little confidence in differentiating outcomes between bioprosthetic and mechanical valves, though risk of death appears lower in patients who receive mechanical valves. Measuring mortality at 30 days, 27 (95% CI 4–45) fewer deaths per 1000 occurred in patients with mechanical valves than in those with bioprosthetic valves. At follow-up approaching 10 years, 145 (95% CI 34–239) fewer deaths per 1000 occurred in patients with mechanical valves than in those with bioprosthetic valves. However, mechanical valves are associated with significantly increased rates of stroke and bleeding. The certainty of evidence ranged from very low to moderate; all outcomes from direct evidence were rated down for very serious risk of bias and imprecision.
We found 4 previously published systematic review and meta-analyses on the topic. The most recent systematic review found a long-term survival benefit for patients who received mechanical valves, similar to findings in this review.17 Despite this similarity, our review has several strengths. Our review includes 4 additional studies than the most recent review. We attribute this to our comprehensive search strategy, allowing the inclusion of 2 studies in Japanese and conference abstracts.41,49–51 We report mortality at specific time points rather than early or late mortality.14–17 Specific time points are critical for 2 reasons. First, prognosis in patients with dialysis-dependent ESKD can vary widely depending on variables not measured (e.g., kidney transplantation) and, second, the length of follow-up between studies varies greatly (30 d to 12 yr).
We report pooled analysis of stroke and gastrointestinal bleeding. Both of these outcomes are important in all patients starting vitamin K antagonist therapy but are especially important in patients with ESKD who are at an increased risk of bleeding at baseline. We also provide a comprehensive estimate of valve deterioration. Prevous reviews have qualitatively described valve deterioration and the most recent review pooled 2 studies.14–17 We pooled 13 studies using the continuity correction, which allowed us to include studies with 0 outcomes in both groups.63 Finally, we applied the GRADE framework to provide clinical practice recommendations for choosing a prosthetic valve for patients with dialysis-dependent ESKD. With the GRADE framework, we used indirect evidence to evaluate risk of bleeding with greater certainty. We identified 5 RCTs from a systematic review that compared warfarin to placebo in the general population.64 We believe that the pooled effect estimate underestimates the true risk of bleeding in patients with dialysis-dependent ESKD, who are at an increased risk of bleeding than the general population. Therefore, we rated down for serious indirectness.
This systematic review shows the paucity of evidence to guide clinicians who must choose a prosthetic valve for patients with dialysis-dependent ESKD. These patients were excluded in previous RCTs that compared outcomes of mechanical and bioprosthetic valves.65–67 Most included studies were underpowered and presented unadjusted results.
Although we found a significantly lower mortality on short- and long-term follow-up with mechanical valves, we did not find an RCT that informed the question. Most included studies did not address confounding or did not appropriately adjust for known confounding variables. This prevents causal inferences; differences in outcomes may be owing to differences in baseline characteristics and residual confounding. For example, bioprosthetic valves may more likely be selected for frail patients with lower life expectancies, while mechanical prostheses may more likely be selected for patients with better overall health or who are awaiting renal transplant, which significantly improves life expectancy.68 Despite this, age and comorbidities did not differ substantially between recipients of bioprosthetic and mechanical valve in this review.
Lower mortality with mechanical valves may also be attributed to survivor bias. Traditionally, structural valve deterioration is defined as a requirement for reoperation.69,70 Patients on dialysis may not be offered reoperation owing to their comorbidities. Among the bioprosthetic valve group, this may result in increased risk of death, decreased reoperation rates and decreased reporting of structural valve deterioration. Studies included in this systematic review reported reoperation, with a median of 3.6 years of follow-up. This length of follow-up is likely insufficient to produce a meaningful result. Another key issue with the literature is publication bias. We suspected publication bias when inspecting funnel plots of 30-day mortality, 1-year mortality, 3-year mortality, 5-year mortality, gastrointestinal bleeding, stroke, postoperative bleeding and endocarditis. We showed publication bias with the arcsine test for gastrointestinal bleeding, stroke and post-operative bleeding. For these outcomes, small studies favouring mechanical valves were missing.
In light of our results, variability in physician practice is expected. Large observational studies with appropriate adjustment or RCTs are required to inform practice. Until higher quality evidence is available, prosthesis choice should be based on discussions of the pros and cons of each prosthetic valve with the patients as well as their values and preferences.
Limitations
We used PlotDigitizer software to extract mortality at specific time points from survival curves. We calculated the proportion deceased from the difference of those who survived at a specific time point and the total sample, assuming that patients on dialysis were unlikely to be lost to follow-up after surgery. We also included studies with 0 events in both groups in the meta-analysis of all outcomes providing conservative and generalizable estimates of infrequent outcomes.6 We used the ROBINS-I tool for transparent and detailed risk of bias assessment19 and GRADE to assess the quality of evidence.20 Despite these strengths, this study also has several limitations. We identified only observational studies with substantial confounding, resulting in very low–to-low quality of evidence. We were also unable to conduct sensitivity and subgroup analyses because of the high risk of bias and lack of reported characteristics. We contacted authors of studies with missing data but were unable to gain additional data needed to perform subgroup analyses. Patients in this review may not reflect the general dialysis population. The average duration of preoperative dialysis was 10 years, but the average 5-year survival rate for ESKD ranges from 42% to 52%.2 Limited data exist to inform the durability of the bioprosthetic valve in the dialysis population. A disadvantage of the bioprosthetic valve in the general population is durability at 10 years. With a median follow-up of 3.45 years, we cannot reliably assess durability in the dialysis population.
Conclusion
Based on very low-quality evidence, risk of death is lower with mechanical valves, but at the cost of increased risk of bleeding and stroke. Residual confounding related to selection bias may account for the association between mechanical valves and death. To further inform clinical practice, future studies should be large enough to allow for adjusted analyses and to generate narrow confidence intervals. Until high-quality evidence is generated, patient values and preferences should guide decisions.
Footnotes
This work was presented at the annual meeting of the European Association for Cardio-Thoracic Surgery, Lisbon, Portugal, Oct. 3–5, 2019.
Competing interests: Emilie Belley-Côté reports grants Roche, Bayer and Bristol-Myers Squibb-Pfizer, and is chair of the data safety monitoring board of the BEEF-BURGER trial. Matthias Bossard reports consulting fees from Abbott Vascular, Amgen, AstraZeneca, Daichii and Vifor. He also reports speaker fees from Abbott Vascular, Amgen, AstraZeneca, Mundipharma, Bayer, Daichii and Vifor. Richard Whitlock reports grants from Boehringer Ingelheim, Bayer and Abbott; consulting fees from Phasebio, Atricure, Cryolife; and participation on a data safety monitoring board with Phasebio. No other competing interests were declared.
Contributors: Kevin Kim, Emilie Belley-Côté, Gordon Guyatt and Richard Whitlock contributed to the conception and design of the work. Kevin Kim, Saurabh Gupta, Arjun Pandey, Ali Alsagheir, Ahmad Makhdoum, Graham McClure, Brooke Newsome, Matthias Bossard, Tetsuya Isayama, Yasuhisa Ikuta, Michael Walsh, Amit Garg and Gordon Guyatt contributed to the acquisition, analysis, and interpretation of data. Kevin Kim, Arjun Pandey, Sophie Gao, Gordon Guyatt and Richard Whitlock drafted the manuscript. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This study did not receive any funding or financial support. Emilie Belley-Côté is supported by a new investigator award from the Heart and Stroke Foundation of Canada and by the E. J. Moran Campbell internal medicine career award. Amit Garg is supported by the Dr. Adam Linton Chair in Kidney Health Analytics and a Clinician Investigator Award fom the Canadian Institutes of Health Research. Richard Whitlock is supported by the Canada Research Chair in Cardiovascular Surgery from the Canadian Insitutes of Health Research.
- Accepted September 12, 2021.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/